 Throughout history there have been seminal moments in time when humankind has been forced to rethink commonly accepted ways of doing things, as well as the beliefs that have held them in place.
Throughout history there have been seminal moments in time when humankind has been forced to rethink commonly accepted ways of doing things, as well as the beliefs that have held them in place.
In many cases these pivots were brought about by milestone events that forced the reexamination of standard operating procedures. When Magellan circumnavigated the globe, proving that the world was not flat, he changed maritime strategy. Issac Newton’s groundbreaking work on the laws of motion and gravity formed the basis for modern physics. Wilbur and Orville Wright’s first powered, sustained airplane flight altered transportation.
The life sciences industry is currently experiencing such a seminal moment. Last year the timeframes we associated with drug development were completely disrupted and redefined. The global pandemic forced our industry to abandon passive acceptance of the decade-long timeline that had been the standard for bringing new drugs to market.
Out of sheer necessity we broke free from traditional processes and procedures to embrace and implement exciting new technology solutions that enabled the slingshotting of new therapies from lab to market in less than a year. The concept and practice of how clinical  data is managed, from the front end of the research and development process to the back end of putting new therapies in the hands of healthcare professionals, underwent a dramatic transformation – one that raised the bar and established a new standard for every clinical development effort to come.
data is managed, from the front end of the research and development process to the back end of putting new therapies in the hands of healthcare professionals, underwent a dramatic transformation – one that raised the bar and established a new standard for every clinical development effort to come.
AI-powered clinical data management solutions substantially contributed as a change agent towards accelerated vaccine and therapeutic development for COVID-19. Now we are charged with ensuring the ongoing integration of this exciting technology with industry’s business strategies in order to continue and sustain the innovation across newer drug development.
AI-based technology solutions are empowering the life sciences industry to achieve this goal in a ubiquitous and permanent way through what I call the “three Cs"— connect, contextualize, and converse with clinical trial data.
Connect
Connection is all about how the clinical data assets that are needed, either for new study planning and design or during study conduct, are integrated, cleaned, harmonized, and mapped. This process of data management is some of the most expensive, resource- intensive, time-consuming and error-prone work that the industry undertakes in relation to every study. Thousands of highly qualified data managers across the pharma and CRO ecosystem traditionally spend countless hours ensuring the highest data quality for the most accurate results.
intensive, time-consuming and error-prone work that the industry undertakes in relation to every study. Thousands of highly qualified data managers across the pharma and CRO ecosystem traditionally spend countless hours ensuring the highest data quality for the most accurate results.
Now, thanks to deep learning and other advances in AI technology, we are realizing a huge change in this clinical data management area. Technology and AI modeling now enable machines to understand the data patterns and intelligent overrides that the domain experts usually enforce, as well as integrate continuous learning models into the business process.
With these AI technology solutions pharma can predict data queries that require critical data changes. The industry leveraged such a smart data query mechanism in 2020 to process more than 100 million data points every day for a 47,000-person COVID-19 vaccine trial. Such an AI system is clearly at the intersection of business and technology, eliminating potentially millions of dollars in wasteful expenses, compressing the hours traditionally needed to manage data, and allowing for consistent results across studies.
Contextualize
The ability to contextualize clinical trial data ensures that researchers, including study managers, CRAs, data monitors, portfolio execs, protocol designers, and the hundreds of other clinical researchers who do the heavy lifting daily, can get the insights they need to prioritize and resolve critical data gaps in real time. Sponsors, CROs, and patients alike benefit from intelligent analytics solutions that capture AEs, alert researchers when target thresholds are breached, and contextualize and curate information to ensure that the right patients are prioritized. The smart data query system used in the COVID-19 vaccine trial mentioned previously was also able to isolate 0.2% of most relevant safety and efficacy data in the trial, ensuring analysis-ready data in only hours instead of days. Smart systems such as this fuel positive disruption and drive collaborative workflows through visibility and information sharing across study stakeholders to optimize innovation and results.
Converse
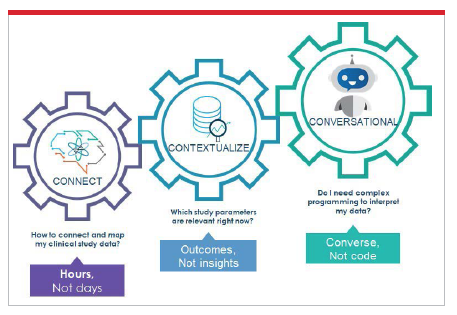 The ability of AI-based virtual assistants to facilitate conversation with clinical data empowers pharmaceutical and biotech companies to address many of the business challenges common to the drug developmental life cycle, bringing increased efficiencies and cost savings to critical outcomes such as patient recruitment, protocol adherence, prediction of study success, continuous process improvement, timely, and accurate analytics insight, patient data privacy, and the ability to leverage previously untapped sources of data.
The ability of AI-based virtual assistants to facilitate conversation with clinical data empowers pharmaceutical and biotech companies to address many of the business challenges common to the drug developmental life cycle, bringing increased efficiencies and cost savings to critical outcomes such as patient recruitment, protocol adherence, prediction of study success, continuous process improvement, timely, and accurate analytics insight, patient data privacy, and the ability to leverage previously untapped sources of data.
Virtual assistants have re-architected the human-computer interaction paradigm. They provide easy-to-use, context and domain-aware conversational experiences, as opposed to the traditional user experience. They can also identify the intent of a query, pinpointing what a researcher would like to do or know. When replying to questions about study conduct, whether from an operational or clinical perspective, AI virtual assistants factor in key parameters, such as the names of persons, organizations, and locations, as well as expressions of times, quantities, monetary values and percentages. Virtual assistants remember the context of previous inquiries and can seamlessly enfold new entities into the discussion to provide rapid clinical operations insights. Queries about various aspects of clinical development, including start-up, enrollment, data quality, and financial risk, result in responses that factor in the intent and specificity of the questions.
This level of precision and penetration enables researchers to gain key data and insights that alleviate the planning, feasibility and conduct challenges inherent in and pervasive throughout the drug development continuum. Clinical experts’ valuable time need not be wasted on a majority of questions to which these virtual assistants can respond.
The Impact
AI-powered technology solutions are irrevocably altering and improving every step of the clinical trials process. They are also offering comprehensive support and unparalleled insights to stakeholders in both the upstream and downstream segments of biopharma. This intelligence is achieving a previously unrealized level of interconnectivity and can be leveraged:
To prevent default to pre-pandemic drug development timelines and standards
To radically improve clinical trial management and execution via clinical intelligence
For data aggregation and automation via smart applications
For better decision-making, fewer delays, reduced drug development costs
The results include improved:
Drug development timelines
Risk management
Performance across clinical studies, systems, sites, and vendors
Collaboration among clinical operations, medical review, data management, biostatistics, and pharmacovigilance teams
Enterprise and industry digital transformation via AI-powered data analytics solutions is upon us. Sponsors and CROs that fail to adopt a technology transformation strategy for drug development will fall behind in the post-pandemic era. Emerging technologies, such as deep learning-based smart data query platforms need to be a core part of every life-science company’s fundamental business strategy. The past two years provided the tailwind for accelerating adoption of AI-powered technology solutions to make fast-tracking of novel therapies the norm, not the exception.(PV)
Saama builds long-lasting relationships with the most respected pharmaceutical companies, nonprofit organizations, universities, and government agencies in the world, by delivering game-changing solutions that improve clinical trial outcomes.
For more information, visit saama.com.
















