Nanomedicine Zeros In On Triple Negative Breast Cancer Cells
Trend Watch: From Breast Cancer to Asthma, Healthtech Tools Develop Care Solutions
 Researchers at George Washington University have shown that a technique of delivering a chemotherapy agent within specially designed nanoparticles can be powerful against triple negative breast cancers. The cells of triple negative breast cancer tumors are difficult to treat because they don’t have receptors for estrogen, progesterone, and HER2 — the main targets used to attack breast cancers.
Researchers at George Washington University have shown that a technique of delivering a chemotherapy agent within specially designed nanoparticles can be powerful against triple negative breast cancers. The cells of triple negative breast cancer tumors are difficult to treat because they don’t have receptors for estrogen, progesterone, and HER2 — the main targets used to attack breast cancers.
The study, published in the journal Precision Medicine, discovered that the smallest nanoparticles that are designed to have the slowest release of doxorubicin, the drug that was studied, had the most powerful killing effect. Next, the nanoparticles will have to be tried in lab animals before going further into clinical trials, which will surely take quite a few years.
“Nanomedicine is a very exciting avenue in modern drug development," says Adam Friedman, M.D., director of the Supportive Oncodermatology Clinic at GW Cancer Center, professor of dermatology at the GW School of Medicine and Health Sciences, and senior author of the study.“This study provides clues for new potential strategies utilizing and manipulating nanotechnology to overcome cancer cell drug resistance. We have our work cut out for us, but this study shows that we are moving in the right direction."
Roche Integrates Insulin Data from Novo Nordisk with Diabetes Management Solutions
 Roche has signed a development and collaboration agreement with Novo Nordisk, a leader in innovating the therapy of diabetes and obesity. Under the terms of the development and collaboration agreement, the partners will integrate insulin data from Novo Nordisk’s connected pen to Roche’s open ecosystem, synchronizing with its suite of digital diabetes management solutions including mySugr.
Roche has signed a development and collaboration agreement with Novo Nordisk, a leader in innovating the therapy of diabetes and obesity. Under the terms of the development and collaboration agreement, the partners will integrate insulin data from Novo Nordisk’s connected pen to Roche’s open ecosystem, synchronizing with its suite of digital diabetes management solutions including mySugr.
“We believe in the tremendous benefits integrated digital diabetes management solutions can bring to people with diabetes, caregivers and healthcare systems as part of an open ecosystem and are excited to partner with Novo Nordisk to further drive innovation in this area," says Marcel Gmuender, global head of Roche Diabetes Care.
“The integration of insulin pen data in our digital health solutions such as mySugr will make it much easier for people with diabetes and their caregivers to track the effect of insulin on blood glucose levels. This enables more efficient and targeted decision support, as they can act on near real-time insights to optimize the personalized diabetes management thereby reducing the risk of costly secondary complications and contributing to improved therapy outcomes and better quality of life."
Asthma Health Tech Tools Gain Traction
Geisinger and AstraZeneca have partnered to improve asthma care by creating a suite of products that integrate into the electronic health record (EHR). The goal is to engage patients and providers in co-managing the disease, especially during intervals between office visits.
The mobile app data is connected to a real-time, Web-based application called Provider Asthma Management Assistant. The assistant combines EHR and patient-reported data feeds to enable asthma educators and respiratory therapists to triage patients on the fly and bring in specialists as needed.
The new asthma app suite includes a mobile tool that allows patients to view asthma-related weather forecasts, log symptoms and asthma triggers, establish medication reminders, track health status over time, and, crucially, communicate with their healthcare team.
In other efforts, an LA Project uses mHealth devices to help low-income kids with asthma. Backed by a $250,000 grant, SmartAirLA will be launching an mHealth project aimed at improving care management and coordination for low-income children living with asthma. Through the grant, the not-for-profit health plan and philanthropic organization Blue Shield of California Foundation is providing smart inhalers to low-income children living with asthma. The connected health project aims to give those children and their caregivers a telehealth link to improve care management and coordination.
Also taking part in the program is Adherium, a Silicon Valley-based developer of digital health tools for medication adherence. Participants in the program will receive the company’s Hailie sensor, which attaches to an inhaler, and accompanying mHealth app platform, available on both Android and iOS.
Asthma is also a chronic condition that’s well-suited for mHealth and telehealth intervention. Studies have shown that smart inhalers, which track medication use and connect users with their care providers, can reduce hospitalizations caused by poor asthma control and improve care management and coordination.
DeepMind Hopes to Train Algorithms to Spot Breast Cancer
Earlier this year, DeepMind joined a groundbreaking research partnership led by the Cancer Research UK Imperial Centre at Imperial College London to explore whether AI technology could help clinicians diagnose breast cancers on mammograms quicker and more effectively.
As a result of this effort, the project is expanding internationally, with The Jikei University Hospital, one of Japan’s foremost medical institutions, joining the collaboration as part of a wider five-year partnership it signed with DeepMind Health.
For the purposes of this research, researchers will study historic, de-identified mammograms from around 30,000 women taken at the hospital between 2007 and 2018. These will be analyzed with AI technology alongside the historic de-identified mammograms already provided by the UK OPTIMAM mammography database in order to investigate whether the technology can spot signs of cancerous tissue on these X-rays more effectively than current screening techniques allow. De-identified breast ultrasounds from around 30,000 women and 3,500 de-identified breast MRI scans will also be shared by The Jikei University Hospital during the course of the project.
Working with partners and data from multiple countries in a single project is a first for DeepMind Health. The company hopes doing so will help it work toward its ambition to create technology that works for everyone around the world, because it will help minimize bias.
Professor Ara Darzi, director of the Cancer Research UK Imperial Centre, says: “It’s early days for this work, but we’re optimistic about the long-term potential for AI technology in this area and hope, in time, to explore how it could help in the analysis of other forms of breast imaging. It’s a hugely exciting opportunity to make a difference to breast cancer treatment across the world."
Vagus Nerve Stimulator Doubles Movement Recovery in Stroke Patients
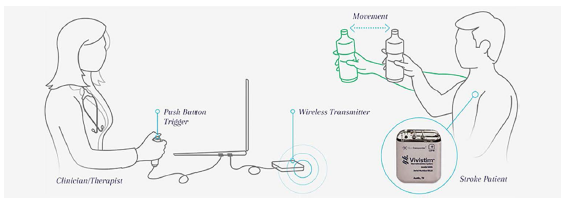 Researchers at the University of Texas at Dallas have developed and helped to commercialize a vagus nerve stimulator therapy, which significantly enhanced — as in doubled — movement recovery in stroke patients undergoing rehab in a recent study.
Researchers at the University of Texas at Dallas have developed and helped to commercialize a vagus nerve stimulator therapy, which significantly enhanced — as in doubled — movement recovery in stroke patients undergoing rehab in a recent study.
The implant is based on the principle that synchronizing vagus nerve stimulation with movement increases neural plasticity in the brain, resulting in enhanced recovery.
This research team developed a device, called the Vivistim, which can be implanted to stimulate the vagus nerve in the neck, which controls the parasympathetic nervous system. They paired the vagal nerve stimulation with traditional physical rehab, and precisely timed the nerve stimulation to occur during movements of the affected limb.
The clinical trial included 17 people across the country who had a stroke between four months and five years prior to selection. After a VNS device was implanted, they received six weeks of in-clinic rehab followed by a home exercise program.
About half were treated with active VNS and the rest received control VNS.
The researchers found that those receiving active VNS scored more than twice as high as control subjects at the 30- and 90-day intervals.
Researchers are currently recruiting up to 120 participants in 15 cities for a more extensive trial.
~~~~~~~~~~~~~~~~~~~~~~~~~
STATEMENT OF OWNERSHIP, MANAGEMENT, AND CIRCULATION
1. Publication title: PharmaVOICE
2. Publication no.: #23626
3. Filing date: September 26, 2018
4. Issue frequency: published monthly except combined issues in July/August and Nov./Dec.
5. No. of issues published annually: 10
6. Annual subscription price: US: $190.
Non-US: $360.
7. Complete mailing address of known office of publication: PharmaLinx LLC, 244 Jacobs Creek Road, Titusville, NJ 08560, Titusville, NJ 08560
8. Complete mailing address of headquarters or general business office of publisher: PharmaLinx LLC, 244 Jacobs Creek Road, Titusville, NJ 08560, Titusville, NJ 08560
9. Full names and complete mailing addresses of publisher, editor, and managing editor:
Lisa Banket, P.O. Box 327, Titusville, NJ 08560, Taren Grom, P.O. Box 327, Titusville, NJ 08560, and Denise Myshko, P.O. Box 327, Titusville, NJ 08560
10. Owner: Lisa Banket, Taren Grom, and
Marah Walsh
11. Known bondholders, mortgagees, and other security holders owning or holding 1 percent or more of total amount of bonds, mortgages, or other securities: (none)
13. Publication title: PharmaVOICE
14. Issue date for circulation data below:
September 2018
15. Extent and nature of circulation (avg. no. copies each issue during preceeding 12 months and actual no. copies of single issue published nearest to filing date, respectively). Total no. copies – 17,939 and 17,900. Individual paid/requested mail subscriptions stated on Form 3541: 17,562 and 17,565. Sales through dealers and carriers, street vendors, counter sales, and other paid or requested distribution outside USPS: 21 and 0. Total paid and/or requested circulation: 17,583 and 17,565. Nonrequested copies stated on Form 3541: 0 and 0.
Free or nominal rate distributed outside the mail: 49 and 38. Total free or nominal rate distributed: 93 and 50. Total distribution: 17,725 and 17,653. Copies not distributed: 214 and 247. Total: 17,939 and 17,900. Percent paid and/or requested circulation: 99.20 and 99.50.
16. This statement of ownership will be printed in the November/December 2018 issue of this publication.
17. I certify that all information furnished on this form is true and complete. I understand that anyone who furnishes false or misleading information on this form or who omits material or information on the form may be subject to criminal sanctions (including fines and imprisonment) and/or civil sanctions (including civil penalties).
(Signed) Marah Walsh, Founding Partner
















