Clinical trials and traditional marketing efforts are no longer the only way to reach today’s patient consumer. To succeed in the era of digital and health savvy consumers, the industry needs to enlist a more proactive patient engagement strategy that anticipates and meets the evolving needs and wants of patients both in clinical trials and out. Pharmaceutical companies can create more value  for consumers by using advanced technology and partnerships that lead to more innovative ways to engage them.
for consumers by using advanced technology and partnerships that lead to more innovative ways to engage them.
Our experts explore various avenues for engagement, such as improving health literacy, capitalizing on retail pharmacy opportunities, gaining deeper insights from more available data, and creating more outcomes based relationships, all with a direct focus on patient-centricity.
“There’s a long history of looking to engage the patient from the pharma perspective, during the development process, as well as once a drug is commercialized," says Karla Anderson, partner, PwC. “However, there’s a variety of factors that are now making that engagement more important, more effective, and more precise."
According to a PwC report, as the healthcare ecosystem continues to evolve, new trends and players are emerging and existing players are adapting. These changes, and three in particular, are creating a need to engage with patients and consumers in new ways. These forces — the emergence of consumerism, the shift within healthcare from volume to value, and regulatory interest in patient perspectives — increase the importance for pharma companies to participate in patient engagement.
“The pharma industry is beginning to change who and how it targets consumers," says Jeff Lee, founder and CEO of mProve Health. “As recently as 10 years ago, those targets were physicians. With the age of the Internet, consumers have a greater voice in their healthcare decision-making."
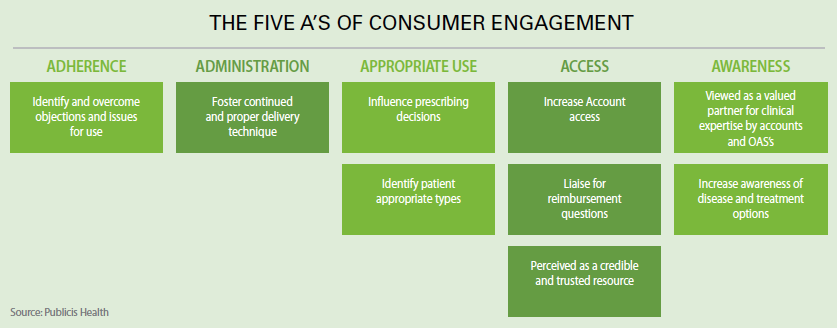 Ms. Anderson agrees, saying patients now have more financial responsibility for their healthcare and have more health information available to them, and this has empowered them. Marketers also can be empowered through the use of technology that enables better patient data collection, allowing them to have a better understanding on how patients behave and the role that the patients’ level of motivation and ability to engage in their therapy plays in successful outcomes.
Ms. Anderson agrees, saying patients now have more financial responsibility for their healthcare and have more health information available to them, and this has empowered them. Marketers also can be empowered through the use of technology that enables better patient data collection, allowing them to have a better understanding on how patients behave and the role that the patients’ level of motivation and ability to engage in their therapy plays in successful outcomes.
“With such a major shift toward outcomes and value in the healthcare arena overall there’s a high recognition that the patient plays an important role in impacting that outcome," Ms. Anderson says. “It’s a convergence of several dynamics that has shifted how pharmaceutical companies think about patient engagement and the types of initiatives that they bring to the marketplace."
Ms. Anderson adds that even the FDA is striving for more patient engagement.
The agency has set up a Patient-Focused Drug Development program to better engage with patients. So far, the FDA has held 21 disease-specific meetings in which they ask patients for perspectives on their diseases, treatments, willingness to participate in clinical research, and tolerance for risk. (Editor’s Note: See the box Questions the FDA is Asking Consumers About Their Diseases)
This approach is likely to expand in the near future. As part of the FDA and the pharmaceutical industry’s proposed revisions to the Prescription Drug User Fee Act (PDUFA), the FDA will be releasing several policies intended to further integrate patients into the regulatory process. Already, the FDA has proposed allowing independent groups, such as patient advocacy organizations, to conduct the patient-focused drug development meetings, potentially accelerating and spreading their patient-engagement efforts.
“People recognize that patient engagement is not a casual opportunity but more of an imperative to design each study around the experience the patient will have," Mr. Lee says. “The pharma industry is about a year or so into the adoption of this concept."
New Avenues for Reaching Consumers
Retail Pharmacy
The retail pharmacy is a trusted environment where patients are in the right mindset at the point of purchase. Jim O’Dea, CEO, Rx-Edge, believes retail pharmacies present the greatest opportunity to interact with the most patients at a time when they are thinking of their health concerns.
 Mr. O’Dea says several Gallup surveys rank the pharmacist as one of the most trusted professions in healthcare, earning a second-place ranking last year. The pharmacy is an ideal place to connect with patients because it is the first place they will go with a healthcare question, as opposed to waiting for an office visit with a physician. While the industry has traditionally viewed the retail pharmacy as a distribution channel for prescription access, pharmacies have begun to position themselves as the first stop on a patient’s healthcare journey.
Mr. O’Dea says several Gallup surveys rank the pharmacist as one of the most trusted professions in healthcare, earning a second-place ranking last year. The pharmacy is an ideal place to connect with patients because it is the first place they will go with a healthcare question, as opposed to waiting for an office visit with a physician. While the industry has traditionally viewed the retail pharmacy as a distribution channel for prescription access, pharmacies have begun to position themselves as the first stop on a patient’s healthcare journey.
“Up to 82% of people will look for an over-the-counter solution to a potential health problem before going to a doctor," Mr. O’Dea says. “That’s why we are so excited about the retail pharmacy space and pharma marketing plugging into it, which is happening more and more."
The pharmacy retailer and the pharmaceutical manufacturer want the same thing — to get the right patient on the right therapy at the right time, and, acquire new patients. However, their tactics for achieving that goal do not always align. Instead of working together toward the common goal, both entities are striving separately for it. Pharma marketers could take advantage of some, if not all, of the pharmacy retail tools being used to engage consumers, instead of creating their own one-offs.
“The pharmaceutical and pharmacy industries are completely aligned on the patient acquisition front and on the adherence front," Mr. O’Dea says. “Where it falls apart in my judgment is on the tactics. My advice to pharma manufacturers is to know what retailers are offering patients, and to align their offerings similarly."
Understanding the Patient Journey
For years the pharmaceutical industry has taken a more narrow view of the patient journey, focusing mostly on how the patient 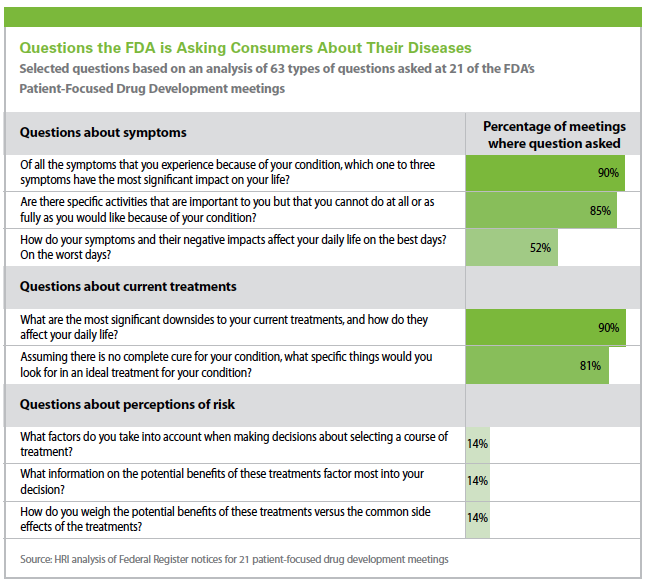 actually gets access to the drug, and how to keep him or her on the prescribed treatment. However, there’s a growing appreciation and awareness from pharmaceutical manufacturers that this narrow view is only one of the many components that a patient copes with.
actually gets access to the drug, and how to keep him or her on the prescribed treatment. However, there’s a growing appreciation and awareness from pharmaceutical manufacturers that this narrow view is only one of the many components that a patient copes with.
“There’s a growing appreciation that the patient journey is much more complex and more personal than it was thought to be in the early days," Ms. Anderson says. “The thinking around the patient journey is evolving to be much more multi-dimensional. The journey is not only about the clinical disease and getting appropriate treatment, it’s also about patients’ challenges from a lifestyle perspective, their economic and logistical access to healthcare, and their psycho-social conditions relative to their disease."
This type of rich insight is gained from analyzing patient data, which is also more prolific these days than it once was.
‘The industry is gaining more insights by talking to patients and understanding what they’re really struggling with," Ms. Anderson says. “It is reflected in a lot of the programs that pharmaceutical companies are looking to put into the market to connect with patients; companies do acknowledge a broader set of needs than just ‘here’s how to take my drug and here’s how to manage being on that drug.’ There is much more sensitivity to all of the other factors that the patient has to manage that may end up being barriers to taking the drug properly and that begins with really understanding what the challenges are, and can the drug be easier to use."
Making the patient journey easier should be the ultimate goal of all patient engagement programs. From the development process all the way to market, patient engagement programs need to be focused on helping to motivate the patient, to understand the value of the product, and to comprehend the outcome that they would derive, Ms. Anderson says.
Tools that enable direct interaction with the patients are taking hold and becoming not only an integral part of clinical and postmarketing programs, but also a strategic enabler for patient engagement, says Chitra Lele, Ph.D., chief scientific officer, Sciformix. Some of the most popular are apps and devices for cardiac monitoring, diabetes management, multi-parameter tracking, etc.
“In today’s digital age, technology enables reach into, and engagement with patients at a much wider level than ever before," she says. “Patient engagement extends beyond clinical practice and has been widely accepted as critical for clinical research over the past decade. Novel technologies such as activity trackers and mHealth devices are making their way into clinical development programs as a means of data capture and serve to encourage patient engagement in the process."
Beginning with the discovery phase, patient input is important to determine the relevance of the research questions and patient  participation is required to generate real world data around unmet needs and therapeutic burden.
participation is required to generate real world data around unmet needs and therapeutic burden.
“At the preclinical stage, patient inputs around meaningful endpoints and PROs are very useful," Dr. Lele says. “Patient inputs also help determine eligibility criteria and the informed consent process."
Increased Engagement in Clinical Trials
At the clinical trial stage patient engagement helps with patient recruitment and adherence, Dr. Lele adds. Furthermore, patient feedback helps to shape trial adaptations or modifications and patient preference studies. How the patient community views the results, in the context of real world evidence and effectiveness of treatments, has a fundamental impact on market access, payer reimbursements and more.
Mr. Lee has observed pharma creating more patient centered study designs and engaging with advocacy groups and crowd sourcing protocols. Pharma has taken strides to engage patients during the study to both improve their trial experience as well as generate insights that will be meaningful for those patients when they become consumers. He has also noticed a shift to engaging with patients on a much more customer service basis through the use of satisfaction surveys.
“We’re now seeing more sponsors take the patient satisfaction concept to heart and survey the patients in a trial, like a soft patient report of information, to see how they are experiencing the study, how they’re having their interactions with the site and how confident they are in their overall participation in the study," he says.
That allows not just a good study design, but calibration of the study experience mid-trial.
“Technology has made it easier to collect information from patients and generate these insights and we’re seeing more of our sponsors bake in that type of data collection," Mr. Lee says.
The satisfaction surveys work well to steer programs in the right direction, and patients who get these communications will stay committed to a study longer because of them.
“We are seeing tremendous correlation between participants who get these surveys and their retention rate," Mr. Lee says.
While patients benefit from the insights, the fact that the data is so measurable is a boon to the sponsors.
“We can correlate patients interactions with their mobile device to the data collected from them as well as the progress and compliance they have in the study," he explains. “Those data points haven’t existed before digital-based patient engagement."
Health Literacy Challenges
 Even in today’s advancing digital era and video capabilities, the pharmaceutical industry still engages most frequently with patients via the written word. This includes labels, disclaimers, dosing guides, instructions, and more. The problem is that most patient materials are written at a college level when only one-quarter of all adults have a college education. Further, only 12% of the U.S. population has adequate health literacy, which is necessary to effectively navigate the complex healthcare system.
Even in today’s advancing digital era and video capabilities, the pharmaceutical industry still engages most frequently with patients via the written word. This includes labels, disclaimers, dosing guides, instructions, and more. The problem is that most patient materials are written at a college level when only one-quarter of all adults have a college education. Further, only 12% of the U.S. population has adequate health literacy, which is necessary to effectively navigate the complex healthcare system.
If there is one strategy that needs to be adopted to drive change in patient engagement, it’s the need to simplify communication and verify comprehension from patient diagnosis to prescribed treatment, says Stephanie Reynders, executive director, PMRG/Health Literacy Initiative.
“But we can’t simply make things easier to read, as readability is only one factor in health literacy, and it alone is insufficient to effect change," she says.
Low health literacy can affect people from all walks of life and all levels of education. This is because health literacy is dynamic and context based. Consider someone who is newly diagnosed with a disease she knows little about. Chances are she may feel overwhelmed and have difficulty making sense of all the new information coming at her in the immediate aftermath of her  diagnosis. Other patients may, however, encounter more routine health literacy challenges, such as those experienced by patients who speak English as a second language.
diagnosis. Other patients may, however, encounter more routine health literacy challenges, such as those experienced by patients who speak English as a second language.
By including a broader scope of patients and casting a wider net, pharma will better understand the patient journey from diagnosis to maintenance and the overall perspectives of their consumers. This will allow pharma to create better materials and products to support their consumers. Unfortunately, those who deal with lower health literacy day-in-and-day-out shy away from research panels and clinical trials, thus, market research doesn’t get their input in qualitative and quantitative research.
“If pharma can find a way to engage with these patients, communications won’t be the only thing that improves," Ms. Reynders says. “The potential benefits become endless." (PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
5 Important Elements for Patient Engagement
To create connections with patients, the engagement strategies should include some or all of the following elements:
1. Be holistic — Clinical intervention affects all aspects of the patient’s life. To that extent, the patient will be much more involved if the engagement is not limited to the “pill" but more on total wellness. The patient will need a “coach" more than a clinical nurse though the coach can be a nurse.
2. Be personal – A website and an answering machine will not do it. The recommendation is to employ a live person as a strong communication medium with a maximum of one warm transfer. Taking time to study the patient’s learning style will also be important to be effective. Resources can include a website with educational resources, etc.
3. Measure periodically – Consistency, while good to keep in touch with the patient, might not be good for tactics. Knowledge
and technology get updated every 24 hours, so pharma needs to revisit both engagement and content tactics periodically. The only way pharma can keep up, is to measure responsiveness to these interventions. Key retention and engagement measures should be developed and measured.
4. Leverage technology – There have been great advances in the areas of artificial intelligence, deep learning, and machine learning. These patterns of recognition learning algorithms will help the engagement be relevant and will continually optimize the amount of time spent and thereby decreasing investment over time.
5. Involve the HCP – Not many patients trust a pharmaceutical company to take care of them. Providing a conduit for HCP involvement/engagement in total care will add more credibility to the process. The HCP, should at a minimum, have knowledge of all engagements with their patients.
















