After COVID-19, pharma companies are eyeing different ways to ensure their supply chains remain resilient to future disruptions.
When the world was hit by the COVID-19 pandemic, supply chains around the world faced almost instant disruption. The pandemic exposed vulnerabilities and highlighted the need for a radical overhaul to meet the demands of a fast-evolving marketplace.
With so many manufacturing sites concentrated in one or two geographies, the impact of a major crisis was always going to be huge. According to a report from Deloitte, more than 200 of the Fortune Global 500 companies — across all industries — have a presence in the Chinese province of Wuhan.
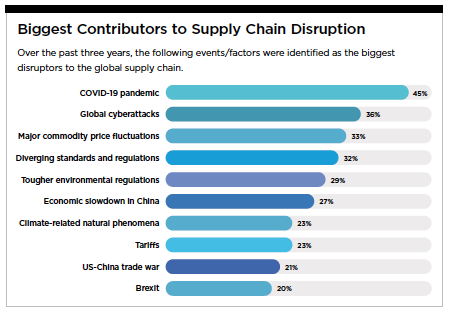
The pandemic was simply the catalyst, with many other disruptive forces affecting the supply chain, such as trade disagreements, geopolitical tensions, and environmental catastrophes. Companies have long understood the need to take steps to improve their supply-chain processes.
According to a PwC Health Research Institute survey, 94% of life-sciences executives, as well as 86% of executives from providers, prioritized improving their supply-chain operations during 2021.
 What became all too apparent was that reliance on low-cost markets, in particular China and India, exposed companies to disruption and product shortages, particularly with regard to active pharmaceutical ingredients (APIs). The focus for companies has therefore been on building flexibility and additional fail-safes into the system and to identify suppliers in different geographies to determine the impact of potential disruptions.
What became all too apparent was that reliance on low-cost markets, in particular China and India, exposed companies to disruption and product shortages, particularly with regard to active pharmaceutical ingredients (APIs). The focus for companies has therefore been on building flexibility and additional fail-safes into the system and to identify suppliers in different geographies to determine the impact of potential disruptions.
Sixty percent of respondents to a report by The Economist Intelligence Unit, commissioned by GEP, agree that excess capacity and supply-chain resilience is more important than speed and efficiency, and 54% say companies must introduce significant change to better manage supply-chain disruptions in the coming years.
Some industry experts advise having at least two trusted suppliers from different parts of the world and ensuring they retain sufficient raw materials in-house. Others suggest increasing inventory levels, which, while requiring a greater amount of investment, does ensure requirements are met if there are disruptions from a supplier. Some companies are working to bolster their relationships with existing suppliers, some have established risk-management teams and processes, and many are investing in technology.
More consideration is also being given to nearshoring or onshoring sources of supplies. A study conducted on behalf of the European Parliament discussed the economic and political justifications for bringing production back to Europe to address supply concerns. What the report noted was that although “a more proactive policy approach to mitigate security of supply concerns and to promote strategic autonomy in the EU is important, reshoring should be primarily focused on specific critical sectors and products with pronounced supply bottlenecks." The report highlights pharmaceuticals as a critical sector, noting that to protect supplies, “governments may wish to resort to other and arguably more interventionist policy options, including reshoring."
As the EU study highlights, supply-chain shortages are not purely a COVID-19 issue. In Europe, medicines shortages have been raised as a source of concern from at least 2017, and an OECD study found that drug shortages in OECD countries increased by more than 60% between 2017 and 2019.
Supply-Chain Efficiency
Nevertheless, the pandemic has brought to the fore the need for more efficient processes within the supply chain.
 Several logistics and supply-chain companies were quick to take action. 2021 PharmaVOICE 100 honoree Heather Zenk was recognized by her company AmerisourceBergen precisely because of her astute thinking in a time of crisis. She played a key role in protecting and securing the supply chain amid the pandemic and bridging the gap between manufacturers and providers to get COVID patients the treatments they need. She led a COVID-19 task force to adapt the company’s approach to business continuity plans during the pandemic to add increased focus on human capital.
Several logistics and supply-chain companies were quick to take action. 2021 PharmaVOICE 100 honoree Heather Zenk was recognized by her company AmerisourceBergen precisely because of her astute thinking in a time of crisis. She played a key role in protecting and securing the supply chain amid the pandemic and bridging the gap between manufacturers and providers to get COVID patients the treatments they need. She led a COVID-19 task force to adapt the company’s approach to business continuity plans during the pandemic to add increased focus on human capital.
“While the pharmaceutical supply chain performed very well, we came to work every day thinking about how we could adapt our processes, or even create incremental solutions, to help our manufacturer partners, provider customers, and the public at large," Heather told PharmaVOICE. “This time has taught me that scary moments in life and career can foster tremendous growth, if approached with the right intention and mindset."
Increased automation is flagged as a key component of a more efficient supply chain, as is greater use of data analytics to identify waste, track supplies, and safeguard the supply chain. And advanced technologies such as artificial intelligence and machine learning are increasingly becoming vital to improve visibility across the supply chain.
Pharma Company Action
Many large pharma companies have adopted strategies to bolster their supply chains. Early in the pandemic, Roche faced a huge spike in demand for its polymerase-chain reaction (PCR) tests for the virus, and went from first knowledge of the genetic sequence of the virus to authorization of its test kits in six weeks. That meant having to rush products through manufacturing.
The president and CEO of Roche Diagnostics North America shared the company’s experience at a Smart Manufacturing Summit. To manage the supply chain, Roche Diagnostics quickly brought together a dedicated pandemic response team. The company also expanded its global supplier network, building in supply-chain redundancies for critical items. Relationships went on full alert ensuring collaboration and transparency with suppliers. The company also hired hundreds more people at their Indianapolis and New Jersey manufacturing sites to boost output of their PCR tests.
Matthew Winterman, jhad of end-to-end supply chain at Roche, responded to the Economist Intelligence Unit report that during 2020, Roche teams overcame many logistical challenges and “ultimately succeeded in keeping complex supply chains running — ensuring vital medicines continued to reach our patients."
GSK, meanwhile, began implementing new processes to improve quality and oversight of the supply chain well before the pandemic. The company is partnering with organizations such as Siemens to use AI to automate manufacturing functions. In 2016, GSK developed the IIM Digitisation Lab to demonstrate what a data-based strategy for manufacturing would look like. The company also works with a diverse range of suppliers to improve innovation and growth, and has placed emphasis on supporting ethnic minorities, women, LGBT+, veteran and disability-owned businesses amongst its suppliers.
Among services companies, some are building digital platforms so their clients have real-time insight into their clinical and commercial supply chains. In some countries, logistics companies are collaborating with medical reps around the use of AI and other digital solutions to create SMS and email notifications around inventory from pickup to final delivery. More effort is going into improving RFID technology to create best practices and standardization across the supply chain.
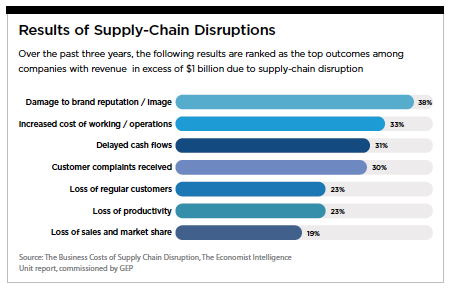
To safeguard against future supply chain disruption, companies must consider a range of strategies as well as digital solutions. Patients — and company reputations — depend on it.(PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Executive viewpoints
 Joanne Santomauro
Joanne Santomauro
CEO
Ancillare
Early Planning is Key
Early and thorough planning is the best way to ensure a fully integrated supply chain. This is the foundation of Clinical Trial Ancillary Supply Chain (CTASC). To some, “ancillary" might imply “secondary" — but not for us. Our proven model accounts for ancillary supplies and equipment early in the trial life cycle, demonstrating that ancillary supply chain is critical to meeting key milestones, including site activation timelines, first patient in, and closeouts.
Dependable Direct-to-Patient Services
Decentralized trials are gaining favor among developers, investigators, and patients. The industry needs dependable direct-to-patient services that ensure consistency across all encounters. Standardized kitting is an important way to maintain data integrity and improve ease of use for patients and investigators. During the pandemic, we helped sponsors develop custom kits for safety and speed and continue to innovate in this space.