 Advancements in technology over the past five years have led to increased interest around the topic of mHealth — the monitoring and delivery of healthcare through mobile devices. Approximately 93% of us now own a mobile phone and, with an increased desire from the pharmaceutical industry to use electronic data capture over traditional paper methods, it’s no surprise that the use of mobile phones has become commonplace within healthcare.
Advancements in technology over the past five years have led to increased interest around the topic of mHealth — the monitoring and delivery of healthcare through mobile devices. Approximately 93% of us now own a mobile phone and, with an increased desire from the pharmaceutical industry to use electronic data capture over traditional paper methods, it’s no surprise that the use of mobile phones has become commonplace within healthcare.
The use of mobile devices as a method of data capture has opened the door to an easier, and more effective, way of generating real-world data (RWD). This creates a larger base of real-world evidence (RWE) for pharma to inform decisions about the cost-effectiveness and patient value of a new product to the healthcare system.
Due to the increased availability and capability of mobile devices across the globe, pharmaceutical industry leaders are beginning to embrace the concept of collecting data directly from patients going about their day-to-day lives, as opposed to sporadic data collection during controlled site visits. As a result, this method of data collection has moved from the domain of the early adopters to that of the “early majority" or “pragmatists" as defined in the technology adoption model described in Geoffrey A. Moore’s “Crossing the Chasm."
Using Technological Advancements to Meet Market Needs
The concept of collecting data directly from patients to inform decision-making is not new, but has only recently become practical on the scale required to support post-marketing research programs routinely.
 Until recently, sponsors tended to view paper assessments as the most economical means of collecting data directly from patients. Paper questionnaires, it seemed, were much less expensive than the alternative of providing patients with electronic devices loaded with applications for reporting. However, paper involves heavy back-end costs as responses must be manually checked and entered into a database — a time-consuming, labor-intensive chore. Plus, data quality suffers from a combination of waning compliance and data-entry errors.
Until recently, sponsors tended to view paper assessments as the most economical means of collecting data directly from patients. Paper questionnaires, it seemed, were much less expensive than the alternative of providing patients with electronic devices loaded with applications for reporting. However, paper involves heavy back-end costs as responses must be manually checked and entered into a database — a time-consuming, labor-intensive chore. Plus, data quality suffers from a combination of waning compliance and data-entry errors.
Today, it’s commonly accepted within the pharmaceutical industry that electronic data collection is more efficient and economical than paper-based solutions, given the wide availability of mobile devices and advances in user interface (see “Mobile Devices Blanket the World"). Cost is now less of a barrier to adoption as sponsors don’t need to provide customized devices to patients who are willing and able to use their mobile devices for self-reporting.
In today’s digital world multiple market forces have also converged to create a rising demand for patient-collected data. Firstly, the pharmaceutical industry is making a concerted effort to understand the patient experience over time, recognizing that treatment is not just about epidemiology or biology. To fully serve patients and improve their care, pharmaceutical companies are eager to understand how patients are feeling, how they make decisions, how their lives are impacted by their condition/disease, and what challenges they face functionally and emotionally. Secondly, payers are demanding evidence of a product’s value for cost, and RWD collected from patients can bolster evidence that a product adds value beyond the standard of care. This information may become crucial in an increasingly competitive marketplace. Thirdly, regulators are moving toward a more patient-centered focus, this is particularly true in some therapeutic areas — notably oncology — where regulators are requesting assessments of patient symptomatology that can only be derived from patients themselves.
Applications of Patient-Collected RWD
 Patient-focused RWD programs can be designed to elicit subjective feedback on their symptoms and side effects, response duration, treatment adherence, treatment satisfaction, activity levels, emotional state, lifestyle, quality of life, and any other relevant contextual elements.
Patient-focused RWD programs can be designed to elicit subjective feedback on their symptoms and side effects, response duration, treatment adherence, treatment satisfaction, activity levels, emotional state, lifestyle, quality of life, and any other relevant contextual elements.
Ultimately, this information, when analyzed and interpreted, contributes to a rich source of RWE that can inform a wide range of important healthcare decisions throughout all stages of the product lifecycle. These RWE-informed healthcare benefits range from demonstrating product safety and value to informing future program designs. As such, RWE is impacting decisions such as product reimbursement and market access now more than ever, resulting in pharmaceutical companies beginning to realize the true potential of patient-collected RWD and the value it can provide:
Obtain a better understanding of treatment pathways and the patient’s disease trajectory
Support regulatory submissions with evidence of a product’s treatment benefit in terms of symptom relief and holistic patient impact
Articulate the value of new therapies to support initial product pricing and market access decisions
For established products, defend against brand challenges and costly label changes
Calculate the total cost of care, when combined with other RWD sources
Support risk management and the monitoring of drug side effects
Compare alternative treatment approaches
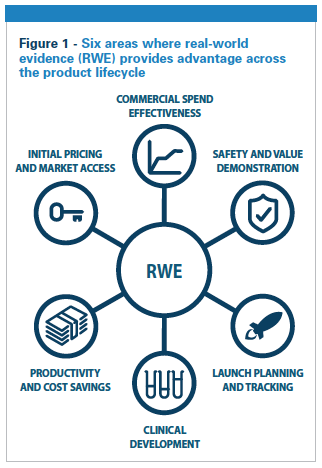
Combining data collected directly from patients with other RWD sources provides a more holistic view of the patient’s treatment experience and changing health status. These data enable sponsors to fine-tune their estimations of a drug’s overall value and its impact on efficacy and safety. The most appropriate combination of data sources will likely be different for each given research need, but patient-collected RWD enriches RWE analysis. Hughes et al. estimate that RWE can potentially generate $1 billion for a top-10 pharmaceutical product when it is applied collectively in six major areas (see figure 1).
Benefits of Electronic Data Capture Solutions
Compared with paper-based surveys, electronic solutions offer multiple advantages:
Improved data quality: As data are transferred automatically from the mobile device to a database, there are no errors resulting from manual data entry. The tool can be set to accept only data within specified parameters, preventing the entry of outlying data. A patient, for example, would not be able to enter a body temperature of 88° Fahrenheit.
Resource savings: Incoming data do not have to be manually entered and checked.
Strong patient compliance: With paper-based collection methodologies, it is common for participation to drop off during post-approval programs, especially if programs run over long durations. Continuing advances in smartphone and other portable technologies are paving the way for ever-better digital patient engagement and data capture. It’s easy for patients to complete questionnaires and diaries on mobile devices in a way that integrates into their everyday lives and improves participation and adherence.
Rapid deployment: Research programs can be implemented quickly around the world.
Unlimited participation: The methodology can be extended to large, heterogeneous populations across different diseases.
As direct-from-patient RWD continues to contribute to the RWE base, it will undoubtedly play an important role in ever-widening applications, particularly as more companies start using RWE strategically. Certainly, as RWD is hailed as the “new currency in healthcare," there is a growing advocacy for RWD collection earlier in the clinical development process.
 It is likely that patient engagement and participation in the real world will become more entwined across the entire product lifecycle, from development through to post-approval. Exactly how this will be optimized remains unclear. Yet, what is clear is that electronic data capture solutions and developments to enhance their affordability, rapid implementation, and ease-of-use have made great strides in recent years.
It is likely that patient engagement and participation in the real world will become more entwined across the entire product lifecycle, from development through to post-approval. Exactly how this will be optimized remains unclear. Yet, what is clear is that electronic data capture solutions and developments to enhance their affordability, rapid implementation, and ease-of-use have made great strides in recent years.
Furthermore, the ability of patients to use their own devices makes RWD programs a wholly better experience both for them and for program sponsors looking for an affordable way to collect rich and robust RWD to contribute to the RWE base.
Continuing advances in smartphone and other portable technologies will pave the way for better digital engagement and data capture solutions to improve patient care from the comfort of their own homes. Through improved program organization and better support via technological developments, program sponsors are able to recruit and retain large groups of patients and maintain their participation throughout the duration of the program.
As such, the RWD that these programs generate continue to improve and proliferate. This means that decisions about new products and their applications can be better informed and more efficiently made, such as in developing precision medicines matched to an individual patient’s specific disease. Overall, this can only be good news for the development and accessibility of new treatments and ultimately, people’s health. (PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Remote Patient Monitoring Devices
The subjective information collected from electronic assessments that patients complete can be supplemented with more objective data from remote patient monitoring devices, including wearables and medical-grade sensors. These devices are passive, objective, and always on, continuously collecting data on the patient’s state of well-being. Currently, use of such devices outside traditional clinical research is limited due to the cost of provisioning devices. However, as these technologies become more commonplace they are expected to play a larger part in RWD capture.
~~~~~~~~~~~~~~~~~~~~~~~~~
Mobile Devices Blanket the World
The statistics on mobile phone ownership around the world make it clear that the availability of hardware is no longer an obstacle to electronic data collection from patients.
In 2016, 93% of the population owned a mobile phone with 68% of those people owning a smartphone. This was a significant increase from 42% smartphone ownership in 2013.
https://www.consumerbarometer.com/en/graph-builder/?question=M1 with a sample size of 56 countries.
At the same time, smartphone ownership in emerging and developing countries is rising at an astounding rate — from a median of 21% in 2013 to 37% in 2015. In almost every country the Pew organization surveyed, overwhelming majorities own some form of mobile device, if not a smartphone.
ERT minimizes uncertainty and risk in clinical research so you can move ahead quickly and with confidence.
For more information, contact [email protected] or visit ert.com.

















