DTRA Launches to Democratize and Accelerate Clinical Trials
 A new global industry coalition — Decentralized Trials & Research Alliance (DTRA) — aims to dramatically increase access for all patient populations in clinical trials and research. DTRA’s goal is to unite stakeholders, including healthcare companies, regulators, patient groups, and research organizations, to make clinical trial participation widely accessible by advancing policies, research practices, and new technologies in decentralized clinical research.
A new global industry coalition — Decentralized Trials & Research Alliance (DTRA) — aims to dramatically increase access for all patient populations in clinical trials and research. DTRA’s goal is to unite stakeholders, including healthcare companies, regulators, patient groups, and research organizations, to make clinical trial participation widely accessible by advancing policies, research practices, and new technologies in decentralized clinical research.
 “The benefits of decentralized research methodologies have been apparent for some time, but adoption has been slow due to many factors including culture and the lack of a forum for stakeholders to collaborate," says Amir Kalali, M.D., life science executive, founder of several collaborative life-sciences communities, and co-convenor of DTRA. (Dr. Kalali is a PharmaVOICE 100 Red Jacket honoree.) “The COVID-19 pandemic has forced organizations to adopt decentralized methodologies which have the potential to broadly accelerate drug development."
“The benefits of decentralized research methodologies have been apparent for some time, but adoption has been slow due to many factors including culture and the lack of a forum for stakeholders to collaborate," says Amir Kalali, M.D., life science executive, founder of several collaborative life-sciences communities, and co-convenor of DTRA. (Dr. Kalali is a PharmaVOICE 100 Red Jacket honoree.) “The COVID-19 pandemic has forced organizations to adopt decentralized methodologies which have the potential to broadly accelerate drug development."
Experts estimate that COVID-19 may set back non-pandemic clinical trials by several years due to prospective patients’ inability or reluctance to schedule visits at physical research locations. Decentralized clinical trial approaches to conducting research facilitate participation by a more diverse patient population and could ease COVID-19-imposed difficulties for both patients and clinical investigators. Additionally, COVID-19 further exposed the operational challenges of the inclusion of representative patient populations in clinical trials by race, age, and geographic location and amplified the disparities and inclusion biases that have become hurdles for potential trial participants.
“Now is the time to share ideas and insights that will chart the future course of clinical trials, accelerating drug development and saving lives," says Craig Lipset, DTRA co-convener, clinical innovation advisor, and a pioneer in decentralized trials. (Mr. Lipset is also a PharmaVOICE 100 Red Jacket honoree.) “We have a responsibility to advance the health of people with unmet medical needs, and by convening stakeholders we can remove remaining barriers to adopting new policies and practices that can impact patients today."
Nearly 50 companies have already signed up as DTRA member organizations to provide expertise to identify and address gaps and needs and advance best practices through effective education and communication. Organizations interested in taking part can visit DTRA.org or follow DTRA on Twitter and LinkedIn.
Americans Exhibit Vaccine Hesitancy
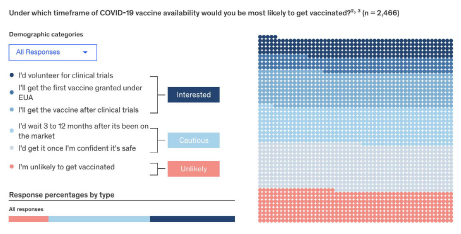 According to a mid-December 2020 report from McKinsey & Company, more than half the respondents in the United States report they are likely to delay or decline vaccination despite regulatory approval, with safety concerns being the main driver of vaccine hesitancy.
According to a mid-December 2020 report from McKinsey & Company, more than half the respondents in the United States report they are likely to delay or decline vaccination despite regulatory approval, with safety concerns being the main driver of vaccine hesitancy.
MMRF Launches CureCloud
The Multiple Myeloma Research Foundation (MMRF) has launched the first at-home genomic test for cancer at no cost. At a time when hospitals are overcrowded with COVID-19 patients, the CureCloud allows patients to get testing in the convenience of their home.
A phlebotomist is sent to the patient’s home to collect a blood biopsy, avoiding a painful and invasive bone marrow biopsy. In just 4 to 6 weeks, myeloma patients will receive a visualized, CLIA grade report that includes their data, along with a map of their genomic sequencing, and a personalized treatment option to discuss with their doctor on the best, most tailored treatment options for their specific subtype. It also includes a visualization tool for the patient to compare themselves to similar patients to learn what has worked best for them to grant them further insights into their treatment options. The MMRF makes the information available to the entire myeloma community of doctors, patients, scientists, and researchers. This will help patients and doctors make better decisions about their treatments faster.
Telehealth Is Convenient Alternative to In-Person HCP Visits
 A new Health Union survey reveals that people with chronic conditions feel that telehealth, despite its convenience and increased use throughout the COVID-19 pandemic, is generally less preferable than in-person visits, but can still serve as an occasionally suitable alternative.
A new Health Union survey reveals that people with chronic conditions feel that telehealth, despite its convenience and increased use throughout the COVID-19 pandemic, is generally less preferable than in-person visits, but can still serve as an occasionally suitable alternative.
The survey is the fifth in Health Union’s ongoing COVID-19 Consumer Attitudes and Health Behaviors Survey series that captures “snapshots in time" that track the perspectives and health behaviors of people with chronic conditions throughout the pandemic.
Telehealth use continues to increase during the pandemic for people living with chronic health conditions. Nearly three-fourths of respondents of this wave 5 survey said they have had at least one telehealth appointment, up from 63% from the wave 4 survey, which fielded in July.
Fortunately, two-thirds of respondents who have had telehealth appointments considered their experiences to be positive, with convenience being a primary reason.
And 44% of all respondents said they were “extremely likely" to consider using telehealth after the pandemic is under control. This number was even higher for people living with autoimmune conditions, such as ankylosing spondylitis (61%), Crohn’s disease (61%), and plaque psoriasis (56%).
However, the survey findings reveal that increased use doesn’t necessarily translate into a clear preference for telehealth. Likewise, telehealth may not always make sense — either to the patient or the healthcare professional (HCP) — for certain types of visits. Instead, telehealth appears to be viewed as an important and convenient tool that, depending on the situation and a number of factors, like preference and comfortability, can be used along with in-person visits to provide patients the care they need.
Unsurprisingly, after living through the pandemic for nearly eight months, nearly all respondents reported having at least one HCP visit. These visits appear to be a combination of both telehealth and in-person, with 63% reporting using both. Only 10% of respondents have just used telehealth during the pandemic, while nearly a quarter have only seen their HCP in person.
Respondents don’t seem to have an outright preference for telehealth. Instead, they seemingly prefer in-person visits or an either-or scenario, but the reason for the visit appears to be a major factor. More than seven in 10 said they prefer only in-person visits for annual checkups, 65% prefer them for unplanned visits, and 46% prefer them for discussing new symptoms, which often requires a physical evaluation. On the other hand, a large portion of respondents said they were fine with either type of visit for discussions about changing or starting new treatments, as well as evaluating whether a new medication is working.
Unsurprisingly, more than six in 10 who have used telehealth rated convenience as at least somewhat better than in-person office visits. That being said, respondents found in-person and telehealth visits to be roughly the same for a number of measurements, including the quality of conversation, the ability to ask questions or voice concerns and the amount of time spent with the HCP.
Greater Gift Gifts $50K Scholarship
 Greater Gift, a nonprofit founded in 2010 with a mission to increase awareness of clinical research, especially among underrepresented communities, announces a $50,000 scholarship to support a minority graduate student enrolled in Wake Forest University’s newly launched Master’s in Clinical Research Management Program. The scholarship is named in honor of Mary Hofmann Murphy, a Winston-Salem native and clinical research participant. The inaugural scholarship was awarded to Darius Ford of Raleigh, a student enrolled in the first cohort of students in the program.
Greater Gift, a nonprofit founded in 2010 with a mission to increase awareness of clinical research, especially among underrepresented communities, announces a $50,000 scholarship to support a minority graduate student enrolled in Wake Forest University’s newly launched Master’s in Clinical Research Management Program. The scholarship is named in honor of Mary Hofmann Murphy, a Winston-Salem native and clinical research participant. The inaugural scholarship was awarded to Darius Ford of Raleigh, a student enrolled in the first cohort of students in the program.
Greater Gift’s goal is to further the engagement of underrepresented groups in clinical research by investing in future leaders in the industry. According to Executive Director Lilly Skok Bunch, “We will only have real equity in clinical research participation when the research industry is reflective of our community. This scholarship is a step in that direction."(PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Pharmaceutical Positioning
 Mike Rea, CEO of IDEA Pharma, who has worked on some of the most successful new drug and medical product launches of the last 20 years, has written a new book — Pharmaceutical Positioning — the world’s first book on positioning pharmaceuticals.
Mike Rea, CEO of IDEA Pharma, who has worked on some of the most successful new drug and medical product launches of the last 20 years, has written a new book — Pharmaceutical Positioning — the world’s first book on positioning pharmaceuticals.
As a discipline, positioning emerged in the 20th century as a powerful concept in product and brand marketing. This book, for the first time ever, applies proven and new positioning methods to pharmaceuticals. This book captures many of the lessons on positioning that underpin successful development and launch. Whether your product is first to create a new market, or fifth into an old market, positioning is a fundamental part of whether or not your product will be successful, and how successful it will be.
“The 1980 Ries and Trout book on positioning has served us well, but it has never really fit pharma," Mr. Rea says. “Given the importance of what we do, the opportunity to rethink positioning for a pharmaceutical audience is critical to increase the relevance of great positioning for what we develop, as well as how we communicate it. They said ‘positioning is not what you do to a product.’ I counter that it is absolutely what you do to a product — in fact, it is everything that you do to help a molecule become a product."
Mr. Rea is a PharmaVOICE 100 Red Jacket honoree.
~~~~~~~~~~~~~~~~~~~~~~~~~
Case Study:
More Than Ever, New Therapies Require Reimbursement Support
Provided By: RxCrossroads by McKesson
Contributed Articles:
Why Your Third-Party Logistics Partner Should Scale With You
Provided By: RxCrossroads by McKesson
Real World Applications of AI-Based Automation in Life Sciences by Keeping Humans in the Loop (HITL) And Machines in the Loop (MITL)
Provided By: Indegene
Advanced Data and Analytics in Life Sciences: The Winning Prescription
Provided By: Indegene
eBook:
Innovating for the Future of Drug Development
Provided By: Parexel
Podcast:
Agents of Change: Lessons from Guiding Business Transformations From Start to Finish
Sponsored by: Gagen MacDonald
Therapeutic Digests:
Central Nervous System
Provided by: ThinkGen and PharmaVOICE
Rare Diseases
Provided by: ThinkGen and PharmaVOICE
Webinars/Virtual Panels:
The Art and Science of Oncolytic Commercialization in 2021 and Beyond
Sponsored by: 1798, a Fingerpaint Company
(OnDemand) Why Centralizing Medical Imaging File Capture & Adjudication Enhances Decision Making
Sponsored by: Datatrak International
(OnDemand) Silicone Layer in Glass Primary Packaging
Sponsored by: Stevanato Group
White Papers:
5 Things Clinical Trial Sponsors Must Digitize Now to Emerge Stronger in 2021
Provided By: ArcheMedX
The Evidence Is In: Integrated Hub and Copay Services Really Do Yield Better Results
Provided By: TrialCard
Engaging CRCs to Meet Site Challenges
Provided By: WCG
~~~~~~~~~~~~~~~~~~~~~~~~~
PharmaVOICE 100 News
 Brion Brandes Joins Incedo
Brion Brandes Joins Incedo
Brion Brandes joins Incedo as VP, commercial solutions, life sciences. Incedo provides end-to-end digital transformation consulting, data science & analytics, technology services and solutions. Mr. Brandes has more than 25 years of experience in life sciences and healthcare and has held leadership roles in business development, sales, product/regional marketing, patient support services, and strategic alliances. These various roles have allowed him to gain experience helping biopharma clients develop solutions that meet their short- and long-term needs. Mr. Brandes is a PharmaVOICE 100 — 2016 honoree.
 Andrea Heslin Smiley — Board Directorship
Andrea Heslin Smiley — Board Directorship
Andrea Heslin Smiley has joined the board of directors of Rockwell Medical, a biopharmaceutical company dedicated to transforming the treatment of iron deficiency and iron deficiency anemia management and improving outcomes for patients around the world. Ms. Smiley brings close to three decades of commercialization and general management experience in the biopharmaceutical industry. She serves as president and CEO of VMS BioMarketing, a provider of patient and healthcare provider support. Ms. Smiley is a PharmaVOICE 100 — 2019 honoree.
~~~~~~~~~~~~~~~~~~~~~~~~~
Woman of the Week Podcast Series
November
Jill O’Donnell-Tormey, CEO, Cancer Research Institute (CRI)
Donna LaVoie, President and CEO, LaVoie Health Science
Tanya du Plessis, VP of Data Strategies and Solutions, Bioforum the Data Masters
Elaine O’Hara, Chief Commercial Officer, Head of North America Commercial Operations, Sanofi Pasteur, the vaccines division of Sanofi