Colon Cancer Screening without the Prep
Alex Ovadia, CEO of Check-Cap, talks about the company’s ingestible imaging capsule-based system for colorectal cancer screening.
 Colorectal cancer (CRC) — the term referring to both rectal and colon cancers — is the third most common cancer diagnosed in both men and women and the second-leading cause of cancer-related deaths. Despite being highly preventable and/or treatable with early screening, according to the American Cancer Society, CRC is expected to account for about 51,020 deaths in 2019.
Colorectal cancer (CRC) — the term referring to both rectal and colon cancers — is the third most common cancer diagnosed in both men and women and the second-leading cause of cancer-related deaths. Despite being highly preventable and/or treatable with early screening, according to the American Cancer Society, CRC is expected to account for about 51,020 deaths in 2019.
In recent years, a significant decline in the incidence of CRC has been attributed to regular screening, which should be carried out between the ages of 50 and 75 years. Yet, in 2015 only 63% of eligible adults were current with screening recommendations. In June 2018, the American Cancer Society updated CRC screening guidelines, lowering the recommended age to begin screening from 50 to 45 years.
There are many reasons why a large percentage of the population does not undergo screening. One of the most significant factors is the discomfort of specific features of certain testing methodologies, including laxative bowel preparation, invasiveness, and sedation.
Check-Cap, a clinical-stage medical diagnostics company, is looking to make the screening process easier and less intrusive. The company was founded in 2005 with the vision of developing the world’s first ingestible imaging capsule-based system — C-Scan — for preparation-free colorectal cancer screening.
The C-Scan system consists of several components, including the C-Scan Cap, which is a single-use ingestible scanning capsule that generates imaging data and positioning data; C-Scan Track, which tracks the capsule and records imaging data and positioning data; and C-Scan View, a stand-alone application that processes and displays structural information. The technology includes ultra-low dose X-ray and wireless communications. According to Alex Ovadia, CEO of Check-Cap, the 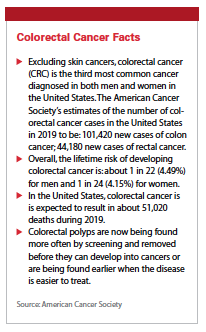 capsule generates information on the contours of the inside of the colon as it passes naturally. This information is used to create 2D and 3D maps of the inner surface of the colon.
capsule generates information on the contours of the inside of the colon as it passes naturally. This information is used to create 2D and 3D maps of the inner surface of the colon.
“The C-Scan system has the potential to increase compliance for colorectal cancer screening," he says. “Our system does not require any sedation, there is no risk of colon perforation for the patient, there is no pain, and there is no interventional procedure. It’s a completely autonomous procedure."
The only requirement for the patient is the need to take a tiny amount of contrast material, 15 mL, with three meals prior to using the system.
Mr. Ovadia says the exposure to radiation from the system is negligible. “We performed a comprehensive analysis and found the amount of exposure to the patient in total throughout the procedure is 0.05 millisieverts, which is about the same level as a chest X-ray, a transatlantic flight, or 10 days on Earth," he explains “A visual colonoscopy exposes the patient to roughly 2 to 5 millisieverts."
He says C-Scan compares favorably with other polyp detection methods, demonstrating 76% sensitivity and 80% specificity for benign or precancerous polyps in the colon.
In April 2019, the company began a U.S. pilot study of the C-Scan system, following Institutional Review Board approval and an Investigational Device Exemption application approval from the FDA.
The first patients who ingested the C-Scan capsule were part of a study conducted at the NYU School of Medicine. The single-arm pilot study (NCT03735407) will enroll up to 45 subjects considered to be of average risk for polyps and colon cancer. The study is evaluating the safety, usability, and subject compliance of the C-Scan system. In May, the company announced that Mayo Clinic joined the ongoing pilot study of the C-Scan system as the second trial site in the U.S.
The company received a CE Mark for C-Scan early in 2018, as well as approval for marketing in Israel in 2018. Mr. Ovadia says the goal is to begin a pivotal study by the middle of next year, with possible filing with the FDA in late 2021. (PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Colorectal Cancer Facts
Excluding skin cancers, colorectal cancer (CRC) is the third most common cancer diagnosed in both men and women in the United States. The American Cancer Society’s estimates of the number of colorectal cancer cases in the United States in 2019 to be: 101,420 new cases of colon cancer; 44,180 new cases of rectal cancer.
Overall, the lifetime risk of developing colorectal cancer is: about 1 in 22 (4.49%) for men and 1 in 24 (4.15%) for women.
In the United States, colorectal cancer is is expected to result in about 51,020 deaths during 2019.
Colorectal polyps are now being found more often by screening and removed before they can develop into cancers or are being found earlier when the disease is easier to treat.
Source: American Cancer Society
















