Pharmaceutical outsourcing remains big business, as companies cut internal costs and with the rise of smaller specialist companies focused on rare diseases and genomics-based therapies.
The move to outsource is growing, with reports that companies are turning to partners more now than they did five years ago. This has led to a steep rise in the number of people employed at CROs – from about 30,000 15 years ago to around 100,000 today.
At the same time, outsourcing organizations are following pharma companies down the path of mergers and acquisitions. For example, Syneos Health was the result of a merger between INC Research and inVentiv Health, and in August 2018, the company acquired Kinapse to expand its ability to provide end-to-end solutions to life-sciences  companies. Other big mergers have included Quintiles and IMS Health as well as the acquisition of DrugDev by Quintiles, PPD’s acquisition of Acurian, and LabCorp’s purchase of Covance. Capital investors have also been acquiring CROs, for example, Pamplona Capital bought Parexel and Amulet Capital Partners acquired SynteractHCR.
companies. Other big mergers have included Quintiles and IMS Health as well as the acquisition of DrugDev by Quintiles, PPD’s acquisition of Acurian, and LabCorp’s purchase of Covance. Capital investors have also been acquiring CROs, for example, Pamplona Capital bought Parexel and Amulet Capital Partners acquired SynteractHCR.
The Impact of M&A
According to a 2017 report from Industry Standard Research, the top nine CROs control 60% of the market, a figure that is likely to have risen with more M&A activity.
For pharmaceutical companies there may be concern about fewer choices, but for large pharma companies working with just a small number of big, consolidated CROs opens the door to preferred provider agreements.
Larger CROs are also more likely to have global presence, which means sponsors don’t have to search for support from different vendors as they enter new markets. These big companies also are more likely to have the latest technologies and to invest in solutions to manage site identification, selection, and start up.
Often the M&A activity is designed to expand the types of services CROs offer to pharma companies. For example, in 2017, Parexel bought specialty CRO Anolinx to gain the data science expertise it needed to provide clients with support in informatics and epidemiology. These are increasingly important services for pharma companies, so having a partner with those skills does add value.
Many such CROs are looking to provide a “one-stop-shop" to pharmaceutical clients, which is ironic as they often have multiple external partners.
While the M&A trend may be worrisome for smaller companies that can’t afford to work with large CROs or that don’t believe they will get the support they need, industry experts note that there are still many small, niche CROs in the industry and that this is unlikely to change any time soon.
There are other concerns that these large M&As raise. For example, smaller companies that serve CROs — such as patient recruitment organizations — find themselves with less work as companies merge. In addition, disruption is inevitable during a merger so clinical trials being carried out by a company that is in the process of being acquired will likely be affected. One impact might be due to labs closing, forcing data to be transferred to a different site.
Why Companies Outsource
The good news is that for larger pharma companies at least, the CRO M&A activity isn’t expected to detract from one of the major objectives of outsourcing: to cut costs. In fact, companies may save money and resources by not having to set up agreements with multiple partners.
Given the high costs of bringing a drug to market together with the huge risks involved, pharmaceutical companies have looked to service providers to help them tackle many aspects of their R&D and manufacturing. In fact, for some products, half  or nearly half of development is now outsourced. This includes 51% for vaccines, 46% for blood factors, and 44% for hormones.
or nearly half of development is now outsourced. This includes 51% for vaccines, 46% for blood factors, and 44% for hormones.
Often, the start-up costs involved in producing and manufacturing a product are too high for small companies to absorb, so outsourcing makes sense. Biopharmaceutical analysis can also be costly and for smaller companies it often doesn’t make sense to acquire the equipment needed to carry out analysis of compounds.
Because CROs are carrying out studies on behalf of many clients, they perhaps have more reason to invest in technology, with a Tufts Center for the Study of Drug Development report finding CROs invest 10% more than sponsor companies.
Efficiency goes hand-in-glove with cost savings, and there is evidence that CROs may be more efficient at site selection, feasibility assessment, and study activation. The Tufts report found CROs report completing site-related activities between six and 11 weeks faster than sponsors and have a shorter cycle time of site identification, selection, and study start up.
So why else do companies outsource? Specialist knowledge is a key priority for many companies when it comes to outsourcing. Sponsor companies often turn to vendor partners for scientific and regulatory expertise that they don’t have in-house, from managing regulatory submissions to advising on guidances by the regulatory authorities. Increasingly, CROs offer more complex services such as data analytics, another priority for sponsors.
Pharmaceutical companies also look for access to specialty equipment with partners, particularly with regard to contract manufacturing.
It’s likely that CROs and other outsourcing businesses will continue to look for ways to expand their reach and services, perhaps through partnerships or through M&A. Sponsor companies should monitor these trends to assess what will work best for them now and into the future.(PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Executive Viewpoints
 Clareece West
Clareece West
VP and General Manager,
Cardinal Health Regulatory Sciences
Mega Mergers
For pharma companies, mega-mergers can often lead to greater efficiencies and a reduction in overall costs by identifying and eliminating redundancies between the merging companies. Outsourced service partners with experience in managing integrations and aligning resources can play a critical role. An integration period can be a good time for pharma companies to evaluate which of their trusted service providers can help reduce costs in lower volume areas or their non-core services, which in turn may increase the new company margins.
The Future Of Outsourcing
For the CRO industry, the future is built around big data and their ability to leverage data on behalf of pharma companies to make decisions that reduce costs, improve profitability and to help get medicines and devices to market faster. As more therapies come to market, drug developers will be required to provide more concrete evidence of clinical superiority and cost-effectiveness of their products to regulators, payers and prescribers, which will lead CROs to become increasingly focused on delivering new sources of data. To meet the growing market demand, I believe we will continue to see more M&A deals between CROs and data/technology companies.
~~~~~~~~~~~~~~~~~~~~~~~~~
M&A: Outsourcing
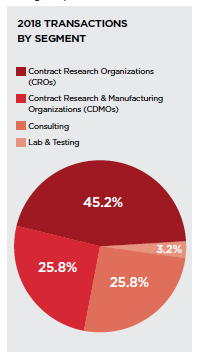
Contract research organizations (CROs) remain the most targeted segment in the pharmaceutical outsourcing industry, accounting for 45.2% of deals to-date in 2018 compared with 38.3% in all of 2017. Buyers have sought to cut clinical trial times and reduce spending through these strategic acquisitions.
Buyer Preferences
Scalable business models with proven results: Businesses that have displayed strong growth characteristics with the ability to capitalize on significant future growth opportunities have been highly valued by buyers.
 Reputation for delivering superior solutions to life sciences sector: Acquirers have sought outsourced providers with proven capabilities, instruments, and technical staff to deliver high quality and responsive outsourced services.
Reputation for delivering superior solutions to life sciences sector: Acquirers have sought outsourced providers with proven capabilities, instruments, and technical staff to deliver high quality and responsive outsourced services.
Tenured management team: Strategic suitors, and in particular private equity groups, have targeted businesses with strong management experience and depth.
Rigorous quality assurance protocols: A sterling audit record with the
FDA and clients has been viewed as a critical attribute for success in the life-sciences outsourcing sector and has attracted buyer attention.
Recurring revenue and visibility: Businesses that have developed recurring revenue streams and long-term visibility are highly sought after in the space.
Source: Capstone Headwaters
Across its various functions a market authorization holder may work with 350+ external partner contacts, unsurprisingly many in the industry wish to shrink this number. Manufacturers are seeking strategic partnerships so they can maintain a global reach with the technologies they require but via a smaller base of contacts.
Source: PharmaIQ
In drug discovery alone, outsourcing is expected to become a $43.7 billion business by 2026, rising from $19.2 billion in 2016.
Source: Biopharma Trend


















