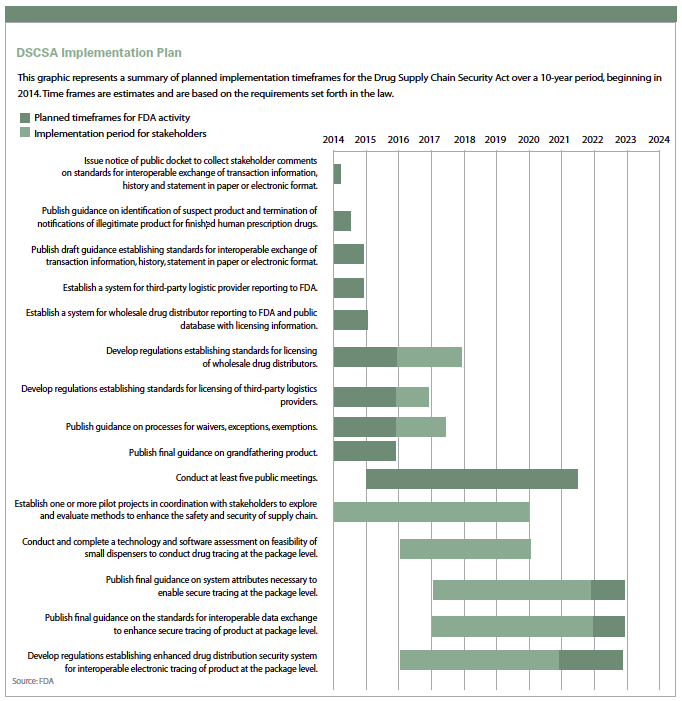
The first deadline of the 10-year DSCSA Implementation Plan takes effect this month, and experts say many drug manufacturers are not ready.
The Drug Supply Chain Security Act (DSCSA) Implementation Plan regulation, which began phasing in on Jan. 1, 2015, provides a national policy for combating counterfeiting, theft, and diversion in the U.S. pharmaceutical supply chain. The law establishes extensive new regulations providing for the tracing and verification of products and transactions across all changes of ownership, from manufacturer to dispenser. As a result, the entire industry — and all of its many moving parts — is facing complicated data management, data exchange, and business process challenges that will impact operations and all supplier and trading partner relationships.
The first deadline requires pharmaceutical companies to include product transaction information in an electronic or paper record every time ownership is transferred. The document must include transaction data listing lot level information.
The documentation must be kept for six years. On the next deadline, in November 2017, pharmaceutical transaction information will no longer be allowed on paper. Transaction information must be available electronically.
~~~~~~~~~~~~~~~~~~~~~~~~~
Fast Fact
75% to 80% of the global drug supply will be covered under global track and trace regulations by 2018 — upward of 10 billions of units a year that companies across the industry need to manage.
Source: TraceLink
~~~~~~~~~~~~~~~~~~~~~~~~~
Also in November 2017, a product identifier must be on the label of each case and individual product. The DSCSA preempts all existing and future state pharmaceutical serialization and pedigree efforts such as those that previously existed, for example, e-pedigree regulations in California and Florida.
Given the diversity of partners and systems, interoperability between trading partners is crucial and flexibility is key to this plan.
Companies may use current electronic or paper-based methodologies as long as these methods meet all the provisions of the DSCSA. Methods documented in the guidance include EDI ASN (such as the HDMA ASN), EPCIS (such as the GS1 EPCIS v1.1), Web portals, and paper (such as packing slips or invoices).
Starting this month, companies take a first step on a 10-year journey toward capturing, maintaining, and providing all supply chain information in full compliance with all provisions of the Tracing and Verification regulations in DSCSA section 582. The next deadlines loom in 2017.
Virtus Pharmaceuticals, a specialty pharmaceutical company focused on the development and marketing of prescription products to pharmaceutical wholesalers, chain drug stores, and other retail merchants in the United States, has been preparing for the DSCSA requirements for almost a year.
“We began working on DSCSA requirements last April and it’s taken quite some time to get to a point where we are now testing and implementing the actual system," says Louis Sanchez, CEO of Virtus.
Track and trace in the supply chain isn’t new. While drug makers with products that are high-risk for counterfeiting and theft have implemented track and trace systems, the new regulation applies to all and will spur an uptake of investment in such systems.
According to Frost & Sullivan, track and trace solutions markets in both the United States and in Europe will continue to grow as pharmaceutical companies strive to comply with the legislation.
“In addition, differentiation and more efficient processes are critical for companies to compete," says Jennifer Lazar, life sciences and connected health global program director at Frost & Sullivan. “As a result, track and trace solutions that enhance efficiency will become key tools for companies in establishing a competitive advantage."
Mr. Sanchez credits the regulations with forcing the industry to be more proactive and better prepared to battle counterfeiting or contamination risks. Knowing where a drug product is during the manufacturing process and knowing where it is going once it leaves a company’s distribution will make the reaction time substantially lower when responding to an unplanned event.
“This system will allow us to immediately respond to upstream and downstream partners should an issue arise," Mr. Sanchez says. “The regulations alone cannot stop counterfeiting or contaminated drugs, but this system does give us better capability of responding in real time."
According to reports from Frost & Sullivan, counterfeit drugs are estimated to cost the industry between 7% and 10% of global pharmaceutical market revenue.
“It is a serious problem for pharmaceutical brands, due to the direct loss of revenue and the indirect loss of brand equity when consumers are adversely affected by fake drugs," Ms. Lazar says.
Other companies that are leading the charge toward implementing DSCSA requirements are Pfizer and GSK.
These major drug makers have been facing the threat of counterfeiting for years, and the complying with the new regulations will enhance the efforts they already have in place.
Ms. Lazar notes that in general, counterfeit and theft are more prevalent among expensive, life critical drugs such as HIV medicine and cancer drugs, as is the case with GSK’s HIV drug, Trizivir. GSK is has a trial program in place for an RFID based track and trace solution at a per bottle level for Trizivir to reduce counterfeit and theft risks.
However, the main reason for the deployment is not to reduce stolen goods, but to enhance efficiency in the processes throughout the supply and distribution channel, she says.
Pfizer has been preparing for the California e-pedigree law for several years and is many steps ahead in applying its learnings and solutions, such as serialization and cloud-based IT solutions for the federal mandate. Pfizer’s Viagra bottle RFID track and trace solution in the United States is a good example of a functioning RFID track and trace solution.
“Major pharmaceutical companies such as Pfizer are leading the way," Ms. Lazar says.
 According to Shabbir Dahod, president of TraceLink, whose company works with Pfizer, the company is years ahead of the industry when it comes to track and trace.
According to Shabbir Dahod, president of TraceLink, whose company works with Pfizer, the company is years ahead of the industry when it comes to track and trace.
In fact, Pfizer won CIO magazine’s award in 2013 for its track and trace serialization Project that includes serialization of all products and shares the terabytes of product data with hundreds of global manufacturing partners. Pfizer’s cloud-based data-management platform simplifies supply chain management while ensuring compliance with government regulations by providing a single repository for serial numbers, product data, partner information, real-time supply network transactions and event data.
As a result, Pfizer is able to respond faster to requests for information from global partners, reduce the time required to track and trace products through its global supply chain, speed up time-to-compliance with evolving regulations and protect patient safety.
Mr. Sanchez at Virtus says meeting compliance requirements is not the only driving factor behind implementing a track and trace system, there are many other benefits, for example, a big one is safety.
“Quality and safety are our top priorities at Virtus," he says. “This system will help us better align our quality processes. As our process develops and evolves it will enhance our systems so that we can be properly prepared in case any issues arise."
Along with compliance and safety, there are other long-term benefits to the industry wide implementation, such as increased flexibility and efficiency, leading to cost savings.
“Track and trace solutions support pharmaceutical companies in competing more effectively," Ms. Lazar says. “The wealth of business intelligence data accumulated by such solutions can be used to increase sales and efficiency in processes."
This is done, she adds, through lower costs enabled by visibility of such key parameters as the most efficient sales outlets; better selling product categories; more efficient distribution, production and repackaging (if this occurs) processes.
In addition, the solution may also help organizations raise their green credentials due to lower resource usage in production and lower gas miles consumed by the distribution fleet.
The DSCSA regulations can make recall and return management faster and easier, says Dave DeJean, VP, center of excellence, Systech.
“Serialization will allow more proficient target recalls, as the supply chain partners will know immediately what product went to what store and when," he says. “This will help get unsafe products off the shelf faster."
It will also create a more accurate return process, as there will be up-to-date information available on how much product was initially shipped to each supplier.
“Rather than harmonizing return costs, companies can pay out specifically and that is a big benefit," Mr. DeJean says.
The accuracy and volume of internal data available will also create a more efficient supply chain.
Data regarding packaging lines, inventory turns, and a real wealth of data down to the unit can provide companies with information where their products are and where they are in their internal operation and externally where they are in the supply chain.
Serialization can also be used to boost brand engagement, Mr. DeJean says.
“Serialization provides a level of confidence in consumers that the product they are buying is authentic, but it can also be used to target content such as instructions and medical information to pharmacists or individual consumers."
Short-Term vs. Long-Term Planning
As the January 2015 deadline passes, companies will be in three categories of readiness, Mr. Dahod says. One group will be compliant or nearly compliant using a short-term fix; one group will be compliant with a well-thought out, long-term solution; and one group will lag behind all others, struggling to catch up to the missed deadline.
Mr. Dahod says his conversations about the implementation of serialization has illustrated that some companies will still be scrambling in January, caught unaware, or will be unprepared.
He says when speaking to industry leaders in the months before the first deadline, he found that many leaders had failed to make a decision on how to proceed or were unaware of the compliance requirements.
“We expect there will be some companies struggling in January because they have not even learned enough about the DSCSA to know yet what it will take to be compliant," he says “These companies are putting their relationships with wholesale partners who need to receive their product at risk."
The largest group will most likely be those with short-term goals for meeting the requirements. Mr. Dahod estimates that almost half of the industry is working on the DSCSA requirements with short-term glasses on.
This is not an uncommon response to compliance deadlines, he says, however, it is not the wisest decision, as companies will have to reconfigure the system for longer term use as more deadlines need to be met and requirements change from where they stand today.
“Many customers are taking short cuts and so their efforts are not scalable, and they will need to replace those short cuts with real solutions so that they can continue to meet requirements," he says. “A short-term focus is not viable, or wise, but often, companies have no choice, as time to make the deadline is running out and they need something in place. A short-term solution will work until they get the timing and budget over the next year or two to implement on a larger scale."
The natural inclination is to focus only on the short-term requirements of the 2017 deadline. Mr. DeJean suggests companies need to plan for the long term, look ahead to all of the requirements needed to be compliant in 2023, and build the entire system, and only implement the different functions as needed.
That is a more cost-effective and timely approach than having to continually modify a system built around a one-time, short-term goal.
“We see a lot of companies making short-sighted decisions based on the first thing they have to do — they have to serialize on the line — so they make a decision on packaging equipment and make that their strategy, rather than looking long term at the ever-changing landscape," Mr. DeJean says. “Companies should be aware that whatever they do today, in a few years, it will be different, so systems need to be configurable rather than customized and they have to have the capability to adapt and support new or changing regulations in the future."
Challenges Ahead
The guidance brings a promise of a more secure drug delivery system for all stakeholders — pharma, physicians, pharmacists, and patients — but it also brings seemingly insurmountable challenges in terms of data management and implementation.
“These types of systems touch every part of a businesses supply chain," Mr. DeJean says. “Everything is being impacted by these regulations."
So buy in from the top is crucial to implementing a system that will operate across all internal functions, as well as be applicable for external partners. Getting stakeholder inclusion is not only a challenge, but also a best practice, according to Mr. DeJean.
“Companies need to get everyone on board to make sure all the right decisions are being made in all the right places," he says. “From customer care, packaging, IT, and contracts with wholesalers, it all must align in a very complex system."
On top of the complexity, companies are also faced with a scale well-beyond anything they could anticipate. This is a challenge that most companies are not aware of at the start of the process. From a practice perspective manufacturers need to understand the scale of the data and transactions that have to be processed. There are many layers of processes across networks that need to be integrated and managed.
“Many companies vastly underestimate the scale of the amount of data that will need to be generated and the scale of transaction processes that need to occur for them to complete all their operations," Mr. Dahod says. “For example, if a company is shipping 100 million units a year, that becomes 100 million units a year within the ERP system, including all documented quantities, purchase orders, internal documents, and production orders. Then if those 100 million units have to be stored and accounted under seven transactions, pretty quickly within a year, companies are managing 700 million transactions. Manufacturers have never had to deal with anything along this magnitude; they need to really understand the scale and what other systems and processes will be impacted and how to be properly prepared."
Another challenge is integrating an effective IT solution, since the service industry has not yet caught up to providing solutions for the newer regulatory requirements.
 “IT integration seems to be the most difficult hurdle to overcome," says Paul Alexander, global engineering director, Catalent Pharma Solutions. “Reliable middleware or mapping service providers are still developing their offerings and this may slow down the implementation process.
“IT integration seems to be the most difficult hurdle to overcome," says Paul Alexander, global engineering director, Catalent Pharma Solutions. “Reliable middleware or mapping service providers are still developing their offerings and this may slow down the implementation process.
“Additionally, regulatory and client requirements are not always harmonized and do not always follow the same standards, such as GS1, leading to a greater number of customization needs," he says.
In the meantime, Mr. Alexander suggests that manufacturers develop a serialization master plan for a global implementation with baked-in flexibility. Teams must also be assembled to coordinate between external regulatory bodies and clients.
According to Mr. Alexander, a best practice is to break the project up into manageable sub-tasks or categories, allowing execution in a staggered, parallel implementation.
Recommended project categories include, but are not limited to: the planned plant or line level solution; Electronic Product Code Information Services (EPCIS) and/or Number Range Management (NRM); the internal interfaces at plant or line level; how warehouse management will work; and integration for transition from one business to another.
Manufacturers also need to study their supply chain to understand for which markets existing and future products are destined.
As part of this study, they also need to determine if aggregation is required to support their own, or their customer’s, supply chain needs. Although aggregation is not always a regulatory requirement, it may be difficult to split or reassemble downstream pallets if one cannot easily identify the contents of each shipper case without opening the box.
“Manufacturers should not just think about packaging when implementing serialization, but also consider what is needed in case of warehouse damage, whether additional sampling is required, and if an automated solution for rework is warranted to assure data integrity," Mr. Alexander says.
As the DSCSA takes hold in the United States, there are other track and trace regulations already in effect, or proposed, all over the world. Track and trace regulations exist in the EU, Brazil, China, Turkey, Korea, and Argentina.
Our experts say by 2018 all of Europe will have to be serialized. There are pending regulations proposed in Saudi Arabia, Jordan, Ukraine, Columbia, and Mexico.
According to TraceLink, 75% to 80% of the global drug supply will be covered under global track and trace regulations by 2018 — more than 10 billions of units a year that companies across the industry need to manage.
This creates a huge complexity from a project stand point because they have to look at each one of those serialization requirements and look at what is different and make sure their one system can support those differences, Mr. DeJean says.
If a manufacturer doesn’t get the system correct, it runs the risk of not being able to move its product through the supply chain.
“The teams working on this — both within manufacturers and from the solution provider standpoint — are doing some very heavy lifting," he says. (PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Thought leader quotes:
 The scale of the generated data and number of processes that has to occur to complete all the DSCSA operations is larger than anything the industry has known.
The scale of the generated data and number of processes that has to occur to complete all the DSCSA operations is larger than anything the industry has known.
Shabbir Dahod / TraceLink
 Manufacturers should not focus solely on packaging when implementing serialization.
Manufacturers should not focus solely on packaging when implementing serialization.
Paul Alexander / Catalent
~~~~~~~~~~~~~~~~~~~~~~~~~
DSCSA Guidance Overview
- Organizations covered by the guidance: Basically any manufacturers, wholesale distributors, repackagers, and dispensers that have a DSCSA compliance requirement.
- Products covered by the guidance: Basically the products defined in DSCSA section 581(13) as having a DSCSA compliance requirement.
- Transactions covered by the guidance: Basically all transactions as defined under DSCSA section 581(13) not explicitly exempted in the law.
Source: FDA
~~~~~~~~~~~~~~~~~~~~~~~~~


















