Portfolio value is traditionally associated with innovation value; i.e., intellectual property, market attractiveness, potential clinical utility, and other product-centric attributes. Less commonly considered is the differentiation afforded by the partnering process employed during clinical development. Maximizing portfolio value by considering a wider audience of stakeholders in key outsourcing decisions thus becomes an exercise as valuable as any in the discovery/development process.
Portfolio value is traditionally associated with innovation value; i.e., intellectual property, market attractiveness, potential clinical utility, and other product-centric attributes. Less commonly considered is the differentiation afforded by the partnering process employed during clinical development. Maximizing portfolio value by considering a wider audience of stakeholders in key outsourcing decisions thus becomes an exercise as valuable as any in the discovery/development process.
The decision to outsource or to retain development services is multivariate. The long and occasionally circuitous pathway from concept to IND/IDE submission and new product approval occasionally precludes linear, codified models of compound evaluation.
In this environment, competency and integration across disciplines and partners is a prerequisite for success. Given the many antecedents and inflection points which vary in timing and complexity, CRO/sponsor alignment becomes a differentiator.
That Was Then, This Is Now
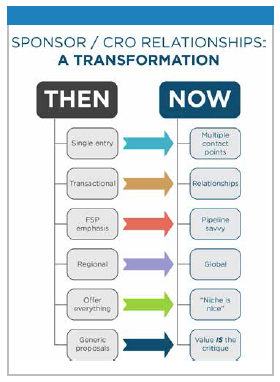
Originally built upon service competency and extension of consulting service, the current CRO business model may have little resemblance to those initially providing a catalyst for these services. For example, sponsor/CRO relationships historically were built upon single entry points for engagement, transactional relationships characteristic of functional service providers, regional as opposed to global capabilities, and often resulted in proposals which failed to highlight the potential for differentiated CRO services.
Now, CRO/sponsor engagements are exemplified by a need to manage multiple contact points, exquisite sensitivity to relationship management, and a CRO staff that is “pipeline savvy."
Global presence is needed not only to enhance patient access for later phase trials and to provide a platform for international  commercialization, but also to facilitate access to difficult to find and more difficult to treat patients for niche indications evaluating complex patient phenotypes and innovative therapeutics. Indeed value often is in the critique provided by a CRO of a proposed protocol and operational plan.
commercialization, but also to facilitate access to difficult to find and more difficult to treat patients for niche indications evaluating complex patient phenotypes and innovative therapeutics. Indeed value often is in the critique provided by a CRO of a proposed protocol and operational plan.
Moving from an R&D development practice in which scale, infrastructure, and processes dominated, the new relationship model is one that additionally must acknowledge contributions from many staff with remarkably different perspectives.
Sponsor/CRO relationships now are strategic, reflect a return to deep science for both products and therapeutic areas, and are characterized by clinical development programs increasing in complexity with demands for value-added activities.
Defining the Box, Then Stepping Out of It
The relationship between cause and effect in therapeutic innovation and advanced clinical trial methodology remains controversial.
Demands for repurposed products, novel chemical or biological entities require proficiency with both established, as well as remarkably innovative clinical development programs.
 Although there is “portfolio value" in recapitulating well-trodden, predictable development pathways, there is parallel increasing emphasis for a “therapeutic neighbors" strategy within clinical development in which pharmacological targets — rather than indications — are initially evaluated. This can be prevalent in rare and orphan disease drug development where experience with both product and the indication are insufficient to enable confidence in subsequent program parameters.
Although there is “portfolio value" in recapitulating well-trodden, predictable development pathways, there is parallel increasing emphasis for a “therapeutic neighbors" strategy within clinical development in which pharmacological targets — rather than indications — are initially evaluated. This can be prevalent in rare and orphan disease drug development where experience with both product and the indication are insufficient to enable confidence in subsequent program parameters.
In this mosaic, a portfolio of clinical development options extracted from experiences across — as well as within — multiple therapeutic areas, becomes an invaluable asset given that precedent may have only limited utility, and predictable development to inflection points becomes a key determinant of company value.
Looking Beyond the Obvious
Current research and development efforts occur in increasingly complex healthcare settings. Neither orderly nor entirely rational, stakeholders introduce requirements for diverse data perspectives from patients, physicians, caregivers, and payers. Given that formulary placement and reimbursement mechanisms have a dominant role in determining adoption, coverage, pricing, and ultimately patient access, a unique set of guidances is required to design programs which would resonate in that environment, while simultaneously securing product registration.
Value from this perspective is achieved by demonstrating improved patient outcomes with reductions in healthcare utilization and cost.
A development program must therefore develop regulatory and peer-reviewed publications upon which decisions regarding product placement can be effected, include patients with concomitant medicines and comorbidities driving healthcare utilization, and assess outcomes representing quantifiable economic readouts (and not just measures). In this setting, it is the impact of innovative therapy on a healthcare system, as well as an individual patient which becomes paramount, and capturing those data is mandatory.
Demonstrating evidence-based value during development enhances portfolio value by supporting provider adoption, payer coverage, and premium pricing which will result in optimal payment and market access.
On the Importance of Passion
 Sponsors dictate key metrics for CRO engagement at study onset. CROs thus require both operational and therapeutic acumen to inform the many parameters that dictate success.
Sponsors dictate key metrics for CRO engagement at study onset. CROs thus require both operational and therapeutic acumen to inform the many parameters that dictate success.
Of the 17 items considered in a multivariate analysis describing the CRO selection process from a sponsor’s perspective, team chemistry and experience are most important. A project execution plan, aptitude in problem solving, and lastly, price were also key variables.
This relative weighting is anticipated given the existence of one- and two-product portfolios, which demand excellence on every study, the innovation in biologics, devices, and small molecules, and the increasing need for outsourcing on either a strategic, or niche level.
Partnership with a CRO that has “one ear to the ground, and one eye on heaven" with a commitment to provide sustained, differentiated services adds considerably to portfolio value. (PV)
Worldwide Clinical Trials, a global CRO, has the mission to facilitate the development of life-changing medicines by integrating clinical trials methodology, operational, and efficient technology.
For more information, visit wwctrials.com.