First and Only Vaccine Approved for Meningitis
Pfizer’s Trumenba a Key Public Health Advance
A serious and life-threatening disease can now be prevented with the first and only FDA-approved vaccine in its class. Pfizer’s Trumenba Meningococcal Group B Vaccine was granted accelerated approval by the FDA in October 2014.
The approval of Trumenba is an important public health advance in helping to protect adolescents and young adults — 10 to 25 years of age — from invasive meningococcal serogroup B disease, otherwise known as meningitis B.
 Vaccines have been available and recommended since 2005 to help protect against four other serogroups of meningococcal disease, and the hope is that Trumenba will become the recommended vaccine in routine adolescent immunization programs to help prevent meningococcal B disease.
Vaccines have been available and recommended since 2005 to help protect against four other serogroups of meningococcal disease, and the hope is that Trumenba will become the recommended vaccine in routine adolescent immunization programs to help prevent meningococcal B disease.
Meningococcal disease can be unpredictable and occur quickly and without warning. Outbreaks and cases of meningococcal group B disease occurred in the United States in 2013 at Princeton University and 2014 at the University of California Santa Barbara.

This strain of meningitis is characterized by high fatality rates and rapid onset, often within 24 hours. In the United States, for people 11 to 24 years of age, about 30% of meningococcal disease cases involve serogroup B, and 10% of these cases result in death. As many as 60% of adolescent survivors of meningococcal disease suffer from permanent life-altering consequences, such as hearing loss, neurologic damage, or loss of a limb.
Trumenba was approved through the FDA’s Breakthrough Therapy designation and Priority Review programs.
Ebola Research
Experimental Ebola Therapeutic Enters Trials
 Just a few short months ago, healthcare workers in Africa struggled to contain an outbreak of Ebola. With no options available, doctors turned to an experimental medication ZMapp, which is comprised of a series of three different monoclonal antibodies that work to prevent the spread of the disease within the body. The three different antibodies that comprise ZMapp are currently grown in tobacco plants. Antibody genes are infiltrated into tobacco plants to transiently manufacture the ZMapp antibodies.
Just a few short months ago, healthcare workers in Africa struggled to contain an outbreak of Ebola. With no options available, doctors turned to an experimental medication ZMapp, which is comprised of a series of three different monoclonal antibodies that work to prevent the spread of the disease within the body. The three different antibodies that comprise ZMapp are currently grown in tobacco plants. Antibody genes are infiltrated into tobacco plants to transiently manufacture the ZMapp antibodies.
The development of ZMapp is a group effort – a great example of scientists working together to get the best results. This public-private partnership is comprised of a consortium of scientists from the Public Health Agency of Canada, Defyrus, the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID), Kentucky BioProcessing, and Mapp Biopharmaceutical, among other institutions. Mapp Bio has been working on multiple plant-based antibody therapies and preventives since 2003.
While ZMapp appeared to stop the spread of the virus in monkeys, it had yet to be tested in humans.
Under an emergency protocol, ZMapp was given to nine patients, including the first two U.S. doctors working in Liberia who were infected in July 2014; they were treated, and survived.
Now, formal clinical trials of ZMapp are about to begin. In February, Mapp Biopharmaceutical received an IND from the FDA to begin clinical trials. The trials will begin in Liberia, and the Institutes of Health and the Liberian government will oversee the clinical trials.
 Breaking New Ground in Preventive Dental Care
Breaking New Ground in Preventive Dental Care
CHX’s Topical Antimicrobial
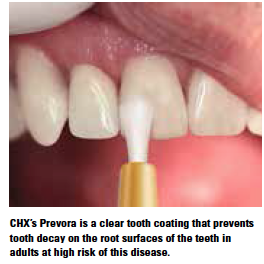
Awaiting approval from the FDA is a topical antimicrobial to treat the cause of dental decay. CHX Technologies’ Prevora is the only drug that treats the cause of dental caries (tooth decay) in adults. It is approved in Europe and Canada, and a new drug application is pending with the FDA.
Prevora delivers 10% chlorhexidine to the tooth surface where it remains at bactericidal concentrations for days and inhibitory concentrations for weeks. The coating is clear, readily applied, and has no adverse effects.
Dental caries remains one of the most prevalent chronic disease in both children and adults — 92% of adults 20 to 64 of age have had dental caries in their permanent teeth — even though it is largely preventable. Dental decay results from a chronic, low-grade asymptomatic bacterial infection.
In the United States, there is a growing aging population affected by a number of chronic diseases, which encourage dental decay. For example compared with healthy controls, a diabetic has two times the level of decay, some arthritic patients five times the decay, and Parkinson’s patients five times the decay. Certain medications such as diuretics and antidepressants often contribute to a dry mouth, which in turn, increases the risks of dental decay.
Dental decay is also linked to overall health. The tooth root is a significant risk factor for a coronary event, and is proven to be more important than cholesterol in heart health. Other studies have shown that the bacteria causing dental decay is the most common microorganism in diseased arterial plaque of end-stage patients in the CCU.
CHX is marketing Prevora in Canada using social media networks and a new point-of-purchase and point-of-advocacy group of dental teams called Partners in Prevention.
 A New Way to Treat Cancer
A New Way to Treat Cancer
Merck’s Keytruda is an Advance Over Current Therapies
Merck’s Keytruda was approved in the United States in September 2014 for patients with unresectable or metastatic melanoma and disease progression following treatment with ipilimumab, a type of immunotherapy.
Keytruda is the first approved drug in the United States that blocks a cellular pathway known as PD-1, which restricts the body’s immune system from attacking melanoma cells.
Melanoma, which accounts for about 5% of all new cancers in the United States, occurs when cancer cells form in skin cells that make the pigment responsible for color in the skin.
According to the National Cancer Institute, an estimated 76,100 Americans will be diagnosed with melanoma and 9,710 will die from the disease this year.
Keytruda, anti-PD-1 (programmed death receptor-1), is a humanized monoclonal antibody that works by blocking the interaction between PD-1 and its ligands, PD-L1 and PD-L2.
The FDA granted Keytruda breakthrough therapy designation because the sponsor, Merck, demonstrated through preliminary clinical evidence that the drug may offer a substantial improvement over available therapies.
Keytruda also received priority review and orphan product designation. Priority review is granted to drugs that have the potential, at the time the application was submitted, to be a significant improvement in safety or effectiveness in the treatment of a serious condition. Orphan product designation is given to drugs intended to treat rare diseases.
The product is also being studied in patients with classic Hodgkin lymphoma and in patients with advanced triple-negative breast cancer.
One Cell vs. Many Cancers
Unum’s Therapy Targets Many Cancer Types
Unum Therapeutics is using the immune system to fight cancer by developing cellular immunotherapies.
Just three months after its final financing round, Unum began its first clinical program based on its antibody-coupled T-cell receptor (ACTR) technology.
Recruitment is already under way for a Phase I clinical trial evaluating ATTCK20, the combination of ACTR T-cells with Rituxan, one of the earliest monoclonal antibodies to be approved by the FDA.
Researchers will test this concept at a variety of dose levels in patients with B-cell malignancies who haven’t responded to Rituxan or who have relapsed after treatment.
Following an initial proof-of-concept with Rituxan, Unum will explore combinations with other tumor targeting antibodies in other kinds of cancers.
The ultimate vision of the biotech company is to produce a single cell therapy that can drive patient cures in many different cancer types.
The breakthrough moment came when Dario Campana, the scientific founder of Unum, realized that it would be possible to effectively combine the useful properties of two different immune cells into a single novel cell type.
Natural killer cells (NK cells) are responsible for attacking infected or cancerous cells that have been recognized by antibodies. T-cells can also attack cells and additionally can proliferate and expand in response, meaning they can have a much more potent effect.
Unlike NK cells, T-cells however are unable to be targeted by antibodies. By fusing fragments from receptor genes expressed in NK cells and T-cells and then expressing the chimera in a T-cell, Unum’s researchers were able to create what is effectively a new type of immune cell — an antibody-targetable T-cell. (PV)
—————————————————————————-
Drug Therapies
ACTR
• unumrx.com/#!technology/c1iwz
Keytruda
• keytruda.com
Prevora
• prevora.com
Trumenba
• trumenba.com
ZMapp
• mappbio.com/zmappfaq.pdf


















