Industry experts discuss the various avenues — immunotherapy, epigenetics, biomarkers, and other targeting technology — companies are taking to address cancer research.
Great progress has been made in the fight against cancer. Advances in molecular and genomic research have unveiled complexities and 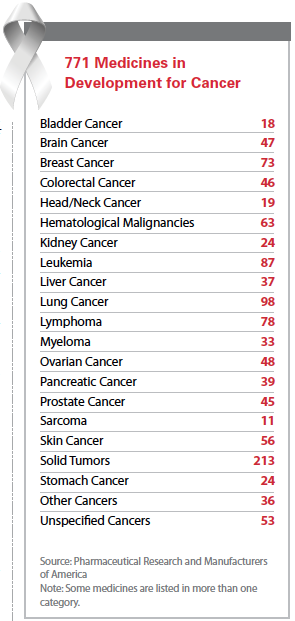 changed the perception of cancer, which we now know is more than 200 unique diseases. Our understanding of cancer has grown, and as a result, treatments are targeting the molecular triggers that cause normal cells to become cancerous. Researchers are using new technologies — from the fields of computational chemistry, imaging technology, nanotechnology, and health information — to develop new avenues of cancer treatments.
changed the perception of cancer, which we now know is more than 200 unique diseases. Our understanding of cancer has grown, and as a result, treatments are targeting the molecular triggers that cause normal cells to become cancerous. Researchers are using new technologies — from the fields of computational chemistry, imaging technology, nanotechnology, and health information — to develop new avenues of cancer treatments.
Although great progress has been made, cancer remains the second-leading cause of death in the United States behind heart disease. Many cancers are not detected until their latest stages, and others are resistant to treatment.
Pharmaceutical company research investments into finding efficacious treatments remain high with cancer therapies accounting for more than 30% of all preclinical and Phase I clinical development projects; 22 new molecular entities were launched in the last two years alone, according to a report from IMS Institute for Healthcare Informatics.
 Cancer remains the biggest portion of the overall drug development pipeline in the earlier phases with four times the number of drugs in the pipeline than the next largest therapeutic class, according to IMS Institute for Healthcare Informatics. After an innovation slowdown through 2008, the oncology pipeline has increased with more accelerated approvals (34% of breakthrough therapy designations are for cancer) and a shift to non-biologics. But there are fewer cancer drugs progressing to Phase II and III trials, which indicates both the high levels of early-phase activity and the difficulties in generating successful results in the clinic, according to IMS.
Cancer remains the biggest portion of the overall drug development pipeline in the earlier phases with four times the number of drugs in the pipeline than the next largest therapeutic class, according to IMS Institute for Healthcare Informatics. After an innovation slowdown through 2008, the oncology pipeline has increased with more accelerated approvals (34% of breakthrough therapy designations are for cancer) and a shift to non-biologics. But there are fewer cancer drugs progressing to Phase II and III trials, which indicates both the high levels of early-phase activity and the difficulties in generating successful results in the clinic, according to IMS.
Many of the medicines in the pipeline today are using novel approaches to attack cancer at the molecular level. About 80% of cancer pipeline drugs are potentially first-in-class treatments, according to the Tufts Center for the Study of Drug Development.
There are 771 medicines and vaccines either in clinical trials or awaiting review by the FDA, according to a September 2014 report from the Pharmaceutical Research and Manufacturers of America (PhRMA).
Currently, there are 3,137 active cancer clinical trials in the United States. Of those, 1,824 are now seeking volunteers to participate or have not yet started recruiting patients.
Immunotherapy
A hot area of cancer research is immunotherapy or immuno-oncology, which is expected to be a game-changing approach to treating cancer. Experts say immuno-oncology may well constitute a new mode of cancer treatment, alongside surgery and chemotherapy.
Researchers have been studying the immune system and cancer for some time. But experts say the approval of Provenge, Dendreon’s immunotherapy for advanced prostate cancer, provided the evidence that the immune system could be triggered to fight cancer. Provenge, approved in the United States in April 2010 and in the EU in 2013, is an autologous cellular immunotherapy that uses a patient’s own cells to stimulate an immune response against cancer.
More recently, two companies — Bristol-Myers Squibb and Merck — received U.S. approval in 2014 for programmed death receptor-1 (PD-1) blocking antibodies. These are “checkpoint inhibitors," a class of monoclonal antibodies that inhibit pathways responsible for blocking the response of T-cells to antigens. This class works to overcome mechanisms that tumors use to resist T cell-mediated antitumor immunity.
Bristol-Myers Squibb in December 2014 received approval for Opdivo (nivolumab) injection. Opdivo is a human PD-1 blocking antibody indicated for the treatment of patients with unresectable or metastatic melanoma and disease progression following therapy with Yervoy (ipilimumab.)
In September 2014, Merck received U.S. approval for Keytruda for patients with unresectable or metastatic melanoma and disease progression following treatment with ipilimumab.
Other companies also are working on PD-1 research, which is based on the interaction between PD-1 and its ligands, PD-L1 and PD-L2.
For example, in November 2014, Pfizer and Merck KGaA announced an agreement to jointly develop and commercialize MSB0010718C, an  investigational anti-PD-L1 antibody currently in two development programs in Merck KGaA’s pipeline as a potential treatment for multiple types of cancer. In a Phase I trial, more than 550 patients have been treated with MSB0010718C across multiple types of cancers.
investigational anti-PD-L1 antibody currently in two development programs in Merck KGaA’s pipeline as a potential treatment for multiple types of cancer. In a Phase I trial, more than 550 patients have been treated with MSB0010718C across multiple types of cancers.
Pfizer is also working separately on other programs in immuno-oncology and actively exploring a variety of novel agents, including checkpoint modulating antibodies, CAR-T therapies, bi-functional monoclonal antibodies, and vaccine-based immunotherapy regimens.
Pfizer’s 4-1BB agonist antibody is currently in Phase I trials, with several other immunotherapeutic agents expected to begin clinical testing in 2015, including a monoclonal antibody against receptor OX40 (CD134), a PD-1 monoclonal antibody, and a vaccine-based regimen for prostate cancer.
Pfizer is exploring combining immunotherapies with its oncology portfolio through its own development efforts as well as in collaboration with partners.
Other companies also are approaching stimulating the immune system against cancer from different avenues.
Cel-Sci, for example, is using TNF alpha to kill cancer cells. The company’s Multikine is injected around the tumor. One of the components of Multikine, TNF alpha, will then kill cancer cells, which releases antigens specific to that person’s own cancer. The immune response then occurs against that person’s antigen.
“This is important because cancer patients could be expressing a different antigen because each person is genetically different," says Geert Kersten, CEO of Cel-Sci Corp.
“Think of it as an in vivo vaccination against a tumor."
The company is conducting a large Phase III clinical study with Multikine in head and neck cancer patients with advanced disease who are treatment naïve (i.e., have received no prior treatment). The current recommendation for initial treatment of these patients is surgery followed by radiotherapy.
In Cel-Sci’s ongoing study, patients are administered Multikine before these other treatments are given.
Mr. Kersten points out that head and neck cancer remains an area of unmet need.
People with this cancer generally are treated with surgery, followed by radiation and chemotherapy. Overall, mortality rates for head and neck cancers have declined since 2001, according to the National Cancer Institute.
Still, an estimated 55,070 people developed head and neck cancer in 2014 and there were an estimated 12,000 deaths, according to the American Society of Clinical Oncology.
Mr. Kersten says in the trials Multikine is being given for three weeks before patients undergo surgery and chemotherapy. The impact of the therapy will be measured through an increase in overall survival.
“The primary goal is not the elimination of the tumor, which the surgeon will remove," he says. “The goal is the elimination of the micrometastases in order to reduce recurrence rate. If we can reduce recurrence, we can increase survival rates. We are given only three weeks for Multikine to work because an experimental drug is not allowed to delay a proven therapy.
“The most logical time to boost the immune system is while patients still have an intact immune system," he continues. “We believe that immunotherapy has a much lesser chance of working in recurrent cancer because the lymphatic system has likely been affected by surgery, therefore the immune system cells can no longer travel around. Additionally, the immune system has been weakened by chemotherapy and radiation."
Phase II trial results of Multikine show that 12% of patients had zero remaining cancer cells by pathology. Of the remaining patients, cells examined on slides showed that the number of cancer cells was 50% less when compared with controls.
Mr. Kersten says the FDA would like to see a 10% increase in overall survival rates as an endpoint for Phase III trials.
Standard chemotherapy can have a devastating impact on a person’s immune system.
Yaniv Bejerano, co-founder and CEO of ImmuneXcite, says company researchers are designing an immunotherapy that is at the intersection of innate and adaptive immunity. Using neutrophils as the initiator of this immune cascade, a type of white blood cell that is an essential part of the immune system, ImmuneXcite is able to bring a robust adaptive immune response.
Neutrophils are the first line of defense against infectious agents and are effective killers of bacteria and fungi. ImmuneXcite’s co-founders have identified a unique carbohydrate from fungal cell walls that signals neutrophils. The company’s technology enables the generation of monoclonal antibody constructs with a novel effector function that generates a cascade of immune response that starts with neutrophils and results in immunity to cancer.
“Chemotherapy can diminish the number of neutrophils that are in essence the initiator of the cascade we are researching," Mr. Bejerano says. “As part of the trial design, inclusion criteria will include patients having a minimal number of neutrophil counts because they may be susceptible to fungal infections."
ImmuneXcite’s product candidate is being studied in preclinical trials for colorectal cancer, and the company’s executives say they hope to begin clinical trials in the first half of 2016.
“We are pursuing an indication in colorectal cancer, specifically patients who have overexpression of EGFR but also have a mutation of KRAS and BRAF that prevents them from responding to EGFR therapy," he says. “Right now, these patients don’t have a lot of options because often chemotherapy doesn’t work for them."
Ifat Rubin-Bejerano, Ph.D., co-founder and chief scientific officer at ImmuneXcite, says the hope is that the Phase I trial will also show an association between efficacy and neutrophil infiltration into tumors.
“We are looking to use the neutrophil infiltration as a pharmacodynamic marker in a Phase I trial that would provide a proof of the mechanism in humans and assist in dose selection," she says. “This will be achieved using an imaging technique that is approved for the detection of neutrophils, which is not generally used in the context of cancer."
Epigenetics
Researchers have discovered that cancer can be influenced by changes in gene expression caused not only by genetic mutations but also by chemical modifications of DNA, which are called epigenetic changes. Epigenetics is a regulatory system that controls gene expression without affecting the makeup of the genes themselves.
Regulation of gene transcription has emerged as a key biological determinant of protein production and cellular differentiation and plays a significant pathogenic role in a number of human diseases.
“When the regulation mechanism is disrupted, gene expression is disrupted and that can lead to misregulated cell growth either by an inability to divide or an inability of cells to develop properly," says Robert Gould, Ph.D., CEO of Epizyme. “This is manifested in human beings as cancer — either solid or blood-borne cancers."
Epizyme is working on identifying the genetic alterations that create oncogenes (cancer causing genes) and then designing small molecule therapeutics to inhibit the oncogene. The company’s focus is on creating small molecule inhibitors of histone methyltransferases (HMTs), a class of epigenetic enzymes.
Since the human genome has been mapped, many cancers have had their DNA sequenced. Genetic changes in the DNA code that differ from a cancer cell and a normal cell can be changed in these epigenetic enzymes, which control gene expression.
In these cases, the alterations within these enzymes seem to be critical to misregulated cellular growth activity, Dr. Gould says.
“One distinguishing factor about epigenetics is that it’s attacking a fundamental change within the cell that is contributing to the aberrant cellular growth," he says. “Through our contributions to the field, we’ve learned there’s a particular family of enzymes called HMTs that play a critical role in gene expression. It is a large family of enzymes, which we have defined as having 96 members. We further identified about 20 of those 96 members as being critically involved in cancer cell growth versus normal cell growth, and we have created chemical matter against 13 of those 20 targets. We think there is a huge opportunity through HMTs to attack a fundamental driver of a variety of solid cancers, such as hematologic cancers."
The global epigenetics market was valued at an estimated $413.24 million in 2014 and is expected to grow at a CAGR of 13.64% between 2014 and 2019 to reach $783.17 million in 2019, according to MarketsandMarkets.
Furthermore, the oncology research segment accounts for the largest share of the epigenetics market.
Epizyme has two clinical programs. One is an inhibitor of an enzyme called DOT1L, which plays a critical role in controlling gene expression. This particular enzyme is misregulated in a subset of acute leukemia. DOT1L is in Phase I trials to treat MLL, mixed lineage leukemia, meaning the disease has characteristics of both acute lymphoblastic leukemia (ALL) and acute myeloid leukemia (AML).
A second Epizyme program is focused on a small molecule inhibitor of a different enzyme called EZH2, whose activity is misregulated in non-Hodgkin lymphoma. Epizyme and its partner Eisai are planning a Phase II trial to begin in 2015.
Biomarkers
Patients with melanoma, metastatic lung, breast, and brain cancers as well as leukemia are now being routinely offered a molecular diagnosis to allow their physicians to select tailored treatments. A majority of companies have embraced personalized medicine, and almost half of the preclinical and Phase I assets in the pharma pipeline have associated diagnostics, especially in oncology, immunology, and CNS, according to a 2013 analysis by McKinsey.
Oncology is one of the most active areas of research of personalized medicines. Examples of such targeted therapies on the market include Novartis’ Gleevec, AstraZeneca’s Iressa, and Genentech’s Tarceva.
Decision Resources predicts oncology therapies will continue to dominate the predictive personalized medicine market, capturing 88% of U.S. sales in 2019. In addition, predictive personalized drugs to treat cancer indications are forecast to account for more than one-third of total U.S. oncology sales in 2019.
Shaji Kumar, M.D., a principal investigator and professor of medicine at the Mayo Clinic College of Medicine, says the increasing application of genetic techniques to understanding cancers have enabled researchers to identify specific changes in subgroups of patients, who can then be targeted for therapy.
“We are starting to understand that cancers are very different diseases," he says. “Even cancers that we call by one name are very different in different people. This concept of targeting patients based on specific abnormalities is in the very early stages and only time will tell if this is the right way to go partly because cancer changes over time."
Dr. Kumar is an investigator working with Millennium on the company’s MLN9708, an oral proteasome inhibitor for multiple myeloma.
The product is currently in Phase III trials. Data from a Phase II clinical trial show ixazomib has the potential to extend depth of response for patients following therapy with ixazomib-lenalidomide-dexamethasone. Millennium’s MLN9708 will offer an alternative to Velcade, which is commonly used in treating myeloma.
Paul Harkin, managing director of diagnostics at Almac agrees that oncology drug development is experiencing a dramatic change because of biomarkers.
“The majority of the companies we work with have biomarkers linked to their investigational products," he says. “We are seeing quite an upsurge in the number of biomarker-driven clinical trials. Patients are being selected for participation in trials based on an investigational biomarker inclusion criteria. The majority of the industry is conducting Phase I and Phase II trials with biomarker-driven trials, and the true impact of the diagnostic on drug development won’t be seen for another 10 years."
Mr. Harkin points out there are additional costs in biomarker-driven trials.
“In early development phases, it is more costly to include a biomarker with a therapeutic," he says. “And the entire trial is more complicated because we can’t just enroll all comers. Patients must be preselected, and to do this we need to have the test results back before the patient can be enrolled in the trial."
Individual companies may not be able to take advantage of scale in terms of testing. When screening patients for a trial, companies may only do 50 or so tests over the trial and samples come in one at a time, Mr. Harkin says.
“We have to dedicate full-time resources to an individual sample to turn it around in seven days," he says. “The resource allocation of what’s required to run these types of tests hasn’t penetrated yet with sponsors."
Mr. Harkin says the hope — and the expectation — is that the increased costs that are seen in Phase I and Phase II trials will be offset by an increase in the number of drugs that are successful in Phase III trials.
Dirk Reitsma, M.D., VP of global product development and therapeutic area head for oncology at PPD, says another challenge with  biomarker trials is the need to screen so many more patients to find those who have a specific subtype of cancer.
biomarker trials is the need to screen so many more patients to find those who have a specific subtype of cancer.
“We had one client that needed about 200 patients for a trial, but in order to find those patients, the company had to screen about 2,000 patients," Dr. Reitsma says. “This is a big challenge and it is altering the way we have to run trials."
Companies are starting to develop partnerships to take advantage of scale in screening as a way to accelerate development timelines. Some are participating in multi-entity, multi-arm, multi-drug protocols.
One of the first of these new development models was the I-SPY trials for breast cancer. I-SPY 2 was launched in 2010 and is a clinical trial for women with newly diagnosed, locally advanced breast cancer to test whether adding investigational drugs to standard chemotherapy is better than standard chemotherapy alone before having surgery.
The trial is sponsored by the Biomarkers Consortium, a partnership led by the Foundation for the National Institutes of Health (FNIH), the FDA, the National Institutes of Health (NIH), and a large number of partners from major pharmaceutical companies, leading academic medical centers, and nonprofit and patient advocacy groups.
A distinctive feature of the trial is that it screens multiple drugs from multiple companies, up to 12 different cancer drugs over the course of the trial. This allows the I-SPY 2 team to graduate, drop, and add drugs throughout the course of the trial without having to stop the trial to write a whole new protocol.
Dr. Reitsma predicts trials similar to the I-SPY protocol will become the workhorses of future drug development.
“Some may see this as a competitive trial, but one drug may work best in one cancer subtype and another may work best in another subtype so, for example, it is unlikely that one drug will emerge the winner to treat all types of breast cancers," Dr. Reitsma says. “Many of the drugs in this trial are going to end up winners. And so far, that’s what has happened. The two drugs that have graduated in their biomarker defined subsets treat different subsets of patients and will not be in competition with each other."
Other Research Areas
Other companies are using additional types of targeting technologies, including antibody drug conjugates, which pair up monoclonal antibodies (mAB) with anticancer agents.
One company in this space is Actinium Pharmaceuticals. The company’s product candidates are based on technology co-developed with Memorial Sloan Kettering Cancer Center (MSKCC) that combine the cancer targeting precision of mAb for targeting specific types of cells with the power of alpha emitting radioisotopes and a broadly applicable antibody labeled with beta-emitting radioisotopesin-licensed from Fred Hutchinson Cancer Research Center.
“It has been known for some time that monoclonal antibodies are very efficient in targeting cancer calls," says Dragan Cicic, M.D., chief medical officer of Actinium Pharma. “Each type of cell has a set of markers on the surface, which distinguishes that type of cell from other cells. We can use monoclonal antibodies that hone in only on certain markers on the cell, i.e., the type of cells we want to target. But in many cases cancer cells have developed various defenses that prevent antibodies from causing them much damage even when they target them successfully."
Actinium’s technology attaches sources of radiation to each antibody so when a cancer cell tries to defend itself it is met with the radiation. This technology can deliver radiation directly to cancer cells with minimal effect on healthy tissues.
Dr. Cicic says the company is preparing to launch a Phase III trial in mid 2015 for Iomab-B for bone marrow conditioning prior to transplant in elderly relapsed/refractory acute myeloid leukemia (AML) patients.
Iomab-B has already been used as a myeloconditioning/myeloablative agent in more than 250 patients with incurable blood cancers.
He says a Phase II clinical trial showed a 100% complete response rate, and the endpoint for Phase III will be durable complete response, which is defined as complete response lasting at least six months.
Iomab-B will be used in preparing patients for hematopoietic stem cell transplantation.
“We are going to compare how many patients had a lasting response in the study compared with the control group and the control group will be physicians’ choice of chemotherapy followed by transplant," Dr. Cicic says.
Another Actinium product is Actimab-A, which is a radiolabeled antibody being developed for newly diagnosed AML in patients over 60, and is currently in a multicenter Phase I/II clinical trial.
The company also has other drug candidates that are in early development for other cancers.(PV)
~~~~~~~~~~~~~~~~~~~~~~~~~~~~
A New Paradigm for Patient Recruitment
Recruiting patients for oncology clinical trials remains a problem and only about 3% of all cancer patients enroll in a clinical trial. Among the many challenges are: limited availability of clinical trials in community oncology settings, a minority of cancer patients seeking out clinical trials, cancer patients often being older with other illnesses and which preclude them from — participating in trial protocols, and physicians have busy practices and screening patients for clinical trials can be time consuming.
“Information we get from clinical trials tend to be somewhat biased because the vast majority of the cancer patients don’t participate clinical trials," says Shaji Kumar, M.D., a principal investigator and professor of medicine at the Mayo Clinic College of Medicine. “This means we may end up with a different picture of cancer treatment from what it actually is. Hence, it is important we also carefully obtain information from real-life settings to supplement the information from clinical trials."
As pharmaceutical companies continue to develop more specific and targeted therapies, often based on molecular or genomic abnormalities, finding and enrolling those patients with the specific alterations can be challenging, leading to more complex trials and longer development timelines.
Phil Breitfeld M.D., VP, head of the Oncology Center of Excellence at Quintiles, believes there is a solution imbedded in that complexity.
“We and others view this as an opportunity to preprofile cancer patients at initial diagnosis by identifying their molecular or genomic abnormalities ahead of the clinical trial opportunity so that now we know where those patients are and we can better recruit them into the appropriate clinical trials."
He points out the costs of molecular profiling are decreasing, and over time it will become more cost-effective to prescreen patients using a molecular panel instead of screening patients for each trial. This genomic data could be used to create a registry of patients with different molecular abnormalities from which to recruit patients.
“We think in the long run, the economics of this will be really clear and there will be wider adoption of this idea," Dr. Breitfeld says. “In a future world, we could imagine delivering trials to sites with eligible patients, which reverses the paradigm that we have today, where we recruit sites and hope patients find the right studies."
There are initiatives already in place to test such a way to find patients. One is the LungCancer Master Protocol, which began in
November 2013, and is an alliance of several partners: the National Cancer Institute; the Foundation of the National Institutes of Health; the Food and Drug Administration; and the advocacy group, Friends of Cancer Research. The trial will be a multi-drug, multi-arm protocol that will evaluate five compounds intended to treat squamous cell lung cancer.
Genomic screening will be used to assign enrolled patients to the treatment arms most likely to provide benefit. The idea is to streamline the drug-approval process by bringing pharma companies together to test multiple experimental drugs in late-stage clinical trials under a single, master protocol.
The five products selected to participate include: MEDI4736, from MedImmune, an anti-PD-L1 monoclonal antibody; AZD4547, from AstraZeneca, a FGFR tyrosine kinase inhibitor; rilotumumab, from Amgen, a hepatocyte growth factor receptor c-MET inhibitor; pictilisib, from Genentech/Roche, a PI3 kinase inhibitor; and Palbociclib, from Pfizer, a CDK4/6 kinase inhibitor.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Thought leader quotes
 The immune oncology space has become very competitive in the last few years, and there are many companies pursing the same types of
The immune oncology space has become very competitive in the last few years, and there are many companies pursing the same types of
molecules.
Yaniv Bejerano/ ImmuneXcite
 Anyone who knows someone who has been treated with chemotherapy knows how devastating it can be. If there is a way to achieve a result without the side effects, this would be a great advance.
Anyone who knows someone who has been treated with chemotherapy knows how devastating it can be. If there is a way to achieve a result without the side effects, this would be a great advance.
Dr. Ifat Rubin-Bejerano/ ImmuneXcite
 Ten years from now, we are going to be able to identify and categorize cancers by the type of genetic changes and then choose therapy based on those changes.
Ten years from now, we are going to be able to identify and categorize cancers by the type of genetic changes and then choose therapy based on those changes.
Dr. Shaji Kumar / Mayo Clinic College of Medicine
 One challenge with biomarker trials is the need to screen many patients to find those with a specific subtype of cancer. This is altering the way we run trials.
One challenge with biomarker trials is the need to screen many patients to find those with a specific subtype of cancer. This is altering the way we run trials.
Dr. Dirk Reitsma / PPD
 The majority of the companies we work with have biomarkers linked to investigational products. We’re seeing quite an upsurge in the number of biomarker-driven clinical trials.
The majority of the companies we work with have biomarkers linked to investigational products. We’re seeing quite an upsurge in the number of biomarker-driven clinical trials.
Paul Harkin / Almac
~~~~~~~~~~~~~~~~~~~~~~~~~~~~


















