Oncology is expected to be the biggest therapeutic class by 2017. In this first article in an ongoing series addressing various aspects involved with oncology, experts examine the market drivers, research highlights, and marketing, sales, and payer considerations of this market.
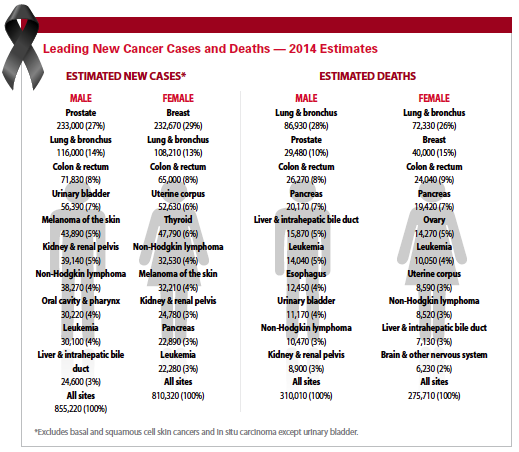
In 2014, there were an estimated 1,665,540 new cancer cases diagnosed and 585,720 cancer deaths in the United States alone. Cancer remains the second most common cause of death in the United States, accounting for almost one of every four deaths.
Cancer is a group of diseases characterized by the uncontrolled growth and spread of abnormal cells. If the spread is not controlled, it can result in death. Cancer is caused by both external factors (tobacco, infectious organisms, chemicals, and radiation) and internal factors (inherited mutations, hormones, immune conditions, and mutations that occur from metabolism).
Cancer is treated with surgery, radiation, chemotherapy, hormone therapy, immune therapy, and targeted therapy. While oncology is considered one therapeutic category, there are more than 200 different types of cancer, which can affect any body organ.
 All cancers involve the malfunction of genes that control cell growth and division. Only a small proportion of cancers are strongly hereditary. Inherited factors play a larger role in determining risk for some cancers (e.g., colorectal, breast, and prostate) than for others. Genetic damage may result from internal factors, such as hormones or the metabolism of nutrients within cells, or external factors, such as tobacco, or excessive exposure to chemicals, sunlight, or ionizing radiation.
All cancers involve the malfunction of genes that control cell growth and division. Only a small proportion of cancers are strongly hereditary. Inherited factors play a larger role in determining risk for some cancers (e.g., colorectal, breast, and prostate) than for others. Genetic damage may result from internal factors, such as hormones or the metabolism of nutrients within cells, or external factors, such as tobacco, or excessive exposure to chemicals, sunlight, or ionizing radiation.
Great strides have been made toward improved cancer prevention, detection, diagnosis, and treatment. As a result, more people are living longer with cancer, and fewer people are dying from cancer. Research supported by pharmaceutical and biotechnology companies, government agencies, and patient advocacy groups has expanded the knowledge of the biology of cancer.
The global market for oncology drugs, including supportive care, reached $91 billion in 2013, as measured at ex-manufacturer prices and not reflecting off-invoice discounts and rebates, according to a May 2014 report from the IMS Institute for Healthcare Informatics. This is up from $71 billion in 2008, and represents a CAGR of 5.4%.
In pharmerging markets, oncology is expected to be the fourth highest spend therapy class by 2017, according to IMS. While the United States and top five European markets have declined in their share of the global market, they still dominate it with 65% of total sales.
Targeted therapies have dramatically increased their share of the oncology market, now accounting for 46% of total sales, up from 11% a decade ago.
 The oncology market is growing partially because of demographics, says Julie Papanek, principal of Canaan Partners, a venture capital firm.
The oncology market is growing partially because of demographics, says Julie Papanek, principal of Canaan Partners, a venture capital firm.
“Cancer is now at the top of the list of killers in the United States," she says. “There is also unmet medical need in this area. There have been reports published showing that more than 40% of total pharma revenue is going to come from oncology."
Ms. Papanek says while advances may seem like incremental wins, when they are added up across multiple lines of therapy they result in tremendous gains in survival.
“My dream is to find products that provide a durable complete response to cancer, the first time patients are treated," she says.
Ms. Papanek says in the future cancer treatment will look similar to the treatment of AIDS/HIV with cocktail therapies providing the best options for survival.
“The goal is to make cancer more of a chronic disease," she says.
In 2014, about 585,720 Americans are expected to die of cancer, almost 1,600 people per day. According to the American Cancer Society, the death rate from cancer has been steadily declining since the 1970s, declining more than 15% in the period from 2000-2011 alone. Five-year survival rates have increased 39% across all cancer types since 1975, with two out of three people diagnosed with cancer surviving at least five years.
The improvement in survival reflects both progress in diagnosing certain cancers at an earlier stage and improvements in treatment. Survival statistics vary greatly by cancer type and stage at diagnosis.
The National Institutes of Health (NIH) estimates that the overall costs of cancer in 2009 were $216.6 billion: $86.6 billion for direct medical costs (total of all health expenditures) and $130.0 billion for indirect mortality costs (cost of lost productivity due to premature death).
Cancer Research Trends
Incredible strides have been made in oncology and as a result, the percentage of the U.S. population living with cancer has more than tripled since the U.S. Congress passed the National Cancer Act in 1971, according the American Association of Cancer Research.
According to the American Cancer Society, 83% of survival gains are attributable to new treatments, including medicines. Between Aug. 1, 2013, and July 31, 2014, the FDA approved six new anticancer therapeutics and new uses for five previously approved anticancer therapeutics, two new cancer imaging agents, and one screening test. Five of these are compounds that target unique molecular and genetic characteristics.
Still, there continues to be huge unmet needs in general in oncology, says Alise Reicin, VP and therapeutic area head, clinical research, at Merck.
“Certainly with targeted therapies that have been developed over the last decade, we’ve seen some good advances," she says. “But patients who don’t have those specific mutations are still using standard chemotherapy, which has limited benefits and patients usually relapse."
Pharmaceutical company investments remain high and cancer therapies account for more than 30% of all preclinical and Phase I clinical developments, with 22 new molecular entities being launched and reaching patients in the last two years alone, according to the report from IMS Institute for Healthcare Informatics.
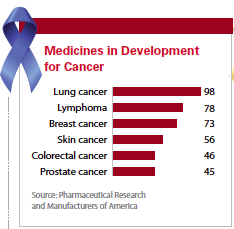
There are 771 medicines and vaccines either in clinical trials or awaiting review by the FDA, according to a September 2014 report from the Pharmaceutical Research and Manufacturers of America (PhRMA). Of the medicines and vaccines in clinical trials, there are 98 for lung cancer, 87 for leukemia, 78 for lymphoma, 73 for breast cancer, 56 for skin cancer, and 48 for ovarian cancer.
Currently, there are 3,137 active clinical trials for cancer in the United States. Of those, 1,824 are now seeking volunteers to participate or have not yet started recruiting patients; the other 1,313 are ongoing but are not currently recruiting new patients, according to PhRMA.
Advances in cancer research have led to an expansion in the clinical use of genomic information. Improvements in the ability to sequence and analyze large amounts of DNA have made it increasingly possible to identify the most appropriate therapy for a patient and to optimize the design and conduct of clinical trials.
Steve Turner, CEO of Protea Biosciences, says the biggest advance in oncology has been the ability to identify genomic aberrations and errors, such as SNPs or the activation of oncogenes.
“Going forward, future advances will be the ability to profile at the molecular level the cancer cell on a comprehensive basis to get past indirect means of identifying the proteins and the metabolics that are being produced by these aberrant tumor cells," he says. “We need big data solutions to identify hundreds of these cells very rapidly and get a molecular snapshot of the tumor cell that can then used to develop much more refined treatments and monitor for recurrence."
 A big driver in the cancer research area is that there is more precision in how different cancers are defined, says Cynthia Schwalm, president and CEO of Ipsen Biopharmaceuticals.
A big driver in the cancer research area is that there is more precision in how different cancers are defined, says Cynthia Schwalm, president and CEO of Ipsen Biopharmaceuticals.
“The drive toward more personalized medicine with genomics and being able to identify patients at the molecular level will lead to changes in how we think about cancer," she says. “We may find that 10 years from now that breast cancer is actually five or six different types of disease, and lung cancer may be five or six different diseases. The way patients will be treated will be much more specialized because we’ll match the person to a personalized therapy and survival rates should continue to increase."
A recent report from MarketsandMarkets predicts the global biomarkers market will grow at a CAGR of 18.5% from 2013 to 2018, to reach $40.8 billion by 2018. Advancement in discovery technologies such as genome sequencing is the prime factor contributing to the growth of this market.
For cancer biomarkers specifically, Transparency Market Research finds that the market for oncology biomarkers was valued at $13.16 billion in 2011 and is expected to be worth $29.78 billion in 2018, reflecting a compound annual growth rate of 14.60% from 2011 to 2018.
Ms. Reicin says many cancer therapies are coming to the market with a diagnostic, and this trend will continue. Merck is currently working on a biomarker for the expression of PDL1 protein in the tumor, which she says indicates that patients with a high expression appear more likely to respond to the company’s recently approved cancer therapy Keytruda. However she pointed out that there are patients who do not express PDL1 in the tumor who also respond, although the early data suggests that the response rate is lower.
Another key area in cancer is likely to be biosimilar therapies. In 2020, oncology biosimilars are estimated to reach between $6 billion and $12 billion in sales, or about 2% to 5% of the total global biologics market, according to the IMS Institute for Healthcare Informatics report. The top three compounds in oncology are biologics, and the top five represent a $20 billion annual opportunity for biosimilar competition.
Research Efforts
Researchers are now exploring new scientific avenues and new technologies for developing cancer therapies. In 2013, eight new orphan drugs were approved for the treatment of cancer, and the first drug launched with an FDA breakthrough designation was a cancer drug: Genentech’s Gazyva (obinutuzumab) to treat patients with previously untreated chronic lymphocytic leukemia (CLL).
One area gaining increased attention in cancer research is immuno-oncology.
“We are on the precipice of a new era in oncology," Ms. Reicin says. “There is a tremendous amount of work that has yet to be done but there is a lot of excitement in the field as well."
Merck recently received approval for Keytruda, which was approved in the United States in September 2014 for patients with unresectable or metastatic melanoma and disease progression following ipilimumab. It is the first anti-PD-1 (programmed death receptor-1) therapy approved in the United States and received FDA’s breakthrough therapy designation for advanced melanoma.
Keytruda is a humanized monoclonal antibody that works by increasing the ability of the body’s immune system to fight advanced melanoma. It blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2.
“We think the immune system does recognize cancer and there is probably something called immuno-editing going on where the immune system finds the cancer and begins the process of trying to kill the tumor," Ms. Reicin says. “But we’re learning now that tumors have developed ways to cloak themselves and deactivate the immune system. Tumors start to express a protein called PD-L1 or PD-L2. These proteins interact with the proteins on T-cells and they are able to deactivate the T-cell so that it no longer recognizes or kills the tumor."
Ms. Reicin says Keytruda works to inhibit that interaction between PD-L1 and PD-L2 on the tumor with T-cells so those T-cells are now activated and can recognize and kill the tumor cells.
Another area getting attention from researchers is in rare cancers and cancers with unmet needs.
“A growth area will be on further defining some of the rare types of cancers," Ms. Schwalm says. “Better molecular testing and much more defined research along with FDA acceptance of what is needed to study patients with rare cancers will lead to high growth in rare forms of cancer."
Ms. Schwalm says FDA acceptance of many of these rare cancers as orphan diseases makes it easier for small companies to do clinical trials in these cancers.
Ipsen is in the final stages of accelerated review for Somatuline Deport, a somatostatin analogue indicated for the long-term treatment of patients with neuroendocrine cancer in the pancreas and certain parts of the colon, a rare cancer.
The product has a unique formulation and a new delivery system, which is a syringe technology that allows for consistent dosing.
“There are benefits for healthcare providers who give the medicine," Ms. Schwalm says. “It has a device that retracts the needle so there is better safety for unintended needle sticks and it is latex free."
But developing therapies for cancer can be challenging. In fact, the complexity of cancer is reflected in the drug development process. Cancer medicines can take an average of 1.5 years longer to develop than medicines for other diseases, according to PhRMA.
In 2014, PhRMA conducted a review of three difficult-to-treat cancers — melanoma, lung cancer, and brain cancer. Since 1998, there have been 96 unsuccessful attempts to develop drugs to treat melanoma, 167 for lung cancer, and 75 for brain cancer.
In the same period we also saw medicines that beat the development odds: seven new drugs to treat melanoma, 10 for lung cancer, and three for brain cancer were approved by the FDA.
Ms. Papanek says researchers are still learning how tumors work.
“Investors and scientists are making informed guesses about what therapies will help patients but sometimes preclinical models are not necessarily representative of what we are seeing in the clinic," she says. “We want to see models and experiments that allow you to predict what will work and what won’t. But some models are limited in their predictive value to patients. It is truly only when you get into the clinic that you learn what therapies work best."
Mr. Turner says the limiting factor in oncology research is qualifying better and more specific biological targets in the tumor cells for therapeutic intervention.
“Nature is a powerful force and we know that genomic changes can be used to identify the cancer cell but there is not a lot of information on the tumor’s DNA and what eventually gets through RNA and through proteins," he says. “We need information on a very comprehensive basis to improve our understanding of what is going on with the cancer biology."
Ms. Schwalm says another challenge in developing cancer therapies, especially for rare cancers, is finding patients.
“There aren’t the large patient populations that you find in some cancers," she says. “Finding the right clinical research network that can get these patients into the system and ensuring that they meet the merits of our protocol is still our biggest challenge. Secondly, these trials are very expensive."
For Ipsen, the challenge of finding patients was made easier because U.S. centers of excellence for neuroendocrine tumors helped to funnel patients to their research.
The Market
By 2018, the oncology products market will account for more than 12% of total drug sales, dwarfing all other drug categories. The total annual U.S. cost of cancer care is projected to reach $175 billion by 2020, an increase of 40% from 2010. According to The State of Cancer Care in America: 2014, a report released by the American Society of Clinical Oncology (ASCO), the U.S. market accounts for 41% of total oncology drug sales.
Due largely to the aging of the population as well as lifestyle changes, the number of new cancer cases in the United States is projected to increase by as much as 42% by 2025. Despite a near doubling of demand for cancer care services, ASCO predicts there will be a shortage of 1,487 oncology physicians in just 10 years. The number of oncologists is projected to grow by only 28%.
Physicians
There are currently about 13,000 oncologists practicing in the United States, the majority in private community practices. As a group, oncologists are highly focused on patient care with more than 80% identifying “direct patient care" as their primary professional activity. According to a November 2014 report in the New York Times, since 2008, 544 private oncology practices were purchased by or entered contractual relationships with hospitals, another 313 closed and 395 reported they were in tough financial straits.
IMS Health estimates that 80% of U.S. cancer care is carried out in community-based oncology clinics. ASCO states that more than 40% of oncologists are now in practices with seven or more physicians, up from 29% in 2012, as smaller practices are aggregated and/or acquired by hospital systems.
According to a recent report from Publicis Touchpoint, one of the leading challenges for privately owned oncology clinics is the U.S. federal government’s 340B Drug Pricing Plan, which allows specified hospitals (and their affiliated oncology clinics) to purchase drugs at discounted prices. This preferential-pricing policy typically pertains to hospitals with a high population of Medicare, Medicaid, and uninsured patients because these institutions are seen as safety-net providers in healthcare.
The number of hospitals participating in 340B pricing has tripled in number since 2005, and the program now includes roughly one-third of the nation’s hospitals. This is driving the location of patient care from physician offices to hospital outpatient facilities.
Payers
According to IMS, the high number of new targeted therapies launched and available for cancer patients has escalated payer scrutiny, as payers want to be sure they are not paying for a higher priced drug that gives the same outcome as a lower priced one would. The average cost per month of branded oncology drug treatment in the U.S. is now about $10,000, up from an average of $5,000 a decade ago. Due to the high level of variability of patient response, the frequent changes to protocol needed for patient care, and underlying issues of equity and patient care, judging value of these treatments for individual patients is filled with challenges.
Experts predict that payers will increasingly choose which drugs make it on formularies based mostly on drug price. To overcome this hurdle, oncology treatment manufacturers are implementing strategies that differentiate their medications and demonstrate their value over the current standard of care. With payers and managed care organizations striving more than ever to limit costs, the industry needs to present drug value as a combination of efficacy, safety, and economics.
For example, biotech GlycoMimetics developed a program for its new treatment for vaso-occlusive crisis (VOC) of sickle cell disease that took into account the medication’s economic value as well as its safety and efficacy. For example, in studies, the drug showed a faster VOC resolution and a shorter length of hospital stay in trials against a placebo. GlycoMimetics used this information to demonstrate that its treatment is not only efficacious, but can also decrease costs to payers.
In terms of being a player in the payer marketplace, the industry should be providing support for oncology reimbursement issues to physicians and patients.
“A cancer diagnosis is a huge burden on patients and their caregivers, and they, and their doctors, shouldn’t have to also worry about navigating a complex reimbursement system," says Mark Reisenauer, VP, oncology sales and marketing, Astellas Pharma US. “As such, Astellas Oncology has designed a comprehensive support solution for XTANDI, called Quick Start, to help providers verify benefits, obtain and track prior authorization, determine patient eligibility for access programs, appeal assistance, triage prescriptions to specialty pharmacies, and facilitate immediate access to treatments."
According to Mr. Reisenauer, as cancer treatment evolves, so too does the landscape of providers and it’s important to consider various perspectives and challenges of all prescribing physicians in the cancer space. At Astellas, a special program has been designed to meet the unique needs of both oncology and urology prescribers.
Decision Resources Group has predicted that XTANDI will be the sales-leading agent across the world’s major markets for prostate cancer, garnering $3.4 billion in 2023.
Sales
The life of the oncology sales rep isn’t getting any easier, either. When it comes to accepting visits from sales reps, oncologists are the most restrictive, limiting access 65% of the time. Many life-sciences companies are training sales reps to possess a science-focused education, consultative sales ability, and superior access skills. In addition, many companies are also beginning to deploy blended teams consisting of a traditional field sales team, along with some combination of nontraditional communication teams.
 The industry should be constantly evaluating the most effective ways of delivering key product information to its customers, focusing on how they prefer to receive information and customizing to meet their needs, Mr. Reisenauer says.
The industry should be constantly evaluating the most effective ways of delivering key product information to its customers, focusing on how they prefer to receive information and customizing to meet their needs, Mr. Reisenauer says.
“At Astellas Oncology, we’re working closely with providers, as well as other key stakeholders like payers and regulators, to understand what data they want and when, and how we can better deliver that data to help us all serve patients better," he says. “Regarding delivery of information, we know that, like most consumers, doctors are spending a significant amount of time per day online. For example, they’re using smartphones nearly 20 times per day to access information for their patients and their practice and then also using tablets and desktops throughout the day. Many of these trips online are for professional purposes so we need to ensure that our marketing efforts are providing information to healthcare providers on demand through the digital channels they’re using most."
According to Publicis Touchpoint in its 2020 Vision, Focus on Oncologists report, many life-sciences companies are doing just that, beginning with improvements to the salesforce. Companies need high-level, exceedingly well-trained sales representatives who can analyze and interpret complex cancer-related information and then educate physicians on the relevance of the latest clinical research.
Sales reps will need to use multichannels, particularly for oncologists, who say preferable alternatives include email, direct mail, and peer-to-peer programs. Alternatives are critical as companies continue to make high-profile targeted cancer drugs, particularly with the new pathways FDA has recently adopted and the patient-centered focus the agency will continue to adopt over the next few years.
To maintain long-term access and relevance to oncologists, companies must develop new, innovative customer engagement models, designed not from the perspective of the pharmaceutical company, but from the perspective of the customer.
Oncology reps are expected, on average, to make five calls a day, but ZS finds that in fact they’re only averaging around three. On average, reps get to visit oncologists only seven or eight times in a year, with even the highest-performing reps getting face time only once a month.
In its 2013 AccessMonitor report, ZS Associates found that oncologists were more likely to open their doors for reps with new drugs to offer — 33% more frequently — but that only lasted for about six months after launch.
In addition, oncology reps with three or more products are able to visit physicians an average of 10 times per year, but those carrying just one or two products only see physicians an average of seven times per year. On average, a rep carrying three or more products is twice as likely to have access to even the most restrictive oncologists — 26% vs. 13% for reps carrying one or two products.
The global oncology market will show growth in the coming years at a 5% rate from $91 billion in 2013, according to IMS.
The oncology market is a very complex, difficult market that will struggle with challenging commercial operations due to limited physician access, payer restrictions, and the focus on economic and clinical value. (PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Thought leader quotes:
 I’ve never seen so much activity in cancer. There are hundreds of -products in development with new targets, new drug platforms, and new modalities.
I’ve never seen so much activity in cancer. There are hundreds of -products in development with new targets, new drug platforms, and new modalities.
Julie Papanek / Canaan Partners
 We are working closely with -providers, as well as other key stakeholders like payers and -regulators, to -understand what data they want and when, and how we can better deliver these data to help us all serve patients better.
We are working closely with -providers, as well as other key stakeholders like payers and -regulators, to -understand what data they want and when, and how we can better deliver these data to help us all serve patients better.
Mark Reisenauer / Astellas Pharma US
 The drive toward more -personalized medicine with -genomics and being able to identify patients at the -molecular level will lead to changes in how we think about cancer.
The drive toward more -personalized medicine with -genomics and being able to identify patients at the -molecular level will lead to changes in how we think about cancer.
Cynthia Schwalm / Ipsen Biopharmaceuticals
~~~~~~~~~~~~~~~~~~~~~~~~~
Fast Fact
The global oncology market reached $91 billion in 2013; ?most sales continue to be in ?the U.S. and Europe.
Source: IMS Institute for Healthcare Informatics
~~~~~~~~~~~~~~~~~~~~~~~~~
Top 5 Advances in ?Modern Oncology
1965: Chemotherapy Cures Hodgkin -Lymphoma
2006: Vaccine Approved To Prevent -Cervical Cancer
2001: Targeted Drug Transforms Treatment For Rare Leukemia
1977: New Treatment Cures Men With -Testicular Cancer
1991: Powerful Anti-Nausea Drugs -Alleviate Major Side Effect Of Cancer -Treatment
Source: American Society for Clinical Oncology
~~~~~~~~~~~~~~~~~~~~~~~~~
250 Years of Advances Against Cancer
1761 – John Hill suggests that there may be a -relationship between the use of tobacco snuff and the development of nasal cancer.
1775 – Percivall Pott identifies a relationship between exposure to chimney soot and the incidence of squamous cell carcinoma of the scrotum among chimney sweeps.
1863 – Rudolph Virchow identifies white blood cells (leukocytes) in cancerous tissue. He coins the term “leukemia."
1882 – William Halsted performs the first radical -mastectomy to treat breast cancer.
1895 – Wilhelm Roentgen -?discovers x-rays.
1898 – Marie and Pierre Curie discover the radioactive elements radium and polonium.
1901 – Francis Henry Williams describes the use of x-rays to cure a patient with squamous cell skin cancer of the lip.
1904 – Hugh Young and William Halsted perform the first radical perineal prostatectomy to treat prostate cancer.
1907 – The American Association for Cancer Research (AACR) is established.
1909 – Paul Ehrlich proposes that the immune -system usually suppresses tumor formation, a concept that becomes known as the “immune surveillance" hypothesis.
1911 – Peyton Rous discovers a virus that causes -cancer in chickens (Rous sarcoma), -establishing that some cancers are caused by infectious agents.
1913 – The American Society for the Control of Cancer, which later becomes known as the American Cancer Society, is established.
1928 – George Papanicolaou discovers that cervical cancer can be detected by examining cells taken from the vagina, which leads to the development of the Pap smear.
1930 – Congress establishes the National Institute of Health in the Ransdell Act.
1932 – David H. Patey develops the modified radical mastectomy for breast cancer.
1937 – Legislation signed by President Franklin D. -Roosevelt establishes the National Cancer -Institute (NCI).
1944 – Oswald Avery, Colin MacLeod, and Maclyn -McCarty identify DNA as the basic genetic -material of cells.
1947 – Sidney Farber shows that aminopterin, a -derivative of folic acid, induces temporary -remissions in children with acute leukemia.
1949 – Nitrogen mustard (mechlorethamine) is -approved by the FDA for treating cancer.
1950 – Ernst Wynder, Evarts Graham, and Richard Doll identify cigarette smoking as an important -factor in the development of lung cancer.
1953 –James Watson and Francis Crick determine that the molecular structure of DNA is a double helix.
1953 – Carl Nordling proposes that cancer cells contain mutations in multiple genes.
1953 – Roy Hertz and Min Chiu Li achieve the first complete cure of a human solid tumor by -chemotherapy with methotrexate.
1958 – NCI researchers Emil Frei, Emil Freireich, and James Holland and their colleagues at NCI -pioneer the use of combination chemotherapy.
1960 – Peter Nowell and David Hungerford describe an unusually small chromosome in the cancer cells of patients with CML, the Philadelphia (Ph) chromosome.
1964 – The U.S. Surgeon General issues a report stating that cigarette smoking is an important health hazard in the U.S.
1964 – For the first time, a virus — the Epstein-Barr virus — is linked to human cancer.
1967 –The guaiac fecal occult blood test (FOBT) is introduced as a screening test for colorectal cancer.
1968 –Baruch Blumberg and colleagues report initial isolation of the virus associated with hepatitis B, a major cause of liver cancer.
1969 – Robert Huebner and George Todaro propose the oncogene hypothesis of cancer.
1971 –Judah Folkman reports that they have -isolated a factor from tumors that stimulates the growth of new blood vessels toward tumors, known as tumor -angiogenesis.
1971 –On Dec. 23, President Nixon signs the National Cancer Act.
1972 – Godfrey Hounsfield describes his invention of computerized tomographic (CT) scanning.
1974 – The FDA approves doxorubicin, an -anthracycline antibiotic from Streptomyces bacteria, for the treatment of cancer.
1976 – Dominique Stehelin, Harold Varmus, J. Michael Bishop, and Peter Vogt discover that the DNA of normal chicken cells contains a gene that is related to the oncogene of avian sarcoma virus.
1976 – Interleukin-2 is discovered in mice, which plays a role in the development of disease-fighting blood cells called T lymphocytes.
1978 – Tamoxifen, an antiestrogen drug, is approved by the FDA for the treatment of breast cancer.
1980 – NCI scientist Robert Gallo and his colleagues isolate human T-cell lymphotrophic virus 1.
1982 – The hepatitis B virus vaccine is approved by the FDA for the prevention of liver cancer.
1984 – The human TP53 (p53) gene ?is cloned.
2011 – The FDA approves the drug vemurafinib to treat melanomas with mutation in the BRAF gene.
2013 – The FDA approves ado-trastuzumab emtansine to treat HER2-positive breast cancer.
2013 – The drug dabrafenib is approved by the FDA to treat inoperable or metastatic melanoma that carries the BRAF gene mutation.
2013 – The drug trametinib is approved by the FDA to treat inoperable or metastatic melanoma that has a mutation in BRAF protein.
~~~~~~~~~~~~~~~~~~~~~~~~~
Cancer Awareness
January
• Cervical Cancer Awareness Month
February
• National Cancer Prevention Month
• Gallbladder and Bile Duct Cancer Awareness Month
March
• Colorectal Cancer Awareness Month
• Multiple Myeloma Awareness Month
• National Kidney Cancer Awareness Month
April
• Testicular Cancer Awareness Month
• Esophageal Cancer Awareness Month
• Head and Neck Cancer Awareness Month
May
• Brain Cancer Awareness Month
• Melanoma and Skin Cancer Awareness Month
June
• National Cancer Survivors Day
July
• Bladder Cancer Awareness Month
• Sarcoma Awareness Week
September
• Childhood Cancer Awareness Month
• Gynecologic Cancer Awareness Month
• Hodgkin’s Lymphoma Awareness Month
• Leukemia Awareness Month
• Lymphoma Awareness Month
• Ovarian Cancer Awareness Month
• Thyroid Cancer Awareness Month
• Prostate Cancer Awareness Month
October
• Breast Cancer Awareness Month
• Liver Cancer Awareness Month
November
• Lung Cancer Awareness Month
• Carcinoid Cancer Awareness Month
• National Family Caregivers Month
• Pancreatic Cancer Awareness Month
• Stomach Cancer Awareness Month
~~~~~~~~~~~~~~~~~~~~~~~~~
Challenge of Today’s Oncologist
In the rapidly changing oncology marketplace, oncologists are facing several challenges:
- Impending oncologist shortage
- Precarious delivery, reimbursement, and pricing environment
- Increased use of clinical pathways
- Complexity of care
- Increase in availability and use of oral agents
- Shift toward hospital-owned practices
- Restricted access to industry resources
Source: Publicis Touchpoint
~~~~~~~~~~~~~~~~~~~~~~~~~
The New Oncology Sales Rep
In order to be successful, the following qualities and characteristics are necessary to excel in the challenging field of oncology.
- Oncologists respond well to sales -representatives with:
- Science-focused education/training
- Science and/or medical training, such as advanced degrees in health sciences
- Recognized credentials, including former nurses and pharmacists
- Significant experience in pharmaceutical sales
To better enable reps to service -oncologists, companies need to:
Develop resources for use by the entire patient care staff, including NPs/PAs and infusion nurses.
Guide field-based teams to -develop meaningful relationships with their -clinics’ billing and reimbursement personnel in order to help them make -informed reimbursement decisions.
Arm the customer-facing team with -decision-making tools and other -resources that can help oncologists navigate complex regimens.
Develop specific educational tools for the office staff on how to manage -patients during therapy and how to address treatment complications.
Take greater care to learn which factors influence patients’ acceptance and adherence to therapy.
Provide more tools, such as contact -centers, social networking sites, and other resources to help support caregivers and patients.
Source: Publicis Touchpoint
~~~~~~~~~~~~~~~~~~~~~~~~~


















