 According to the National Institutes of Health (NIH), more than 80% of clinical trials in the United States fail to meet their patient recruitment timelines. These delays increase costs, deplete resources, and prolong the time to market introduction.
According to the National Institutes of Health (NIH), more than 80% of clinical trials in the United States fail to meet their patient recruitment timelines. These delays increase costs, deplete resources, and prolong the time to market introduction.
Patient recruitment is challenging as it involves multiple stakeholders, including patients, healthcare providers (HCPs), office staff, sponsors, and clinical trial research teams. With clinical trial recruitment, timelines are always accelerated and tight, communications are fragmented, and recruitment rates tend to be disappointingly low.
A few common challenges in recruiting patients for clinical trials include:
Lack of physician awareness of available clinical trials.
Unrealistically restrictive inclusion and exclusion criteria. Research from the National Cancer Institute shows that 40% of oncology trials fail to meet minimum patient enrollment, and more than 60% of Phase III trials fail to meet minimum patient enrollment.
Impractical and inconvenient protocol requirements.
Potential Solutions
How patients learn about clinical trials is dramatically changing. Historically, patients learned about clinical trials through the media, mail,  and their HCPs. In the early 2000s a significant shift began and more patients began learning about clinical trials through e-mail and the Internet, as well as traditional outlets.
and their HCPs. In the early 2000s a significant shift began and more patients began learning about clinical trials through e-mail and the Internet, as well as traditional outlets.
At the same time, stakeholders in clinical research shifted their efforts to place patients at the center of planning and execution. Specific changes include clinical research professionals partnering with patients to play a more active role in informing and setting research agendas, patients providing feedback on protocol design feasibility, patients participating and engaging their peers and communities as volunteers in clinical trials, and disclosing and disseminating clinical trials results to study participants.
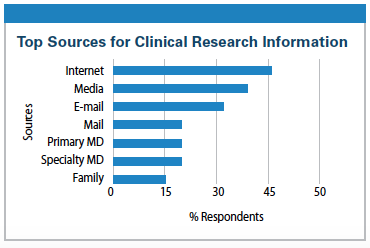
In 2013 the Center for Information and Study on Clinical Research Participation (CISCRP) conducted a global assessment of the changing attitudes of public and patient perceptions about clinical research. In the 2013 CISCRP survey, the top sources for clinical research information are:
Clearly there are many channels that patients are using to obtain clinical research information, and this requires that clinical trial sponsors use multiple channels to communicate about on-going trials.
Best Practices in Patient Recruitment
 Patient recruitment strategies have significantly changed with increased patient engagement in clinical research trials. Additionally, the tools patients are using to obtain information about trials have changed dramatically. There are recommended five best practices for successful patient recruitment:
Patient recruitment strategies have significantly changed with increased patient engagement in clinical research trials. Additionally, the tools patients are using to obtain information about trials have changed dramatically. There are recommended five best practices for successful patient recruitment:
No. 1 Know the target audience.
Key questions to ask include:
What is the target patient population for this study?
Where is the patient population likely to get information/education? Are the
patients likely to engage online and through social media? If so, what channels are patients engaging with?
What types of HCPs treat the condition? To what extent are allied professionals (i.e., pharmacists, social workers, nurses)
involved in treating this condition?
What are the best ways to work with hospitals, health systems, and HCPs to identify eligible patients?
No. 2 Develop a promotional plan.
The plan should be based on the preferences of the target audiences and include outreach to HCPs and advocacy groups, and various media channels such as print, TV, radio, and social media such as Twitter, LinkedIn, and Facebook.
No. 3 Develop mechanisms to screen potential patients.
Once patients are identified have a screening process in place via dedicated call center operators, study websites, and interactive voice response (IVR) systems.
As part of the screening process, sponsors and CROs need to:
Evaluate the study protocol inclusion and exclusion criteria and include these criteria as a part of the pre-screening tool.
Determine if skilled nurses or other HCPs are needed for pre-screening more complex protocols that require medical knowledge or extensive screening.
Consider the need for multilingual communications. If this is needed, translate the pre-screening questionnaire and partner with a pharmaceutical call center that offers multi-lingual operators.
No. 4 Accelerate the process for referring patients to study sites.
Once the initial screening is conducted, develop a process to quickly and seamlessly refer patients to study sites for additional evaluation, which can be done through several options, including:
Warm transfer — a call center operator can initiate a three-way call to connect the potential patient with the closest research site.
Appointment scheduling — include
available site appointments in the call center database so call center operators can immediately schedule an appointment for a referred patient.
Provide appointment confirmation materials, including date and time for appointment, directions to the study site, pre-visit instructions, and study information.
No. 5 Implement a system to track patient referrals and follow up.
Once patients are referred to a study site it is critical that they are quickly contacted by the site for additional evaluation and possible  enrollment in the study. Processes and systems are needed to ensure rapid follow up and appointment scheduling. Some options to consider include:
enrollment in the study. Processes and systems are needed to ensure rapid follow up and appointment scheduling. Some options to consider include:
Site solutions — online solutions to track the progression of patients from referral to enrollment. An online portal provides study sites with a view of the patients referred to their site and provides details on overall enrollment success.
Sponsor solutions — secure, HIPAA-compliant portals provide project teams the ability to actively monitor enrollment across all study sites. Underperforming sites can be quickly identified and resources reallocated as needed. Sponsors can also view the number of patients who have failed screening and can adjust inclusion and exclusion criteria if needed.
Patient recruitment initiatives that include all five of these areas accelerate patient recruitment timelines and enhance the likelihood that the patient enrollment target will be met.
Best Practices for Retaining Patients
Once patients are enrolled in a clinical study, the emphasis shifts to retaining patients in the trial and ensuring they are compliant with all aspects and timelines of the protocol. Enrolled study participants are more likely to adhere to protocol requirements when study-related tasks are top of mind. There are many solutions available to ensure compliance with study-related tasks, including:
Appointment scheduling and reminders via email, mail, and telephone.
Reminders to complete patient diaries and/or interviews.
Online patient diaries.
Reporting mechanisms via telephone and secure Websites, as well as centralized adverse event reporting.
Centralized communications that streamline the information flow and provide after-hours support for questions, follow up with patients, appointment rescheduling, and sharing information directly with site coordinators.
Providing patients with immediate access to operators for questions and easy-to-use tools to complete study-related tasks increase patient 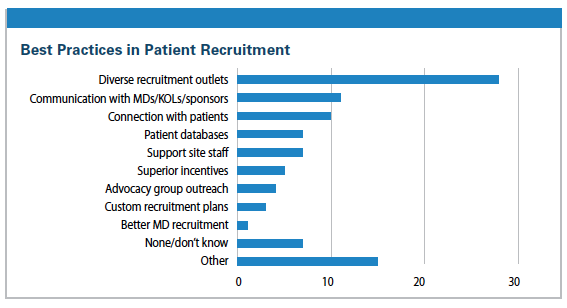 retention in clinical trials.
retention in clinical trials.
In closing, recruitment and retention are critical components to completing clinical trials in a timely manner. In 2014, Industry Standard Research conducted research with industry leaders in patient recruitment and patient network services; 28% indicate the most prevalent best practice is to use a diverse range of recruitment outlets and approaches. Clearly, there is no one preferred approach to recruitment, rather a diverse offering of initiatives is needed for success.
A pharmaceutical call center can support life-sciences companies in developing and implementing customized patient recruitment and retention initiatives specific to the target patient population(s), as well as provide a variety of resources and tools to accelerate enrollment and enhance patient retention. (PV)
Tunstall delivers customized 24/7 healthcare communication services to connect individuals, caregivers, and healthcare providers; drive awareness and knowledge; support patient adherence; and empower patients. Our products and services enhance patient care, improve outcomes, and increase operational efficiencies for hospitals, medical centers, health systems, payers, and life-science companies.
For more information, visit americas.tunstall.com.











