AstraZeneca made a bold move last year and took back control of its servers and networks that pertained to core drug 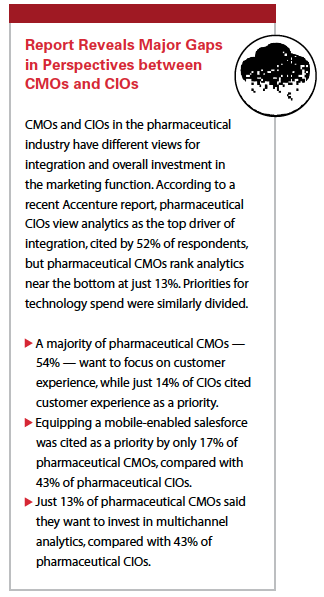 development from outsourcing vendors, which led to a 90% reduction in the number of critical outages, and decreased incident response down to a few hours instead of several days. For other segments of its business, AstraZeneca is using several software-as-service (SaaS) applications. For example, the company recently moved its entire internal communications to the cloud, using Box’s cloud content sharing and collaboration platform. The platform allows secure collaboration companywide of its 51,000 employees across more than 100 countries and with external parties, as well as mobile access to all information. AZ is the largest account in the pharma industry using this particular file-sharing tool.
development from outsourcing vendors, which led to a 90% reduction in the number of critical outages, and decreased incident response down to a few hours instead of several days. For other segments of its business, AstraZeneca is using several software-as-service (SaaS) applications. For example, the company recently moved its entire internal communications to the cloud, using Box’s cloud content sharing and collaboration platform. The platform allows secure collaboration companywide of its 51,000 employees across more than 100 countries and with external parties, as well as mobile access to all information. AZ is the largest account in the pharma industry using this particular file-sharing tool.
AstraZeneca CIO David Smoley expects this agile business model to allow the company to do what it does best: create treatments faster and more cost effectively.
“At AstraZeneca this collaboration is critical as we work to accelerate the process from development to commercialization, to get new medicines to patients faster," Mr. Smoley says. “IT has to be an enabler for the business, giving our people the right tools and capabilities so they can focus on what they are here to do — bring lifesaving medicines to patients."
AstraZeneca has implemented a number of new tools over the past year to help people collaborate in whichever way best suits their needs, Mr. Smoley says.
“I spend time with leaders and teams across the business to understand their needs from the IT organization," he says. “We work hard to make sure we have competent IT talent partnering with each team, focused on supporting their work and on achieving positive business outcomes."
 On a smaller scale, but similar move to use technology to create efficiencies, Sanofi US has streamlined its content management system using DITA Exchange for structured authoring and automated publishing for clinical and CMC documentation processes. The DITA (Darwin Information Typing Architecture) model provides an open XML content standard defining a common structure that promotes the consistent creation, sharing, and re-use of information. By using the structured management content system, Sanofi has gained operational efficiencies throughout the development process, including major cost and time savings, as well as maintaining the integrity and quality of data consistently across functions.
On a smaller scale, but similar move to use technology to create efficiencies, Sanofi US has streamlined its content management system using DITA Exchange for structured authoring and automated publishing for clinical and CMC documentation processes. The DITA (Darwin Information Typing Architecture) model provides an open XML content standard defining a common structure that promotes the consistent creation, sharing, and re-use of information. By using the structured management content system, Sanofi has gained operational efficiencies throughout the development process, including major cost and time savings, as well as maintaining the integrity and quality of data consistently across functions.
While Sanofi is focusing on handling clinical trial data through this advanced technology, it can be  applied to all business forms.
applied to all business forms.
“But the clinical development process is not the limit to using SCM in the industry — it can certainly be applied to early development and to research itself," says Joan Affleck, associate VP/operational leader, Sanofi.
Ms. Affleck credits advances in technology with spurring the renewed interest by the industry for SCM. The major players in the industry have dabbled in SCM in the past, but perhaps abandoned the idea due to lack of infrastructure, time, and technology evolution.
 “In the past several years, major players have come back to this idea and Sanofi is in the forefront of work being done in the clinical space," she says. “Based on interactions with other executives in the industry and reps from regulatory agencies, I would say within the next five years we will see a major increase of use in this kind of content management."
“In the past several years, major players have come back to this idea and Sanofi is in the forefront of work being done in the clinical space," she says. “Based on interactions with other executives in the industry and reps from regulatory agencies, I would say within the next five years we will see a major increase of use in this kind of content management."
SCM is just one small segment of how technology and companywide applications can be mastered with the appropriate approach.
“Key documentation is a major deliverable for the pharma industry and there are increasing requirements related to transparency of the documents around clinical protocols and study results and we want to ensure our data and messages are communicated in a clear and consistent way to all stakeholders," Ms. Affleck says. “If we were to apply these principles and standards in healthcare reporting so that data information about patient status, healthcare needs, and challenges can be harmonized, we have the ability to share information across industry, research, academia, and patient-care delivery in ways that protect patient privacy but also accelerate finding the right treatment for each patient, enhance compliance, and help provide better healthcare for everyone."
These are just two examples of the types of transformations that are being driven by the availability of faster and more efficient models based on revolutionary technology.
“The healthcare industry is facing unprecedented technology-driven change," says Olivier Leclerc, director, McKinsey.
 According to Mr. Leclerc, there are three fundamental technology discontinuities driving the IT transformation: mobile, cloud, and advanced analytics.
According to Mr. Leclerc, there are three fundamental technology discontinuities driving the IT transformation: mobile, cloud, and advanced analytics.
“The market is changing and so are the needs, and technology is enabling extremely rich and rapidly improving insights into consumers, markets, and products," he says.
These challenges also present opportunities, mostly within four categories, Mr. Leclerc says. First, is advanced analytics and real-world evidence. Tapping into data analytics around clinical trials allows drug manufacturers to lower the risk of drugs and to demonstrate the efficacy of their product.
“Analytics also let companies create micro segments for marketing," Mr. Leclerc says.
Another opportunity lies in developing technology around products, such as patient compliance solutions for pharma products or tech enabling medical devices. Life-sciences companies should also look for digital opportunities to enhance consumer relationships. Since both physicians and patients are increasing their use of digital sources, this is the perfect opportunity for pharma to engage with both sets of customers. For example, Mr. Leclerc says, more patients are going and engaging online to learn about their disease, for instance on patient exchange platforms.
Challenges with Technology
Pharma is under extreme pressure to change and with that comes many challenges, says Sastry Chilukuri, partner, McKinsey.
“There is a market shift and some trends that are accelerating will put more pressure on pharma to change," he says. “If we think about the pharma industry today versus 25 years ago, there is more data on products outside the four walls of a pharma company than within; it used to be that very few entities outside a pharma company had more data than it did. Pharma companies had full control of their data and today this is reversed. There has been an explosion of data at all levels — consumer, payer, patient — everyone has access to data in some way, shape, or form."
This creates a lot more pressure to engage at the digital level, Mr. Chilukuri says.
Adapting to the pace of change and understanding that success in this dynamic environment requires an open mind and agility, and these are some of the biggest challenges facing the C-suite, Mr. Smoley of AZ says.
“When it comes to IT, we cannot operate in a world of annual plans because they are obsolete in months, if not weeks," he says. “It’s also a big challenge to sort through all the information and identify what is relevant and important, versus what is noise. Increasingly, IT underpins every activity within a company. No matter what sector you are in, it is likely that competitors who are at the leading edge with IT will have an advantage over their peers. In an innovative company, in a heavily regulated industry, this takes some work to get the right balance."
One of the major challenges Ms. Affleck has observed is the slow turning of the legacy mindset. In document management, in particular, this means moving away from the idea of being document-centric and thinking of a document as a standalone, large compendium of content and embracing the idea of breaking up that document into pieces of content that can be reused across the life cycle of a document road map. With this thinking comes the No. 1 concern of whether doing something new will take more time.
“Timelines are very important in clinical development and for pharma at large, so busy people don’t want to start using a new tool if they are going to have a steep learning curve or if it will make their job more difficult," Ms. Affleck says. “That’s a fear people have."
Even though there are challenges, organizations are going to be forced to move forward to survive the competitive marketplace.
“Rather than just focusing on the challenges, it may be useful to look at what organizations have to do to enable this transition and overcome the challenges that are inherent," says Bhaskar Sambasivan, VP, head of life sciences, NA & UK, Cognizant.
He recommends four steps: a clear understanding and belief across the organization that technology can fundamentally transform businesses and generate significant value, which may have to start with a communication from the CEO; CIOs proactively engaging with business leaders to understand their priorities and demonstrate agility in their organizations to deliver on these priorities; CIOs to focus on building teams and capabilities in-house and through strategic partners to deliver on the dual set of priorities for their organizations, i.e. driving efficiencies as well as generating business value; and finding the right set of strategic partners that can not only help them deliver on this dual mandate, i.e. run better, but also enable differentiated capabilities to gain a competitive business advantage.
IT — In-house or Outsource?
Our thought leaders say it largely depends on each organization to identify where efficiencies can be made, who should be in charge of the transition, and will it be tackled by an in-house organization or outsourced. Not all IT work can or should be in-sourced. Many commoditized and transformational services are best executed with the help of the right strategic partners, given the scale, maturity, and agility that many large service providers can offer. This ensures the right balance between what should be in-house versus partnered. This balance will help organizations keep abreast with the latest technology shifts and ensure that they are always leveraging innovative capabilities that will give them a competitive advantage.
While creating in-sourced capabilities, companies have to carefully consider the kind of capabilities that will give them a lasting advantage. Companies will not want to invest in building capabilities that will be obsolete in three to five years.
“The lifespan of many technologies has been steadily shrinking over time while the advancement of digital technologies — social, mobile, analytics, cloud, automation — have been rapid," Mr. Sambasivan says. “This means incubating necessary and sufficient capabilities that can help companies understand the implication of technologies on their businesses and how to leverage these to build a competitive advantage."
According to Mr. Smoley, AstraZeneca has chosen to in-source because the company believes it gives them a competitive advantage.
“Both the pharma industry and the technology industry are moving so quickly that we believe having our own people on the front line is a good strategic move, to keep ahead of the game and stay innovative," he says. “We can also take advantage of an improved cost of delivery, enabling us to plow more resource into our pipeline and delivering life-changing medicines."
AstraZeneca started by identifying the service lines and capabilities where opportunity for positive business impact was greatest.
“For example, we have launched an IT technology center in Chennai specifically to strengthen our internal resources, taking advantage of the huge supply of IT talent in the city," he says. “We hired 350 AZ IT employees there in 2014 and this will increase to 1,000 this year."
According to Patrick Sim, chief information officer, West Pharmaceutical Services, a company needs to make the decision on outsourcing based upon what value IT brings to the company. It becomes very difficult to rebuild capabilities once everything has been outsourced. Also, the talent market for software and hardware skills remains very competitive, so a company will likely have to pay top salaries, which then offsets the cost savings that may have been realized during the outsourcing period. All of this will take significant time and effort to achieve.
“I would suggest that pharma first focus on bringing in-house capabilities that are critically important to differentiate its business from the competition," Mr. Sims says.
 Mr. Smoley says AstraZeneca has made its decisions based on that theory.
Mr. Smoley says AstraZeneca has made its decisions based on that theory.
“Our in-sourcing strategy is very much based on the benefit to the business," he says. “For example, we started by in-sourcing our application support and maintenance. To best support the company, we needed to truly understand what the organization needs to do and how our systems can support and enable success. We can’t do that if we don’t understand our IT landscape. What systems and applications do we have? Which applications give us a more competitive advantage? Which are barriers to success? Can we enhance and simplify some, and should we divest and converge on others? There are so many questions to answer, and if we aren’t able to answer them, then we are too far removed to deal with challenges effectively. These are the areas where we want to in-source first."
There are times when the need for speed warrants an outsourcing platform, such as automated platform applications that can enhance collaborative decision-making. With the many layers of data sets accruing by the minute in the digital age, life-sciences companies frequently struggle to fuse these data sets — sales and payer plan data, primary market research, etc. — into deeper insights into the market, says Brian Irwin, VP, business strategy, Trinity Pharma Solutions. In particular, companies face the business and technical challenge of creating bridge files and integrating multiple datasets coming from various sources and delivered at different frequencies.
“In the past three to four years there has been a dramatic shift in the marketplace around leveraging data to drive more patient-centric and economic strategies," Mr. Irwin says. “For example, platform data integration can bring together hundreds of pieces of data across commercial enterprise and mobilize them for use, as well as provide a 360-degree view of the patient, provider, and payer."
Outsourcing to these platforms can unlock pretty substantial value that resides in data sources today.
Mr. Irwin says there are 50 different ways to look at data; a significant driver of value is to centralize it in a single guided analytic framework.
Cross-Team Collaboration
As technology gets a stronger foothold in all business processes, the digitalization of the organization spurs more robust cross-team collaboration. The disparity in perceptions between functions is greatest between CIOs and CMOs. According to Shawn Roman, managing director-life sciences, digital customer, at Accenture, CMOs and CIOs in the pharma industry are rarely on the same page — or even the same planet — in their vision for what should be happening in terms of investment in technology to streamline processes.
“Due to this misalignment, the industry is missing many opportunities to optimize the positive impact of digital technologies," he says. “In fact, life sciences as an industry has the greatest gap between the two roles out of nine other industries studied."
Accenture reports that nine out of 10 large pharmaceutical company CIOs (91%) in mature markets believe that their company is in need of greater marketing/IT alignment. By contrast, fewer than two out of three pharmaceutical CMOs (58%) agreed with that statement. This discrepancy of 33% is far greater than the average 14% discrepancy across other industries.
These numbers point to one of the greatest challenges faced by CEOs: getting the two groups to work together. The survey also pointed to a lack of a common vision for technology and understanding between CIOs and CMOs in the pharma industry. For example, two-thirds (67%) of the pharmaceutical company CMO respondents do not view IT as a strategic partner, compared with 50% of CMOs surveyed in all industries, who share that view. Additionally, while 80% of pharmaceutical CIOs see the need for greater alignment with CMOs, just 44% of pharmaceutical CMOs feel that way. CIOs and CMOs in the pharmaceutical industry also have differing attitudes on how to make alignment work between the two functions: almost four out of 10 pharmaceutical CIOs (38%) would favor co-locating IT and marketing staff, an option favored by only 13% of pharmaceutical CMOs. At the same time, nearly four out of 10 pharma CMOs (38%) said they would support creating an IT lead within the marketing function, and a marketing lead within IT, a solution that only 19% of pharma CIOs favored.
“Digital is breaking down the walls of old functional silos in commercial, R&D, supply chain, and IT and the industry needs to  fully embrace digital to drive the types of experiences that consumers and patients in this world are expecting," Mr. Roman says. “Starting top down, collaboration between CIOs and CMOs is absolutely a challenge for today’s CEO. Most CMOs are not viewing CIOs as a partner, as organizational constraints and cultural aspects of pharma have created walls between marketing and sales that really haven’t come down. The industry’s legacy business models need to adapt to a newer landscape that is experiencing significant and generational change. The challenge is that pharma has to operate in the existing world while looking toward the future and that is a sideways shift."
fully embrace digital to drive the types of experiences that consumers and patients in this world are expecting," Mr. Roman says. “Starting top down, collaboration between CIOs and CMOs is absolutely a challenge for today’s CEO. Most CMOs are not viewing CIOs as a partner, as organizational constraints and cultural aspects of pharma have created walls between marketing and sales that really haven’t come down. The industry’s legacy business models need to adapt to a newer landscape that is experiencing significant and generational change. The challenge is that pharma has to operate in the existing world while looking toward the future and that is a sideways shift."
To foster greater collaboration between business functions and IT, CIOs have to articulate a clear vision of how technology can help transform businesses and drive growth and generate value. In certain companies, CIOs request the support of CEOs to help communicate this message top down. It is then important to demonstrate a proof of value through a number of small projects and pilots to generate quick wins, which can then give the confidence to the business leaders and foster greater collaboration, Mr. Sambasivan says.
In addition, whatever the scope of the project, senior business and IT management should be advising the CEO as to where to begin, where to head, what the end result is expected to be and clearly articulate the compelling business case, the associated risks, and mitigation options. (PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Inside the C-Suite
Life-sciences C-suite executives are coping with critical technology issues such as the cloud, data security and storage, asset management, and using more data to accelerate development, clinical trials, improve sales, as well as CRM programs, to name
just a few challenges. Thought leaders provide best practices and solutions that can be used to smooth the transformational
shift across functions and organizations.
Elisa Cascade
President, DrugDev Data Solutions
One challenge to using more data is the lack of a single, unique identifier that can be used to integrate user profile and facility information. To address this issue, DrugDev has developed a process called canonicalization that matches persons and facilities to a unique identifier through a combination of automated algorithms and manual curation. In addition to our own network, the DrugDev Golden Number is being used to integrate information from pharma and CRO CTMS, online profiles, third-party providers, and public sources. This number also enables data sharing with investigator consent for collaborations such as the Investigator Databank and the TransCelerate Investigator Registry.
Zaher El-Assi
President,
Merge eClinical
My take on technology adoption is pretty simple: we thrive or perish based on leadership. The mechanics of it all will work out. It always does. Big data, the cloud, complex systems integrations — they all have huge dependencies and intricacies, but the right team aiming in the right direction can always manage it. The key questions really are, “What are we aiming at? Why? What can we gain? Why should I buy in to this vision?" That’s where good leaders shine. They can articulate what’s ahead in a way that sets — and keeps — everything moving forward faster and with more confidence.
Sujay Jadhav
CEO, goBalto
Workflow-driven tools based upon SOPs help manage and automate the entire lifecycle, by normalizing and tracking decision points and global processes. These tools ensure that “tribal knowledge" is retained during staff turnovers by helping to reduce staffing and training costs. Additionally, these systems can be used to track cycle times and other metrics to enable predictive analytics and drive process improvements. Today, some companies still manage processes with spreadsheets or opt to spend significant budget on customizing tools. Forward-thinking companies use workflow-driven solutions and enjoy reported savings of up to 35%.
Mike Jagielski
CEO, KCR
After the competition between CROs and pharma companies for the same patients across similar programs, technology is the No. 2 challenge for the CRO industry. Making intelligent technology choices is critical particularly for a mid-sized CRO, as implementing many systems does not always mean more efficiency. At KCR we have chosen a path leading to the multipurpose usage of the same technology across different processes. We use our EDC technology as a data management tool but also for our CTMS, start-up project management, and recruitment, and we still see the potential for other applications. We have to keep overhead costs down and minimize training time — it is critical for every CRO, and especially for the very competitive mid-sized CRO market. The eTMF solution will take a bigger market share, and global and complex projects will require easy adaptation of platforms. It is interesting to see how standards will evolve in the future, as we see more unique complexities that require a different approach. Patient engagement technologies will be the next big trend. I believe that at this moment in time, we have no notion about how this will reshape our fundamental understanding of clinical trial conduct. In five to 10 years we will witness new CRO business models developing around technology platforms, combined with medical, clinical, and process knowledge. In a longer perspective of 10 to 15 years we will also probably see the patient being in the center of the value creation chain, with less importance of the investigative site. All signs of today point in this direction.
Jeff Lee
Managing Partner, Cumberland Consulting Group
With mergers and acquisitions on the rise in the pharmaceutical industry, C-Suite executives are confronted with the challenge of fully integrating two companies in short timeframes. Technology plays an important role in this process. To provide better access to data and absorb future acquisitions in a more efficient manner, organizations must consider consolidating systems, using industry-standard technology, implementing commercial off-the-shelf (COTS) systems, and moving to the cloud. SaaS and cloud solutions allow for the flexibility, accessibility, and scalability needed to effectively manage the challenges of evolving IT infrastructure and rapid company growth to meet business objectives.
Ronald Maheu
President and CEO, PharmaCommunications Systems Inc.
As organizations start applying critical technology solutions, the opportunity for synergy becomes immense. Leveraging that synergy is a key best practice element to smooth a company’s trip through the transformational shift. Recognizing this, SaaS vendors have built high-security, high-availability, and scalable platforms that enable companies to connect a group of multi-disciplinary vendors into one cloud, and have them all contribute toward achieving the organization’s business objectives. This can be as simple as CRM and promotional management systems working together, or as complex as a cloud that joins together the entire end-to-end product-development to end-user-marketing continuum.
Courtney McBean
VP of Clinical
Innovation, BioClinica
Thanks to technology, our industry stands to welcome across-the-board change and improvements in how it operates. Technology advances in clinical trials enable risk-based monitoring implementation. Unprecedented improvements await in areas, including informed decision-making, quality and performance, operational efficiencies, and innovation in therapy development. Change management, however poses barriers to technology adoption and progress. Leadership can overcome these by: setting and supporting a transition goal and milestones; fostering a climate of change; and heeding the power of individuals to influence and drive change processes. So why wait? Engage champions of change at functional levels and allow them to lead the way.
Patrick Sim
Chief Information Officer, West Pharmaceutical Services
Understanding the benefits and risks associated with the cloud is the Holy Grail for everyone, and it is not a simple answer. Key considerations in selecting a cloud partner to work with include whether the partner understands the priority of security, has the right focus on it and has the financial backing to fund the investments necessary to protect your information. The ability to invest in their security and ours is a major advantage of the larger cloud providers. It is the responsibility of corporate leadership to determine to whom and how we grant access to these services. I look at it like a banking relationship: banks keep money safe and protected, but the consumer needs to be responsible for understanding how his/her information is accessed. Using technologies like two factor authentication, which many financial institutions offer, coupled with good password discipline, is an example of how you can protect access to your information. The other thing to consider is: Who do you give access to your information? Third parties expose you to an extra level of risk. In the end, cloud services are the future, and we need to keep making it safer and safer, but the bad guys will always be out there trying to extract something of value. We are currently working toward a hybrid cloud environment for many reasons, not just security.
Mike Wilkinson, Ph.D.
Executive VP, Chief
Information Officer, Head of Technology, Innovation and Performance, PPD
While it may be true that pharmaceutical companies are moving away from outsourcing their corporate IT function, we see an opposite trend occurring specifically in the clinical development area. While R&D spend remains relatively flat across the industry, outsourcing clinical development services to CROs is increasing. The primary business driver behind this trend is the potential for faster trials at lower costs. Leaders in pharmaceutical companies are faced with essentially two different models for clinical technology from CROs. The first is a product-based model in which the CRO sells its technology products — either built in-house, purchased, or as a commercial software-as-a service — along with its services in an effort to increase customer loyalty or stickiness. This model has four fundamental disadvantages: CROs are not technology companies and can’t possibly compete with modernizing, upgrading, supporting, and maintaining software; clinical technology is becoming commoditized, so that differentiation, if it exists at all, is fleeting; this model doesn’t account for the importance of the data and analytics; and CROs betting on customer loyalty being created through technology products is causing them to stray from what should be the core focus of a service company. In contrast, I believe the industry will move to the second of the two models. In this approach, the CRO closely partners with high-tech companies to allow pharmaceutical customers to leverage best-in-class technology that is CRO agnostic. In other words, the selection and retention of the CRO is not dependent on the technology. In this model, pharmaceutical companies can reduce their overall technology spend, variabilize what costs they do have, access the most modern, cutting-edge software, get world-class support and maintenance, and expect better integration and “plug and play" capabilities. All this results in better access to data and, most importantly, the opportunity to realize and retain the best CRO based not on its technology but on its people, process, data, service, and quality.
~~~~~~~~~~~~~~~~~~~~~~~~~


















