McKinsey: Telehealth Use 38 Times Higher Than Pre-COVID-19 Use
Trend Watch: Telehealth Gets a Push from COVID-19
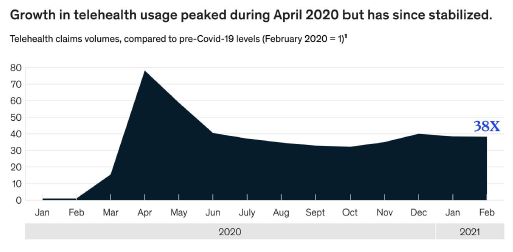 Overall use of telehealth is at levels 38 times higher than before the COVID-19 pandemic, ranging from 13% to 17% of visits across all specialties, according to a recent McKinsey report. Usage has dropped slightly since its peak in spring 2020, but patient and physician attitudes toward telehealth have improved. About 40% of surveyed consumers said they planned to continue using telehealth moving forward, up from 11% before COVID-19. After that initial spike, utilization levels dipped and have been largely stable since June last year.
Overall use of telehealth is at levels 38 times higher than before the COVID-19 pandemic, ranging from 13% to 17% of visits across all specialties, according to a recent McKinsey report. Usage has dropped slightly since its peak in spring 2020, but patient and physician attitudes toward telehealth have improved. About 40% of surveyed consumers said they planned to continue using telehealth moving forward, up from 11% before COVID-19. After that initial spike, utilization levels dipped and have been largely stable since June last year.
But a gap has historically existed between consumers’ expressed interest in digital health products and actual use. McKinsey says continuing to focus on creating a seamless consumer interface, breaking down silos in care provision across virtual and in-person with improved data integration and insights, and proactive consumer engagement will all be important to sustaining and growing consumer use of virtual health as the pandemic wanes.
On the provider side, 58% of physicians continue to view virtual care more favorably than before the pandemic, though that’s down slightly from September, when 64% of physicians were in support. As of April 2021, 84% of doctors were offering telehealth, and 57% said they’d prefer to continue offering this as an option. However, they expect reimbursement: 54% of doctors said they wouldn’t provide virtual care if it was paid at a 15% discount to physical services.
CMS has already significantly expanded reimbursable telehealth codes, adding 144 telehealth services in 2020 temporarily covered by Medicare and codifying nine permanently in a December payment rule. However, the new additions only apply to patients in rural areas in a medical facility, more meaningful changes would require congressional approval.
Spencer Smart Hub Improves Patient Adherence, Relationships
 Spencer Health Solutions promotes home research and healthcare with the world’s first and only smart hub for medication and telecare to support patients at home. The new combination of services enables healthcare providers and biopharmaceutical companies to reach patients at home. Spencer’s smart hub integrates health services into a user-friendly device the size of a small food processor. Spencer provides medications, integrates data from other medical devices in the home, surveys patients for insights and analysis, and enables virtual visits to healthcare providers and participation in clinical research. More than 95% of patients using the Spencer device take medications as prescribed, and more than 90% of patients respond to health status questions displayed on the touch screen.
Spencer Health Solutions promotes home research and healthcare with the world’s first and only smart hub for medication and telecare to support patients at home. The new combination of services enables healthcare providers and biopharmaceutical companies to reach patients at home. Spencer’s smart hub integrates health services into a user-friendly device the size of a small food processor. Spencer provides medications, integrates data from other medical devices in the home, surveys patients for insights and analysis, and enables virtual visits to healthcare providers and participation in clinical research. More than 95% of patients using the Spencer device take medications as prescribed, and more than 90% of patients respond to health status questions displayed on the touch screen.
In addition to dispensing prepackaged medications and surveying patients, the device hosts telehealth virtual visits with healthcare providers. Catalyst Healthcare deployed the Spencer device through a network of pharmacies to improve care with enhanced engagement and personalized patient care.
During the pandemic, survey questions of patients after taking medications were designed to screen patients for COVID-19 symptoms.
“Providing technology to support a broad range of services is important, but the true value of the Spencer smart hub is the relationship patients build with their healthcare providers," says Spencer Health Solutions Founder and CEO Tom Rhoads. “The high level of patient engagement is proof of the value patients place on those relationships."
Amazon Web Services Introduces AWS for Health
Amazon Web Services recently introduced AWS for Health, a range of services aimed at helping healthcare and life-sciences organizations reach their goals. AWS for Health provides proven and easily accessible capabilities that help organizations increase the pace of innovation, unlock the potential of health data, and develop more personalized approaches to therapeutic development and care. AWS will allow organizations to accelerate the digitalization and utilization of their data.
The tools are aimed at addressing a range of needs, including clinical systems, analytics and AI/ML, patient and clinician experience, medical research, finance and operations, and core health IT.
Amazon’s Epic on AWS solution allows users to migrate electronic health record workloads to the cloud, with a goal of increasing performance and automating many traditional IT tasks.
Of particular interest is Amazon HealthLake, which is available in select regions. The HIPAA-eligible service uses machine learning to extract meaningful information from unstructured data, then organize, index, and store that information in chronological order.
By leveraging the Fast Healthcare Interoperability Resources industry standard format, the software enables interoperability, allows users to analyze the newly structured data, and makes it easier for organizations, researchers, and practitioners to collaborate.
The company first announced HealthLake in December 2020, joining a host of other software giants in offering data management and analysis tools. HealthLake is available in eastern and western U.S. regions, with more availability coming soon.
Apple Supplier Rockley Photonics Unveils Wearable Health-Monitoring Tech
 Rockley Photonics has introduced a health monitoring solution that it is calling a “clinic-on-the-wrist" digital health sensor system. The technology uses a miniaturized chip that provides continuous, noninvasive monitoring of core biomarkers.
Rockley Photonics has introduced a health monitoring solution that it is calling a “clinic-on-the-wrist" digital health sensor system. The technology uses a miniaturized chip that provides continuous, noninvasive monitoring of core biomarkers.
Rockley uses infrared spectrophotometers to detect and monitor a much wider range of biomarkers, which could dramatically increase the functionality of wearable devices. The differentiating element of the sensing system is a noninvasive sensor module based on Rockley’s spectroscopy technology. Rockley’s sensor module generates a large number of discrete laser outputs from a single silicon chip covering a broad optical band.
The sensor noninvasively probes beneath the skin to analyze blood, interstitial fluids, and various layers of the dermis for constituents and physical phenomena of interest. Such biomarkers have historically been measurable only by using bench-top equipment.
Rockley is initially launching its full-stack sensing solution as a wristband that contains the sensor module and communicates with an app, and it will be used in a number of human studies in the coming months, but the company suggested that its sensor module and associated reference designs, including hardware and application firmware, will be available for other consumer products.
Apple is the largest customer of Rockley Photonics. Rockley has previously said that its sensors could be in consumer smartwatches and other electronics as soon as next year.
Artificial Pancreas Helps Manage Blood Sugar Levels
 A team at the University of Cambridge and Cambridge University Hospitals NHS Foundation Trust, which previously developed an artificial pancreas with the aim of replacing insulin injections for patients living with type 1 diabetes, has shown the device can be used to support patients living with both type 2 diabetes and kidney failure.
A team at the University of Cambridge and Cambridge University Hospitals NHS Foundation Trust, which previously developed an artificial pancreas with the aim of replacing insulin injections for patients living with type 1 diabetes, has shown the device can be used to support patients living with both type 2 diabetes and kidney failure.
The device can help patients safely and effectively manage their blood sugar levels and reduce the risk of low blood sugar levels. The team recruited 26 patients requiring dialysis between October 2019 and November 2020. Thirteen participants were randomized to receive the artificial pancreas first and 13 to receive standard insulin therapy first. The researchers compared how long patients spent in the target blood sugar range (5.6 to 10.0mmol/L) over a 20-day period as outpatients.
Patients using the artificial pancreas spent on average 53% of their time in the target range, compared with 38% when they used the control treatment. This equated to around 3.5 additional hours every day spent in the target range compared with the control therapy.
The artificial pancreas is a small, portable medical device worn externally on the body and is made up of three functional components: a glucose sensor, a computer algorithm to calculate the insulin dose, and an insulin pump.
The artificial pancreas is powered by software in the user’s smartphone that sends a signal to an insulin pump to adjust the level of insulin the patient receives. A glucose monitor measures the patient’s blood sugar levels and sends these back to the smartphone to enable it to make further adjustments.
Unlike the artificial pancreas for type 1 diabetes, this version is a fully closed-loop system, whereas patients with type 1 diabetes need to tell their artificial pancreas that they are about to eat to allow adjustment of insulin, with the new version, the device functions automatically.
The research was supported by the NIHR Cambridge Biomedical Research Centre, The Novo Nordisk UK Research Foundation, Swiss Society for Endocrinology and Diabetes, and Swiss Diabetes Foundation and Swiss Kidney Foundation. (PV)




















