Robots Measure Vitals to Minimize Interactions with COVID Patients
Trend Watch: COVID-19 Forces Healthtech Innovations from Robots to Partnerships
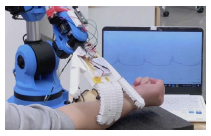 Researchers at Simon Fraser University in Canada have developed three different robots that can sense and measure patients’ healthcare parameters, including electrocardiograms, oxygen levels, respiration rates, and temperature.
Researchers at Simon Fraser University in Canada have developed three different robots that can sense and measure patients’ healthcare parameters, including electrocardiograms, oxygen levels, respiration rates, and temperature.
One of the robots, comprising a robotic arm, contains electrodes in its fingertips and can take measurements when it makes contact with a patient. Another, a smart insole, can monitor a patient’s gait when worn in a shoe. The third, a humanoid robot, can keep track of oxygen levels, which may be useful in the treatment of COVID-19 patients.
In the age of COVID-19, minimizing close contact between patients and clinicians could help to reduce transmission.
The devices were created using 3D printed origami structures, and so far have been able to determine a wide array of basic physiological data in volunteers. At present, the robots are largely passive in how they obtain information, but the researchers have said it may be possible to use artificial intelligence to allow the devices to interact with patients, and perhaps even make decisions such as prescribing medications.
“The recent pandemic demonstrates the need to minimize human-to-human interaction between healthcare workers and patients," says Woo Soo Kim, a researcher involved in the study. “There’s an opportunity for sensing robots to measure essential healthcare information on behalf of care providers in the future."
Such devices may become commonplace in the future for routine medical checkups or health monitoring.
etectRx Targets Mental Health Field In Partnership With Pharmacy
Digital health company etectRx is partnering with Local Health, a subsidiary of OneroRx, which operates telepharmacies and community retail pharmacies. The partnership is structured to evaluate patient acceptability of the etectRx FDA-cleared digital pill ID-Cap System. Local Health, located in Chicago, will recruit and engage patients, dispense the digital pill system, collect data, and publish survey results.
The study focuses on mental health medication. The etectRx technology gives Local Health the ability to intervene in real time, helping patients successfully stay adherent to their medication. Increasing patient use with Local Health allows the companies to benefit the mental health community together.
“We are launching a variety of targeted use cases and have plans to launch programs with pharma and healthcare providers to continue building data supporting the value of digital pills," says Valerie Sullivan, president and CEO of etectRx.
According to the company, the etectRx ID-Cap System holds 98% accuracy when used correctly and has recorded more than 5,900 ingestions across 15 IRB clinical research studies.
The four-part FDA-cleared system consists of an ID-Capsule, reader, patient app, and clinician dashboard.
The ID-Capsule is a digital pill with an embedded ingestible sensor. The sensor communicates a digital signal shortly following ingestion and capsule dissolution. The sensor is naturally and safely eliminated through the patient’s GI tract. The reader is a wearable device that detects and forwards messages transmitted from the ingested sensor to the patient app and clinician dashboard. The patient app allows patients to view ingestion events in real time as well as medication-use history. The app can send notifications to remind patients to take their medication. The clinician dashboard displays the information flow that starts with the ingestion of the ID-capsule and provides the clinician with both real-time notifications and a history of their patients’ ingestion events.
Evernorth Buys MDLive, Brings Telehealth In-house
Cigna’s Evernorth healthcare services business has bought MDLive, a fast-growing provider of telehealth services, which have taken off during the COVID-19 pandemic.
The deal brings a service in-house that usually health insurers pay for via contract. Cigna sees MDLive and its round-the-clock telehealth delivery platform as critical to the Evernorth healthcare services portfolio the insurer launched in September of last year.
“Customers expect more convenient care interactions, and COVID-19 has rapidly accelerated this need," Evernorth Chief Executive Tim Wentworth says. “We see an immediate opportunity to build a new model of care delivery, one that delivers a connected experience with greater affordability, predictability, and simplicity. With the opportunity to serve millions more people, and with more personalized ways to deliver care, we will have an even greater impact on our customers, clients, and partners."
Teladoc’s 2020 Revenue: Win Some, Lose Some
Benefiting from the virtual care services boom related to the COVID-19 pandemic, Teladoc finished 2020 with more than $1 billion in total revenue. However, despite the positive growth metrics, Teladoc Health’s net loss also grew from $19 million in Q4 2019 to $394 million in Q4 2020.
The company delivered 10.6 million virtual visits in 2020, up 156% from 2019. The company’s U.S. paid membership hit 51.8 million, up about 41% from users in 2019. Teladoc reported 3 million virtual visits during the fourth quarter alone. Full year access fees revenue rose 107% year-over-year, while visit fee revenue increased 132%.
“As virtual care shifted to become a consumer expectation in 2020, Teladoc Health not only met the rapidly growing demand, but we transformed our company to define a new category of whole-person virtual care," Jason Gorevic, CEO of Teladoc Health, says. “By accelerating our mission to transform the healthcare experience, we exceeded our fourth-quarter and full-year 2020 expectations and see strong momentum across our global business in 2021 as the market embraces the breadth and depth of our unique capabilities."
Looking ahead to 2021, the company projected the total revenue will be between $1.95 billion to $2 billion for the full year.
Bionaut Introduces Targeted Treatment Robot
Bionaut Labs, a company focused on revolutionizing the treatment of central nervous system disorders (CNS), has come out of stealth mode after $20 million financing led by Khosla Ventures. Other investors include Upfront Ventures, Revolution, Bold Capital, and Compound. Proceeds from the financing support the advancement of its lead therapeutic program in glioma through preclinical development, as well as help advance its program in Huntington’s disease.
“We are thrilled to bring Bionaut Labs out of stealth mode as it typifies the type of new impactful technology companies we like to help build," says Vinod Khosla, founder, Khosla Ventures. “Bionauts hold great promise as a new targeted treatment modality for severe brain disorders for which there are few, if any, effective treatment options. Moreover, the broad therapeutic potential of Bionauts extends to many diseases where conventional therapies are limited or lacking."
A Bionaut is a novel treatment modality that uses remote-controlled microscale robots to deliver biologics, nucleic acids, or small molecule therapies locally to targeted CNS disease areas. Through precision localized brain targeting, Bionaut therapeutics could offer better efficacy and safety that cannot be achieved by other traditional treatment modalities.
“Bionauts can transform the way the biopharmaceutical industry develops treatments, allowing us to engineer the therapeutic index for improved efficacy and better safety," says Michael Shpigelmacher, co-founder and CEO, Bionaut Labs. “They make it possible to get to the precise brain area we want to target, providing the freedom to explore new pathways and remove the device without leaving a footprint."
Bionauts can be constructed in different versions with custom geometries and surface characteristics. Smaller than a millimeter, they contain moving parts controlled remotely by a magnetic controller, allowing them to safely reach the target and release a therapeutic payload from the cargo compartment. Engineering flexibility provides a broad foundation for designing Bionaut therapies for nearly any disease of interest.
Microfluidic Smartphone Dongle Detects COVID-19
 Researchers at Rice University have developed a microfluidic device that can test for the presence of SARS-CoV-2 in a drop of blood and display its results once plugged into a cell phone. The test runs in under an hour, and works by detecting SARS-CoV-2 nucleocapsid protein using antibody-studded magnetic nanobeads. In tests, the device could detect very low levels of the protein, suggesting that it could function as a sensitive diagnostic tool.
Researchers at Rice University have developed a microfluidic device that can test for the presence of SARS-CoV-2 in a drop of blood and display its results once plugged into a cell phone. The test runs in under an hour, and works by detecting SARS-CoV-2 nucleocapsid protein using antibody-studded magnetic nanobeads. In tests, the device could detect very low levels of the protein, suggesting that it could function as a sensitive diagnostic tool.
This device can detect a viral protein in a small drop of blood, such as that from a finger prick. One of the other big benefits of the device is its portability. “What’s great about this device is that it doesn’t require a laboratory," says Peter Lillehoj, a researcher involved in the study. “You can perform the entire test and generate the results at the collection site, health clinic, or even a pharmacy. The entire system is easily transportable and easy to use."
The microfluidic chip contains magnetic nanobeads studded with antibodies against the SARS-CoV-2 nucleocapsid protein.
An external magnet draws the beads towards an electrochemical sensor that generates a current in response to the captured protein. When plugged into a standard smartphone, the device sends these data to an app for easy interpretation of the results.
The test is rapid, providing results in as little as 55 minutes, and can detect as little as 50 picograms of the protein per milliliter.
ObvioHealth and RedHill Biopharma Launch First-in-Kind Remote COVID-19 Therapy Trial
ObvioHealth, a global virtual research organization, has launched a hybrid clinical trial for the treatment of COVID-19, using an innovative study design with digital data capture to remotely monitor patient vitals. ObvioHealth’s platform collects both telemetric and patient-reported information; biomarkers continuously gathered from the devices include temperature, respiratory and pulse rates, blood pressure, pulse oximetry, ECG, and weight. Home healthcare nurses will make in-home visits to check in on patients, collect blood samples for certain laboratory parameters, perform virus PCR swabs at home, and oversee certain patient-reported data capture using devices provided to participants at home. This combined medical monitoring allows the study team to intervene quickly if a patient experiences an adverse event.
Otsuka and Click Therapeutics tap Verily’s Platform for Digital Depression Treatment Study
Otsuka Pharmaceutical and Click Therapeutics are making their major depressive disorder (MDD) clinical trial “remote" and are working with Verily Life Sciences to do so.
The pivotal trial will investigate the effectiveness of digital therapeutics in reducing depressive symptoms in up to 540 adults diagnosed with MDD who are on antidepressant monotherapy. The participation will last 10 weeks.
Verily will provide tools and technology to engage patients and clinicians to increase the pace of studies and collect higher quality and more comprehensive data in a more naturalistic setting. The trial will run off Verily’s Project Baseline platform.
“The COVID-19 pandemic has clearly demonstrated the need for digital treatments and fully remote e-clinical trials that go beyond the pill to empower patients, enhance connectivity between patients and their healthcare team, and ensure more diverse populations can participate in new clinical trials," says Kabir Nath, president and CEO of Otsuka North America Pharmaceutical Business Division.(PV)