 One of the most insidious characteristics of rare diseases is how isolating they can be for patients and their caregivers. Those with a rare disease don’t enjoy the social sport of comparing cholesterol numbers among friends at a dinner party.
One of the most insidious characteristics of rare diseases is how isolating they can be for patients and their caregivers. Those with a rare disease don’t enjoy the social sport of comparing cholesterol numbers among friends at a dinner party.
Theirs is a much lonelier path of confusing symptoms, unusual blood tests, and in many cases, depression associated with a long and frustrating journey to an accurate diagnosis. It can be difficult to explain their quality of life to family members, work colleagues, and even their own doctors.
There are nearly 7,000 identified rare diseases in the world and most are not taught in medical school or covered in medical journals. Even among specialists such as rheumatologists, hematologists, neurologists, etc., there is a dearth of experience with rare diseases, making them difficult to diagnose. It’s not unusual for a patient to bounce from specialist to specialist, trying to find someone who can make a proper diagnosis. According to the rare disease community, Global Genes, it takes an average of six to eight years for a patient to get an accurate diagnosis. This can mean a delay in treatment, if one is available, which can have a lasting, negative impact on the patient.
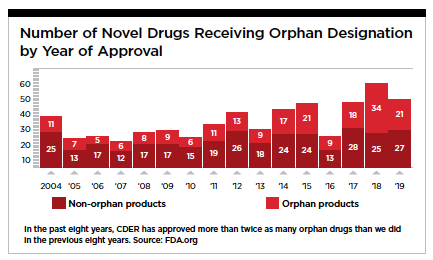
Until the Orphan Drug Act (ODA) of 1983, medicines for rare diseases were far down the priority list for most biopharma companies. Besides being difficult to diagnose and treat, the patient population is small and the potential return on investment couldn’t justify the R&D required to bring a novel drug to market. But  with the ODA’s incentive of extended market exclusivity, the door was opened to work on new therapies for the 95% of rare diseases that don’t have an approved medication.
with the ODA’s incentive of extended market exclusivity, the door was opened to work on new therapies for the 95% of rare diseases that don’t have an approved medication.
The financial incentives worked, and in the past few years, the majority of new FDA-approved drugs are for rare diseases. The confluence of genetics, AI, and a healthy biotech funding environment are fueling this growth. In addition, there is a growing recognition of the important role that advanced analytics, data science, and digital communication plays in positively impacting many aspects of the clinical experience, including drug development, clinical trial recruitment, patient identification, HCP education, and patient community development and support.
Drug Discovery Powered by AI
The speed with which COVID-19 was sequenced and the multiple vaccines developed and approved for emergency use is unprecedented. It demonstrated the art of the possible, from global collaboration, to the effectiveness of the new mRNA technology platform, to a new flexibility among global review boards.
Artificial Intelligence (AI) was instrumental in shrinking the time from concept to clinical trial to approval. This represents a promising new chapter in the speed and effectiveness of new product development in the life sciences.
For patients suffering from one of the thousands of rare diseases without a known therapy, the use of data science to level-up the speed and power of drug discovery could not come sooner. The recent experience in bringing two new mRNA vaccines to market in less than 12 months demonstrates the revolution that is occurring specifically in gene therapy. This will be invaluable to scientists approaching other rare, genetic disorders. For example, it was Moderna’s experience in developing mRNA-based therapies for rare liver diseases that led to its groundbreaking COVID-19 vaccine.
 Another promising approach to finding new therapies for rare diseases is to mine existing drugs for new indications. New effective therapies don’t always come from de-novo drug discovery. Existing or investigational drugs (even those that have failed their primary endpoint) may have potential for other diseases. The power of AI to screen existing or investigational drugs for new indications (called “drug repurposing") was used to identify potential COVID-19 therapies. One team used Amazon Web Services’ AI resources to build a comprehensive COVID-19 knowledge graph by screening 24 million PubMed publications and identifying 41 repurposed drug candidates, including dexamethasone for COVID-19 treatment (Zhou et al., Lancet (2020)).
Another promising approach to finding new therapies for rare diseases is to mine existing drugs for new indications. New effective therapies don’t always come from de-novo drug discovery. Existing or investigational drugs (even those that have failed their primary endpoint) may have potential for other diseases. The power of AI to screen existing or investigational drugs for new indications (called “drug repurposing") was used to identify potential COVID-19 therapies. One team used Amazon Web Services’ AI resources to build a comprehensive COVID-19 knowledge graph by screening 24 million PubMed publications and identifying 41 repurposed drug candidates, including dexamethasone for COVID-19 treatment (Zhou et al., Lancet (2020)).
The Accelerated Move to Digital Trials
For clinical trials hamstrung by social distancing directives, the use of digital communication, remote patient monitoring, and AI processing of large data sets have changed the way sites will manage trials in the future. There is evidence that new digital trial platforms are engaging and retaining more patients at less cost than the traditional methods, changing the clinical trial process for good. The fact that Pfizer and Moderna enrolled participant pools of 43,000 and 30,000, respectively, and reached their primary endpoints in less than four months has raised the bar on expectations for speed and accuracy.
Digital trial methodology will also bring greater efficiency to phase four and real world evidence investigations. It affords patients the opportunity to engage and report from the safety and comfort of their homes rather than having to travel to a hospital or clinic. For example, v-safe is a new CDC mobile app that automatically sends daily text message surveys to individuals after receiving their COVID-19 vaccine to track side effects. This will be one of the lasting benefits brought about by COVID-19.
Finding a Patient in a Haystack
Although there are about 30 million Americans with a rare disease, the total number of patients for any given disease can be very small. For a pharma brand team, identifying the right patients and understanding where they are in their journey is the first step to helping them begin appropriate therapy. For a patient with a rare disease, however, getting an accurate diagnosis can be a long and discouraging process. How can advanced analytics and digital communication help?
Data analytics are now being used to review large data sets of confirmed patients to better understand their disease. When patients present with a progression of divergent symptoms, each step may come with its own misdiagnosis. AI is ideal for pattern recognition and when used across a cohort of patients, it can help to pick out those with an identifiable sequence of symptoms and confidentially flag them for follow up by their healthcare professional.
In other rare diseases, there may be a common penultimate diagnosis. Advanced analytics can be used to scan claims and EMR data to find patients who may be only months away from failing a first-line therapy and being ready for a second-line alternative.
The use of AI to find patterns and predict disease progression represents one of the most powerful ways for reducing the time lapse between initial symptoms and an accurate diagnosis and treatment. For some diseases, AI-based algorithms are helping to shape clinical guidelines and decision support (Doucette et al., A Framework for AI-Based Clinical Decision Support (2012)).
For a pharma brand team, the task of identifying rare disease patients who might be candidates for an innovative therapy follows the fine line of confidentiality and data privacy. An algorithm trained on a specific disease pattern and applied to a regional set of de-identified EMR and claims data can graph where patients are on their disease progression. Even though the data is anonymous, the results can be used to inform specialists in a region about the prevalence of likely patients with the rare disease.
Activating the HCP
Because few rare diseases are covered in medical school, most primary care physicians won’t know what to look for when wrestling with a difficult set of symptoms or test results and may be uncertain in making a referral. Although specialists may be familiar with the most common rare diseases, they will only see a handful, or less, of cases in a year. When new therapies are brought to market, they will typically use an innovative technology or new mechanism of action.
These three factors all contribute to a lack of knowledge about rare diseases, the unique challenges facing patients who suffer, and the therapeutic options available.
Digital content including mechanism of action animations and case studies from researchers and leading clinicians are all important elements in building awareness and understanding among healthcare professionals. An omnichannel media strategy will deliver small, relevant bites of information that are consumable when a clinician is ready. The underlying digital content strategy will deploy a “create once, publish many" method for developing branded and unbranded messaging. For example, closerlook’s award-winning aggNETIC Attraction animated disease education experience was published across multiple digital platforms to support a new clinical story of Krystexxa (pegloticase) among rheumatologists and nephrologists with positive results.
Marketing analytics tools such as closerlook’s Backstage Intelligence are used to segment physicians along their adoption path and trigger appropriate digital content through their preferred media channel. Personality detection software such as the award-winning Backstage Relate identifies the optimal individual learning style of each clinician to create personalized content just for them.
Supporting the Patient Journey
Patients with rare diseases often feel alone and misunderstood. They will seek out others like themselves, often finding them within small, committed online communities of patients and caregivers. It’s not unusual for patients and caregivers they meet through these online patient communities to become close friends. These communities, sometimes centered around a Facebook or Instagram group, combine support with activism, passing around encouragement and the latest news on possible brand and off-brand treatments.
Online communities can be fiercely independent but also hungry for new information. Pharma brands that respect the role of patient communities and make an effort to deliver user-friendly information and insight into current research will be welcome.
Another challenge for patients is the lack of public awareness of their disease. Online content and digital storytelling through the creation of film festival-worthy documentaries or global and small-group meet-ups can be important elements in validating and raising awareness of rare diseases.
The Path to Hope
As demonstrated by the stunning speed with which new vaccines and treatments for COVID-19 were developed and approved, the role of data science and the digital transformation of the drug development and commercialization journey offers a promising approach to unlocking new drugs for patients with rare diseases. This is not lost on the small gene therapy and biotech firms that are intensifying their focus on rare diseases.
There is also an important role that specialized digital agencies can play to help identify new patients, activate physicians, and support the broader patient and caregiver community with AI/advanced analytics and digital communication tools. Together with our biopharma partners, we can bring hope and support to patients in need of a new path to healing.(PV)
closerlook, inc., an analytics-driven digital-native marketing agency serving biopharma brands, creates specialized omnichannel marketing programs that drive demand and sustain customer loyalty by focusing on what matters most — better outcomes for patients and greater business impact for the brands that serve them.
For more information, visit closerlook.com.


















