New opportunities are available in spite of a year not worth repeating.
 Although 2020 was an incredibly challenging year for conducting clinical research studies due to COVID-19, many leaders acknowledge it forced the industry to rethink processes and creatively address the barriers that have often been pain points.
Although 2020 was an incredibly challenging year for conducting clinical research studies due to COVID-19, many leaders acknowledge it forced the industry to rethink processes and creatively address the barriers that have often been pain points.
 Know Rare fielded a survey among professionals in advocacy, medical affairs, and clinical operations in early fall 2020 to capture the major pain points of clinical trial recruitment. The top four areas of concern identified are:
Know Rare fielded a survey among professionals in advocacy, medical affairs, and clinical operations in early fall 2020 to capture the major pain points of clinical trial recruitment. The top four areas of concern identified are:
1. Identifying patients
2. Ability to follow up and track inquiries
3. Getting a signed informed consent form
4. Tracking milestones
Need for Flexibility
The inability to travel created challenges and forced greater flexibility in clinical studies for rare disease.
With many medical centers still closed or operating on a reduced schedule with a backlog of existing patients, carving out visits to screen potential participants may be a luxury they cannot afford.
Many people with rare diseases feel particularly at risk and will be less likely to want to travel for screening. Prior to the pandemic, few centers or doctor’s offices engaged in telehealth visits —either by phone or video chat — and now these are becoming the mainstream for many practices. In fact, at least 30% of all visits scheduled across all specialties through ZocDoc, one of the largest doctor directories, were for video telehealth appointments. Telehealth makes screening less burdensome for patients and is more efficient for study centers that are trying to maximize in-person visits for existing patients.
Mobile devices may also help with disparities in participation in clinical trials among diverse and underserved people across all diseases. Penetration of mobile smart phones is similar among all segments of the population.
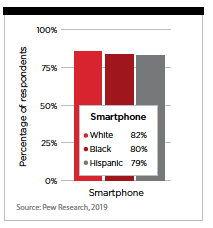 Mobile phones allow trial candidates to participate in screening without missing a work day or without the need to travel. Virtual visits can be scheduled during lunch hour or at break times, offering greater flexibility. Challenges may still remain in some areas, such as the bandwidth needed for video calls and how telehealth visits are reimbursed. Overall, the accelerated adoption for telehealth was due to the pandemic and is one change that both clinicians and patients want to stay.
Mobile phones allow trial candidates to participate in screening without missing a work day or without the need to travel. Virtual visits can be scheduled during lunch hour or at break times, offering greater flexibility. Challenges may still remain in some areas, such as the bandwidth needed for video calls and how telehealth visits are reimbursed. Overall, the accelerated adoption for telehealth was due to the pandemic and is one change that both clinicians and patients want to stay.
According to Jake Wachsman, Know Rare’s chief operating officer and co-founder, new technologies will be part of the solution for trial recruitment. “The industry, healthcare providers, and the FDA were forced by the pandemic to embrace technology at an accelerated pace; it has proven its worth and will continue to do so," he says. “Technology is the engine that fuels our effort to make it more efficient in finding, matching, and screening potential participants for rare disease studies, but it also enables us to track in real time all of the actions of the participant from inquiry to the signing of the informed consent form."
A Solution Built for Patients
With the Know Rare platform, artificial intelligence (AI) matches the application filled out by a potential participant to the database of rare disease studies culled from clinicaltrials.gov. The application can be filled out once, and the system will continually serve up potential clinical studies to participants who match the inclusion and exclusion criteria. Once they match, a potential participant can apply to a study and choose a study center.
Immediately, a patient’s application is routed to the study center team, who receives the profile of the applicant, which they can either accept or decline. If they accept, a telehealth virtual visit is scheduled through the platform, with reminders sent to each party prior to the call — conducted via audio or video. At the virtual visit, the more in-depth screening questions are asked, and if the study team feels the person may qualify, they can present the informed consent for signature. The signing can be done electronically, and whether signed or unsigned, all actions are captured in the platform’s database.
“From our network of sponsors, researchers, and clinical trial coordinators in rare disease, we heard one thing: tracking inquiries is a pain point," says Nina Wachsman, CEO and co-founder of Know Rare, who spent months interviewing stakeholders in the rare disease community before building the Know Rare recruitment platform.
Know Rare is unique in that it provides a dashboard of all actions to sponsors and participating study centers, and makes tracking inquiries burden-free, requiring no input from study center staff, making it easier to follow up on inquiries via smartphone. Additionally, the system can provide the real-time insights that can help spot issues and enable them to be addressed immediately to accelerate recruitment.
Know Rare is an offering built on a commitment to rare patients and the desire to help researchers in the discovery of new therapies for rare disease. The Know Rare platform can ensure a more accessible, ethical recruitment and perhaps a more rewarding experience to people with rare diseases.
For more information, visit knowrare.com.










