A Therapeutic-Area-Specific Approach to Data Capture Solutions
 For data capture, CNS and pain trials are demanding, with complicated eligibility criteria, extensive monitoring requirements, comprehensive data collection, and substantial demands on patients. To design a great solution, we consulted key CRO and pharma opinion leaders in eCOA, ePRO, and direct data capture (DDC).
For data capture, CNS and pain trials are demanding, with complicated eligibility criteria, extensive monitoring requirements, comprehensive data collection, and substantial demands on patients. To design a great solution, we consulted key CRO and pharma opinion leaders in eCOA, ePRO, and direct data capture (DDC).
Their pain points and expectations for service and technology in this space were elucidating. Yes, they outlined desirable functionality. But they also expressed a desire for experience-based guidance. Transactional delivery of ePRO/eCOA/DDC wasn’t enough. These customers wanted an experienced, consultative partner that could deliver not only the most useful 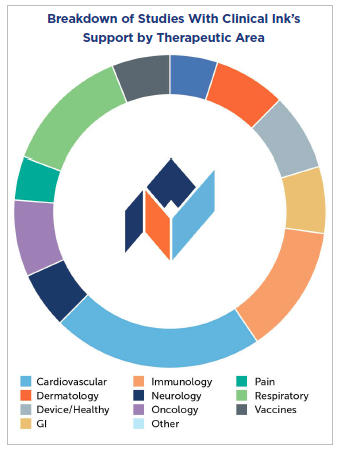 tools, but also best practices for how to use those technologies in the client’s therapeutic area.
tools, but also best practices for how to use those technologies in the client’s therapeutic area.
Clinical Ink’s specialized pain/CNS/neurology offering is the culmination of these conversations.
A Service-First, End-to-End Methodology
From initial meetings through data archiving, facilitating specialized therapeutic area solutions requires specialized services and technologies. Implementing the proper functionality with exceptional quality and timelines requires feedback, guidance, and support, from translation to help desk.
Options such as BYOD, indication-specific questionnaires, eligibility requirements, edit checks, and remote monitoring are important, as are patient engagement and patient centricity. Clinical Ink’s end-to-end methodology incorporates best-in-class services for full therapeutic area suite solutions built on experience with more than 435 unique scales/questionnaires in 250+ global studies.










