Many of us are identifying the proverbial silver linings associated with the changes we have been forced to make in response to the COVID-19 pandemic. One rather sweeping silver lining is the increased and accelerated use of technology in both our business and personal lives, with an obvious example being the now pervasive use of web conferencing.
Many of us are identifying the proverbial silver linings associated with the changes we have been forced to make in response to the COVID-19 pandemic. One rather sweeping silver lining is the increased and accelerated use of technology in both our business and personal lives, with an obvious example being the now pervasive use of web conferencing.
An element that lies at the intersection of business and personal is health, specifically, clinical trials. In the pre-pandemic world, patients, sites and sponsors were all seeking to improve clinical operations for different yet inter-related reasons. Let’s take a look at how COVID-19 has accelerated some of these changes as well as best practices for the post-pandemic clinical ecosystem.
Meeting Clinical Objectives with Virtual Trials
The major objectives for clinical trial operations have not substantially changed with the pandemic. They include safer trials, efficient sites, shortened timelines, regulatory adherence, and engaged patients. What has changed is the desire for increased execution optionality.
Sponsors need to ensure that their trials can successfully persist under any future challenge.
Traditional site-centric execution is no longer sufficient to ensure trial persistence.
The pandemic has brought that dependence to sharp resolution.
In response to this, many sponsors are following the FDA’s March Guidance and implementing alternative methods to safely continue their trials. One of these methods is virtual trials (also 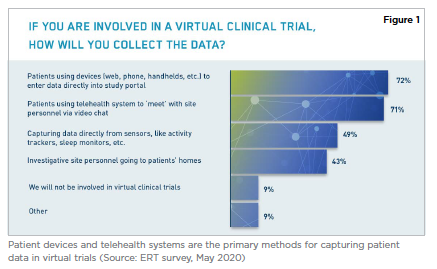 called digital or decentralized trials) or hybrid execution, where traditional site-based and virtual elements are incorporated into trial design.
called digital or decentralized trials) or hybrid execution, where traditional site-based and virtual elements are incorporated into trial design.
According to a survey of clinical professionals conducted by ERT in April/May 2020, 73% of respondents said the virtualization of clinical trials was either very or extremely critical to their organizations. Another 33% stated they were conducting virtual trials prior to the COVID-19 pandemic, 75% claimed to be adding virtual elements to their clinical trials due to the pandemic, and 7% have gone completely virtual. These survey results suggest that compliance with COVID-19 mandates is accelerating the adoption of virtual trials. This may allow some studies to continue that might otherwise have been shut down, and it also allows key stakeholders to benefit from the core advantages of virtual trials. Necessity as the mother of invention has accelerated the adoption of virtual solutions and is changing the paradigm for trial execution.
“COVID-19 mandates are accelerating the adoption of virtual trials, allowing key stakeholders to recognize their core advantages over traditional site-centric models."
Improved Patient Engagement
One of the great benefits of virtual trials is improved patient engagement. Keeping patients engaged from their first to last visit is challenging during the best of times. Well before the onset of the pandemic, sponsors and sites pinpointed patient-centric trials as a primary goal, as many study participants reported a correlation between their lack of engagement and the 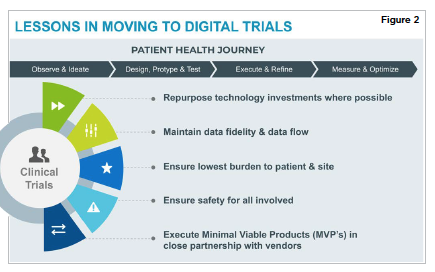 lack of transportation convenience, training, coaching and information on study results.
lack of transportation convenience, training, coaching and information on study results.
Due to restrictions put in place in response to the COVID-19 pandemic, sponsors and CROs face new threats to patient engagement and retention. Restrictions such as social distancing or stay-at-home orders may increase missed visits and patient non-compliance. Functionality that is part of the virtual trials paradigm, such as home monitoring, remote interviews and home visits that engage patients at a distance are critical to keeping them active and ensuring the flow of high quality data.
In the survey, when asked about “the big issue that keeps you up at night," 79% of respondents cited patient recruitment. Further, when asked about the biggest issues impacting clinical trials through 2020, 43% indicated patient related issues such as recruitment, enrollment, retention, and compliance. Virtual trials provide an answer to these issues by addressing the challenges of:
Needing to recruit a sufficient number of targeted patients within a certain radius of the site
Keeping patients engaged and in the study through the last visit
Connecting with patients via virtual visits
Conducting patient home monitoring and assessments
Collecting safety and efficacy data in a home visit by site personnel or clinician
Collecting site-based eCOA assessments remotely
Collecting image scans from local providers not involved in the study
Enabling patients to administer their own safety and efficacy assessments through advanced technology
Offering significantly improved patient convenience
Let’s Focus on Endpoints
We need high fidelity study data we can trust — for regulators, patients and our businesses. The mechanics of virtual trials help deliver trustworthy data — and more of it. Virtual visit capabilities have become increasingly critical due to social distancing, travel restrictions, and diminished site staff levels or closed sites (Figure 1). Tools that visualize performance indicators like study progress and compliance, and analyze safety and efficacy data, are also key to proactive study-management decisions that protect study outcomes. Virtual trials address the data visualization challenges of:
Integrating with ECG, spirometry and imaging equipment
Collating and gaining insight into the study, site and patient data through eCOA
Managing risk of low compliance driven by little/no in-person patient interaction with sites
Connecting imaging readers/radiologists to the tools they need despite travel restrictions
Integrating geographic COVID infection rate data so sponsors and CROs can proactively intervene where patient and site protocol compliance may be at risk
The sources of data in the virtual trials ecosystem are more reliable than many sources within traditional trials. This includes the increased amount of data from devices, imaging and sensors, such as ECGs and spirometers, as well as the data that is more thoroughly and regularly input directly into online portals by patients and clinicians, as opposed to the data transcribed from manual charts and diaries.
Virtual trials bring the trial closer to the patients through eCOA capabilities and at-home assessments provisioned by devices, web and BYOD. They minimize the obstacle of not capturing enough data to close the study and increase the amount and accuracy of analytics through built-in business intelligence and dashboards.
Supporting the Patient Health Journey
Sponsors and CROs can concurrently support the patient health journey and achieve their own objectives and obligations of the clinical trial. As they evolve to virtual trials and take advantage of all technology has to offer, there are some key actions they should take (Figure 2):
Repurpose technology investments where possible. This will save the organization time and money.
Maintain data fidelity and data flow. Try to avoid increased variability, and document novel approaches/procedures/activities in endpoints captured. Accurate and plentiful data increases the value and decreases the timeframe of the study.
Reduce burden to patients and sites. This will increase retention and shorten timeframes. Patient burden can be decreased by not requiring travel, and by making it easy to input data on a regular basis.
Execute Minimal Viable Products (MVPs) in close partnership with vendors. Develop prototypes that can be easily adapted to other customers, trials, and protocols for efficient implementation into specific ecosystems.
According to the recent ERT survey, one-third of companies polled are cancelling one or more studies due to the constraints of the COVID-19 pandemic. Some studies are candidates for virtual trials and some are not. There are situations in which patients simply must visit the study sites. But perhaps some percentage of the one-third could have continued with their studies had they been designed with virtual trial options.
There are certain things that will not be the same post-pandemic, and clinical trials is one of them. Carrying on through the restraints imposed by COVID-19 has forced us as a society to accelerate our digital transformation and adopt the use of technologies faster than we otherwise would have. Grandma and Grandpa probably would not be using web conferencing absent quarantines, and clinical trial sponsors and CROs probably would not be embracing the virtual trial ecosystem as quickly as they are absent pandemic implications. So there are silver linings. Business and families have found a new way to work and connect, and our industry is utilizing a new way to conduct clinical trials that can shorten timeframes, improve the experience for participants and site coordinators, ensure continuity of execution and ultimately, get valuable treatments to patients faster. And that’s what it’s all about.(PV)
ERT is a global data and technology company that minimizes risk and uncertainty in clinical trials, so that sponsors can move ahead quickly — and with confidence.
For more information, visit ert.com.