Apple and Google Partner on COVID-19 Contact Tracing Technology
Trend Watch: IoT technologies are being used across a wide spectrum for COVID-19 services
In April, Apple and Google announced they were building software into smartphones that would tell people if they were recently in contact with someone who was infected with COVID-19. The alliance of these two rivals, which pulled together quickly, shows the “seriousness of the health crisis and the power of the two companies whose software runs almost every smartphone in the world," reported The New York Times.
The companies said they would release the tool within several months, building it into the operating systems of the billions of iPhones and Android devices around the world. The software would enable the smartphones to constantly log other devices they come near, enabling what is known as “contact tracing" of the disease. People would opt in to use the tool and voluntarily report if they became infected.
The partnership enables the use of Bluetooth-enabled contact tracing technology that could help governments and health agencies reduce the spread of the coronavirus. Apple and Google plan to launch a comprehensive solution that includes application programming interfaces (APIs) and operating system-level technology to assist in enabling contact tracing.
Both companies will release APIs that enable interoperability between Android and iOS devices using apps from public health authorities. The official apps will be available for users to download via their respective app stores. Apple and Google will work to enable a broader Bluetooth-based contact tracing platform by building this functionality into the underlying platforms.
The Bluetooth-based contact tracing platform would allow more individuals to participate if they choose to opt-in, as well as enable interaction with a broader ecosystem of apps and government health authorities.
PatientKeeper Launches Mobile Tool to Help Healthcare Providers Treat More Patients
 To meet healthcare providers’ urgent and growing needs for mobile anytime, anywhere access to patient data and secure messaging with physician peers, PatientKeeper created PatientKeeper Clinical Communications Suite Now. Its launch is timely as healthcare providers prepare to treat more patients, more quickly, under surge conditions in a variety of venues during the COVID-19 pandemic.
To meet healthcare providers’ urgent and growing needs for mobile anytime, anywhere access to patient data and secure messaging with physician peers, PatientKeeper created PatientKeeper Clinical Communications Suite Now. Its launch is timely as healthcare providers prepare to treat more patients, more quickly, under surge conditions in a variety of venues during the COVID-19 pandemic.
PatientKeeper Clinical Communications Suite Now optimizes and integrates with MEDITECH acute-care electronic health records systems that do not provide such native functionality.
The Clinical Communications Suite Now provides immediate access to patient records via native iOS and Android apps on smartphones and tablets; securely instant messages care team members, consultants, practice administrators, and any other necessary hospital staff, with access to the full patient record embedded in the message; share quick notes about patients with other providers using a simple “scratch pad" to capture the most salient points — ideal for handing off to coverage and/or in a high-volume, high-throughput crisis care / triage environment; and supports telemedicine practices by enabling remote access to patient records.
MicroMass Launches COVID-19 Compass Website
 To help support others during the COVID-19 pandemic, MicroMass has created a new website, called COVID Compass. On the site, people can find information on how to build life skills to help navigate the “new normal." It is not a news source, but a place to find scientifically-proven techniques and coping mechanisms for remaining physically and emotionally healthy in this challenging time of the COVID pandemic.
To help support others during the COVID-19 pandemic, MicroMass has created a new website, called COVID Compass. On the site, people can find information on how to build life skills to help navigate the “new normal." It is not a news source, but a place to find scientifically-proven techniques and coping mechanisms for remaining physically and emotionally healthy in this challenging time of the COVID pandemic.
The site features videos and downloadable activities centered around three focus areas: finding Reliable information, connecting with people, and reducing stress.
The site, which is based on behavioral science and public health principles, is available to everyone.
UK Diabetes App Broadens Scope to Include COVID-19 Symptoms
A clinically validated app, Intellin, developed by the U.K. company Gendius helps patients manage their diabetes. Gendius is working with the NHS and other healthcare partners to add features that can help users to identify symptoms of COVID-19, while staying on track with their diabetes management. New features released in January also mean that the app can integrate with a wide variety of smart devices, including blood pressure cuffs and other wearables.
The Intellin app uses a smart algorithm to assess comprehensive healthcare information, that has either been added manually or via a direct link to a patient’s medical information, as well as smart device data, to highlight the most likely areas for diabetes complications. Using this data, the app provides tailored, medically approved educational content to help patients manage their diabetes and reduce the likelihood of developing serious complications.
Intellin has now been downloaded more than 68,000 times in countries all across the world, with over 1,000 downloads so far this year in the U.K. alone.
Work is underway to further validate the effectiveness of the Intellin app, including a clinical study with Salford Royal NHS Foundation Trust looking at recurrence of diabetic foot ulcer. Results of this study are expected later in 2020.
Debiotech Working on COVID-19 Microneedle Vaccine Delivery
 Debiotech is collaborating with vaccine companies, foundations, and public organizations to use its unique DebioJect Microneedle to improve the effectiveness of COVID-19 vaccines by reducing the necessary dose to achieve a sufficient immune response.
Debiotech is collaborating with vaccine companies, foundations, and public organizations to use its unique DebioJect Microneedle to improve the effectiveness of COVID-19 vaccines by reducing the necessary dose to achieve a sufficient immune response.
It is already well-documented that the immune response induced by intradermal administration of one-fifth of an intradermal dose for rabies is equivalent to the full dose given intramuscularly. In the case of influenza vaccine, the same intradermally injected dose has been shown to elicit a superior immune response in the elderly. These micro-needles therefore address two key issues related to the current pandemic: faster access to a larger share population with the same vaccine production capacity and better protection of the most vulnerable part of the population.
A recent publication in the Lancet demonstrated the value of the intradermal approach in the case of COVID-19: microneedles were used to deliver a subunit vaccine in a pre-clinical in-vivo animal trial, inducing the production of specific antibodies within two weeks from the injection. “DebioJect has already been successfully used on humans in multiple studies and can be produced at very high scale, thanks to a unique MEMS production process we have been working on for more than 10 years," says Dr. Laurent-Dominique Piveteau, CEO of Debiotech.
ERT Offers First Patient-Administered ECG Assessment for Continuation of Clinical Trials during COVID-19
ERT, a global data and technology company that captures critical endpoint data, entered into a first-of-its-kind partnership with AliveCor, an AI-based, personal ECG technology. The partnership enables ERT to capture digital cardiac safety data with KardiaMobile 6L, the only FDA-cleared personal ECG for patient-administered 6-lead data collection.
The hand-held device records lead II data without the attachment of electrodes. Data captured from the device can be integrated into ERT’s software and workflow platform and read by ERT cardiologists to ensure patient safety during the clinical development of new medical treatments. ERT provides high-quality measurements for QTc, QRS, and other ECG intervals that enable efficacy and safety monitoring during clinical trials.
“We are enabling our customers to continue developing new medical treatments during the COVID-19 pandemic," says Ellen Street, executive VP of cardiac safety, ERT.
Smart Temperature Monitors Could Help Manage Coronavirus
 Polysense Technologies has developed a series of smart human body temperature monitoring products based on Semtech’s LoRa devices.
Polysense Technologies has developed a series of smart human body temperature monitoring products based on Semtech’s LoRa devices.
Real-time data from the temperature sensors enables healthcare workers to efficiently screen individuals with a high temperature. Semtech supplies analogue and mixed-signal semiconductors and advanced algorithms, and Polysense develops IoT solutions for fiber and wireless sensing.
Polysense offers its new LoRa-based human temperature monitoring products in two variants. A wireless sensor for individual use, the WxS x800-IRTM, which uses thermal sensing technologies and embedded LoRa devices to monitor patient temperature in real time. The sensor uses connectivity based on the LoRaWAN protocol to communicate temperature data to cloud-based applications for trend analysis. Alerts are pushed to mobile devices if temperature thresholds are exceeded, allowing the patient to limit exposure to others and seek medical attention.
Polysense’s WxS x810-IRTMS is a standalone thermal meter for use in public spaces. This product enables public health officials to monitor populations on public transportation, at airports, railway stations, schools, shopping centers, and large building entrances. Deployment of the WxS x810-IRTMS takes place in less than five minutes, allowing immediate temperature screening.
TigerConnect Unveils Telehealth System to Help Providers Fight COVID-19
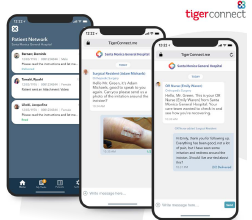 Care collaboration technology vendor TigerConnect has launched TigerTouch, a text message-oriented, audio/visual telehealth system designed to help health systems manage COVID-19 cases.
Care collaboration technology vendor TigerConnect has launched TigerTouch, a text message-oriented, audio/visual telehealth system designed to help health systems manage COVID-19 cases.
TigerTouch allows providers to control communication by initiating communications with a patient’s cell number via text message, thus solving phone-tag and voicemail problems. The system also enables care providers to securely exchange photos, videos, and file attachments on their smartphones.
TigerTouch is a mobile-centric healthcare-communications platform that enables doctors, nurses, and allied health professionals to communicate securely with patients via video, voice, or text with a single, enterprise-grade app. This is achieved without forcing a patient to log in to a portal, download an app or create another password to manage.(PV)


















