Analysts and experts discuss scientific, technological, and access advancements in the oncology area.
Oncology clinical development activities are being undertaken by more than 700 companies and are at record high levels, according a report by the IQVIA Institute for Human Data Science. But despite some improvement in oncology trial productivity and the prospect of further advances over the next five years, development remains high-risk and of long duration.
 The pipeline of oncology drugs in late-stage development expanded 19% in 2018 alone, and 63% since 2013. Within the pipeline, across all phases of clinical development, the most intense activity is focused on almost 450 immunotherapies with more than 60 different mechanisms of action.
The pipeline of oncology drugs in late-stage development expanded 19% in 2018 alone, and 63% since 2013. Within the pipeline, across all phases of clinical development, the most intense activity is focused on almost 450 immunotherapies with more than 60 different mechanisms of action.
Ninety-eight next-generation biotherapeutics — defined as cell, gene, and nucleotide therapies — are also under clinical investigation and leverage 18 different approaches. The combined immunotherapies and next-generation biotherapeutics are targeting almost all cancer tumor types with more than 80 mechanisms of action.
In recent years, record numbers of new oncology drugs have been approved, bringing new treatment options to patients — 15 in 2018 alone (see chart for more information). Additionally, strides have been made in improving cancer outcomes with the cancer death rate down 25% since its peak in 1991, making nearly one in 20 Americans a cancer survivor.
A key contributor in the improvements in survival is the acceleration of innovation in oncology therapies. Through this wave of new drug launches, the promise of cancer immunotherapy is coming to fruition and precision/personalized medicines are continuing to gain adoption.
“We’re in the midst of a tremendously productive period of time in terms of new cancer medicines for a range of tumors, leukemias, and lymphomas, says Michael Kleinrock, research director, IQVIA Institute for Human Data Science. “In the last five years, breakthroughs have evolved quickly."
The breadth and scale of oncology research is unprecedented and on myriad fronts, including immuno-oncology drugs, cell and gene therapies, and biomarker-specific therapies. There are exciting advancements in all of these areas, says Bruce Feinberg, D.O., VP, chief medical officer, Cardinal Health Specialty Solutions.
“There has been a steady annual decline in cancer mortality for the past two decades, most of which can be attributed to better treatments, rather than prevention or earlier diagnosis," he says.
“Targeted- and immuno-oncology agents are primary reasons for this dramatic improvement in survival. But while these classes of therapy can sometimes extend remissions for five years or more, they are rarely curative. Looking forward, cell and gene therapies may have a greater impact because they may have the potential to be curative."
 Genentech’s 2019 Oncology Trend Report supports this hypothesis, with most oncologists believing that technologies such as checkpoint inhibitors (95%), antibody drug conjugates (89%), and cell therapies (86%) will have a moderate to significant impact on oncology treatments in the next two to three years.
Genentech’s 2019 Oncology Trend Report supports this hypothesis, with most oncologists believing that technologies such as checkpoint inhibitors (95%), antibody drug conjugates (89%), and cell therapies (86%) will have a moderate to significant impact on oncology treatments in the next two to three years.
“The trend in oncology is toward specificity," says Edison Liu, M.D., president and CEO of The Jackson Laboratory. “Treatments are progressively being tuned for narrow indications defined by clinical, genetic, biochemical mechanism, and organ characteristics," he says. “At the beginning of cancer pharmacology, chemotherapy primarily targeted dividing cells, and any organ specificity came from trial and error. Now, treatments are crafted on the basis of more refined — and complex — biological mechanisms, or very specific molecular configurations. The new classes of RAS and receptor tyrosine kinase inhibitors that target very specific sequence mutations in RAS or EGFR, and cell therapeutics such as CAR-T therapies that target uniquely expressed antigens are examples of this trend. This biological specificity is at the heart of the personalized medicine movement in oncology."
Trends Driving Oncology Research
Seven IO drugs are currently commercially available: Yervoy (ipilimunab), Opdivo (nivolumab), Rituxan (rituximab), Blincyto (blinatumomab), Interleukin (aldesleukin), Gardisil (recombinant human papillomavirus vaccine), and Kymriah (tisagenlecleucel).
Jordan Clark, chief commercial officer, Diaceutics, says oncology research in 2020 will focus on next-generation IO, including neoantigens and custom cancer vaccines, new KRAS-targeted therapies, and tumor agnostic targets such as NTRK and RET gene fusions. “More research into the tumor microenvironment and the tailoring of combinations and timing of different types of IO therapies will have a significant impact on cancer patients in the future," Mr. Clark says.
“Meanwhile, biomarker pathways such as HRD versus single gene/proteins, namely BCRA, will improve patient outcomes — not just as part of a single line of therapy, but by providing a holistic view of the patient journey."
 Dr. Liu agrees there is still much to learn about oncology immune therapeutics and further advances will be profoundly important. “In many ways, we are at a similar stage as we were for oncogene targeted treatments in the late 1990s."
Dr. Liu agrees there is still much to learn about oncology immune therapeutics and further advances will be profoundly important. “In many ways, we are at a similar stage as we were for oncogene targeted treatments in the late 1990s."
Dr. Liu says understanding the dynamics of tumor evolution and how to eliminate dormant metastatic cancer cells will have the greatest impact on cancer patients.
“Tumor evolution is central to disease progression and the development of therapeutic resistance," he explains. “The process is genetically based and could be modeled if we knew the rules. We have the tools to understand this process in the form of single cell sequencing, advanced animal models of disease, and circulating tumor DNA sequencing. Tumor dormancy is the characteristic of a cancer cell to be quiescent, even for years, only to emerge as a proliferating metastatic tumor.
Understanding this process will be most important to finding approaches that block cancer relapse. Understanding tumor evolution and dormancy does not downgrade the importance of furthering cancer immunology or DNA repair research."
Dr. Clark predicts advancements will continue to be made in the development of cancer vaccines, such as autologous patient-derived immune cell vaccines, tumor antigen-expressing recombinant virus vaccines, and peptide vaccines.
Mr. Clark says big pharma companies, such as AstraZeneca, Novartis, and Roche, are doing great research in the areas of precision medicine and the use of biomarkers in oncology. Another interesting big pharma company is Lilly, which recently announced an oncology R&D initiative with biotech-like agility. The new company, named Loxo Oncology at Lilly, combines the Lilly Research Laboratories (LRL) oncology organization and Loxo Oncology, which was acquired by Lilly in early 2019.
“However, I believe much of the innovation comes from the smaller, single-platform/asset pharma companies that can dedicate efforts to R&D," he says. “We expect that in 2020, the FDA will approve more precision medicine drugs than one-size-fits-all therapies. This will have a colossal impact on patient care and outcomes, and will further drive the movement toward a personalized medicine approach and the need to integrate seamless biomarker testing at the time of launch for each new drug.
“In addition, there will be a general trend to move away from traditional Phase I, II, and III clinical trials, toward a basket/umbrella trial format with adaptive arms," Mr. Clark continues.
 “Increasingly, AI will be used in lab and clinical practices to aid both diagnosis and clinical decision-making. As more data is gathered, we will see more AI technologies integrated into routine clinical practice once the critical mass of credibility has been passed."
“Increasingly, AI will be used in lab and clinical practices to aid both diagnosis and clinical decision-making. As more data is gathered, we will see more AI technologies integrated into routine clinical practice once the critical mass of credibility has been passed."
Precision Medicine and Biomarkers
Our experts agree precision medicines with associated biomarker-driven strategies are driving oncology research bolstered by a variety of clinical and economic factors.
“One of these factors is the ability to improve outcomes through more precise medicines," Dr. Feinberg says. “As we learn more about the genetic underpinnings of cancer, conditions such as lung cancer, which was once considered one disease, has now been segmented into a collection of ultra-orphan conditions, each focused on unique biomarkers or genetic mutations. While these R&D efforts address highly targeted populations, in many cases the clinical outcomes justify the significant investment."
Biomarkers are powerful tools to prescreen populations to achieve better trials results and achieve better outcomes, and as a result more medicines reach the market faster, Mr. Kleinrock says.
“There are two dynamics in the oncology marketplace that are likely to play out: the development of cancer drugs that work for more people and cancer drugs that work for specific, small patient populations," he says. “Biomarkers don’t necessarily narrow indications for a therapy."
Three cancer drugs — Rozlytrek, Keytruda, and Vitrakvi — have now been approved as tumor-agnostic therapies.
Tumor-agnostic therapies use drugs or other substances to treat cancer based on the cancer’s genetic and molecular features without regard to the cancer type or where the cancer started in the body. Tumor-agnostic therapies uses the same drug to treat all cancer types that have the genetic mutation or biomarker that is targeted by the drug.
In 2019, Rozlytrek (entrectinib) was approved as a treatment for adult and adolescent patients whose cancers have the specific genetic defect, NTRK (neurotrophic tyrosine receptor kinase) gene fusion and for whom there are no effective treatments. Keytruda was approved for tumors with microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) tumors in 2017 and Vitrakvi for NTRK gene fusion tumors in 2018.
Across a number of major tumor types, patient treatment protocols are being based on biomarkers rather than by tumor type alone. “Essentially, the FDA is approving drugs that work with a biomarker test result regardless of where in the body the tumor is," Mr. Kleinrock says.
The global precision oncology market is on track to reach $141 billion by 2026, according to BIS Research.
Yet, biomarker adoption still has a way to go. According to Novartis’ most recent Precision Oncology Annual Trend Report, decision-maker confidence in personal oncology biomarker knowledge is variable: 68% of oncologists and 44% of pathologists reported high confidence vs. only 25% of payers. Oncologists reported that the most important factors influencing oncology biomarker use are the predictive power of the test (66% strongly agree), the test is recommended in clinical pathways and/or guidelines (64%), and the companion diagnostic is mandated in the FDA labeling (52%).
The Novartis survey found payers consider the clinical validity of the oncology biomarker test the most important factor driving coverage decisions (72% of payers strongly agree). This is followed by several important considerations, including the oncology biomarker is mandated in a FDA labeling (60%), test results must change patient management (60%), test performance (58%), clinical utility (58%), and inclusion in third-party guidelines (56%).
In 2018, the Centers for Medicare and Medicaid Services (CMS) finalized a nationwide determination to advance innovative personalized medicine for Medicare patients with cancer. This news means that more cancer patients will have access to diagnostic laboratory tests using next-generation sequencing (NGS), which can lead to more informed treatment decisions and better outcomes. Additionally, with this decision comes the hope that more tests and therapies may be approved in the near future.
Opinions on NGS/GSP (glycated serum protein) coverage policies vary widely among payer study participants, as they did not strongly agree with any one strategy. Almost half of all payers surveyed (40%) by Novartis strongly agree that they have already or are likely to adopt the CMS decision to automatically cover FDA-approved NGS companion diagnostics.
Biomarkers that stratify patients likely to respond to therapy are currently included in 39% of oncology trials, an increase of 25% from 2010, a reflection that precision medicine approaches are becoming more commonplace. Modeling the impact of current clinical development trends on future productivity, the availability of pools of prescreened patients and biomarker tests could yield improvements of as much as 104% and 71%, respectively, by 2023, predicts IQVIA.
Oncology Research: High-Risk
Oncology research is a high-risk endeavor. Despite increased levels of pipeline activity, oncology still remains one of the most challenging areas for research and development, fraught with significant failure risks and long timelines. Barriers to new drug adoption also remain, delaying patients from benefiting from treatment advances.
The oncology composite success rate fell to 8.0% in 2018 from 11.7% in 2017, similar to the average level of the past decade, according to IQVIA. And while clinical trial duration timelines still remain higher for oncology trials than other disease areas, this number has generally declined over the past five years, with a seven month reduction in Phase I trials, 11 months in Phase II trials, and more than a year in Phase III trials.
Clinical trial complexity — measured as a combination of endpoints, eligibility criteria, and numbers of subjects, trial sites and countries — has increased sharply for Phase I trials over the past five years. The overall productivity of oncology trials — measured as success rates relative to trial effort (complexity and duration) — has improved by 22% since 2010, but remains far lower than trials for other therapy areas.
“The cumulative success rate — 8% from Phase I through to market — for oncology drugs remains really low," Mr. Kleinrock says. “Fewer than 1 in 10 oncology therapies make it to the market."
“The current failure rate of about 90% from Phase I to approval of a new drug is lowered by about three times when a robust biomarker-driven strategy is used," Mr. Clark says. “Loxo’s Vitrakvi went from preclinical trials to approval in record time. Another recent example is the BEAT AML umbrella trial."
The BEAT AML study, sponsored by The Leukemia & Lymphoma Society, demonstrated the feasibility of using NGS to assign treatment tailored to individual genomics of elderly patients with acute myeloid leukemia within seven days. This may prove to be a major advance, since typically the wait for NGS results is up to one month, and by that time, the disease can rapidly progress.
“Cancer trials have some of the most complex criteria for eligibility and for management, for a number of valid clinical reasons; every cancer patient is a little bit different," Mr. Kleinrock says.
Dr. Liu says issues of trial complexity and risk will have to be addressed with an open mind toward radically different approaches, including novel financing of expensive cancer drugs, more public/private partnerships, and coordinated centers for innovation.
“These suggestions have several things in common: coordination, discipline, novelty, and resources," he says “Better coordination between the for-profit pharmaceutical and nonprofit academic and research communities would create significant value for patients. It should be clear that, today, good drugs cannot be developed and distributed without private companies and access to capital. It is also obvious that research talent in the universities and research institutes are a wellspring of innovation."
New oncology drugs launched in 2018 took a median of 10.5 years from the time of first patent filing to regulatory approval and launch, four years shorter compared with 2017. Even as progress is being made in accelerating the time it takes for scientific advances to reach cancer patients, barriers remain in the areas of registration, diagnostics, infrastructure, and reimbursement, resulting in variability in care and delays in patients benefiting from treatment advances.
Getting Oncology Therapies to Patients
The healthcare community has continued to grapple with managing the costs and complexity of delivering cancer care to more than 1.7 million newly diagnosed cancer patients every year in the United States.
Spending on all medicines used in the treatment of patients with cancer reached almost $150 billion in 2018 up 12.9% for the year and marking the fifth consecutive year of a double-digit increase, according to IQVIA. This increase was driven entirely by therapeutics, which grew 15.9%, while supportive care drugs declined 1.5% in 2018. New brands launched in the past two years and protected brand volume contributed nearly all the positive growth in major developed markets, where spending increases exceeded 13% in each market with the exception of Japan.
Cost is a concern for physicians, payers, and patients. The average annual cost of new medicines continues to trend upward, although the median cost dropped $13,000 in 2018 to $149,000, and cost per product ranged between $90,000 $300,000, according to IQVIA. The mean cost for new oncology brands in 2018 was $175,578, down from $209,406 in 2017, but was above the $143,574 mean from 2012 to 2018. Spending on cancer medicines is heavily concentrated, with the top 38 drugs accounting for 80% of total spending. More than half of cancer drugs generate greater than $143.6 million in annual sales and in aggregate account for only 2.2% of oncology spending.
Growth in cancer therapeutic spending of 11% to 14% is expected on a CAGR basis over the next five years, bringing the total market to between $200 billion and $230 billion, according to IQVIA.
Including supportive care, which is expected to decline by 3% to 6%, overall oncology spending will reach $220 billion to $250 billion, growing 9% to 12% through 2023.
One of the concerns is that the high cost of treatment could cause patients to delay or discontinue therapy. In fact, according to Genentech’s 2019 Oncology Trend Report, 19% of patients did just  that. The survey also notes that oncology practices estimate that a slightly higher percentage of patients are applying for patient-assistance programs via drug manufacturers — 32% on average of patients this year versus 27% last year. Among patients that sought support from assistance programs, 50% received support (vs. 47% last year). Among those seeking support from external foundations, 46% of patients who applied received support, up from 38% last year.
that. The survey also notes that oncology practices estimate that a slightly higher percentage of patients are applying for patient-assistance programs via drug manufacturers — 32% on average of patients this year versus 27% last year. Among patients that sought support from assistance programs, 50% received support (vs. 47% last year). Among those seeking support from external foundations, 46% of patients who applied received support, up from 38% last year.
Coverage decisions are increasingly being integrated across pharmacy and medical benefits.
Managed care organizations view real-world evidence (RWE) as important for coverage decisions; while they are concerned about site-of-care shifts to hospitals and academic center settings increasing costs, they are excited about the potential for more data collection. Genentech’s report finds oncologists are more comfortable evaluating surrogate endpoints, with about half as many managed care organizations expressing confidence in their readiness to evaluate gold-standard endpoints, for example overall survival. MCOs view real-world data (RWD) and patient-reported outcomes as important for determining coverage, though many consider RWD to have limited usefulness due to a lack of methodological rigor. Oncologists are divided on the matter of prescribing drugs in the absence of traditional clinical evidence and, overall, are less comfortable to do so compared with 2017 responses.
“With the passage of the 21st Century Cures Act, policymakers have been pushing for more RWE in regulatory submissions," Dr. Feinberg says. “This is important because clinical trial data, which serves as the foundation for most clinical decisions, provides a limited, and often not representative, assessment of a treatment’s efficacy and toxicity. In oncology, only 3% of all adult patients participate in clinical trials — and they tend to be younger, healthier, and less diverse than patients who don’t participate. To understand the other 97%, we need to rely on RWE. In just the past six months, five oncology drugs have gained FDA approval with a submission dataset that included RWE, and that trend is expected to continue."
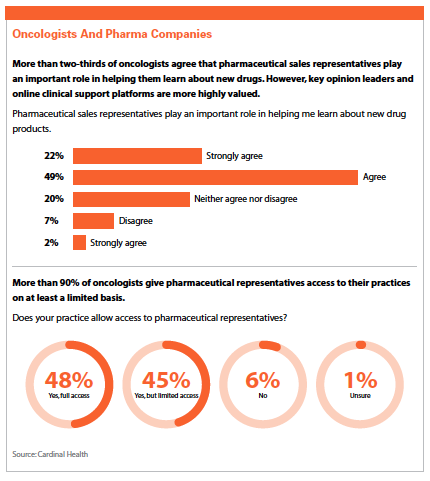
According to a recent survey by Cardinal Health, oncologists want to receive more real-world evidence from pharmaceutical companies, particularly patient outcomes and comparative effectiveness studies.
Cardinal Health surveyed oncologists to understand their attitudes about RWE and the role this plays in supporting their decisions about patient care. At the time of the survey in 2017, their responses indicated a mix of growing recognition of the importance of RWE, balanced with some skepticism and reluctance to incorporate RWE into their own decision-making process.
In the two years since that report, a growing consensus has been building around the importance of RWE in everything from regulatory submissions to reimbursement strategies. The FDA has openly advocated for the use of more RWE in regulatory submissions and in 2019 issued guidance to sponsors to clarify and simplify the process for including RWE in regulatory applications. At the same time, as value-based reimbursement models such as the Oncology Care Model have become more widely adopted, providers are increasingly being asked to provide RWE data to demonstrate patient outcomes.
 In Cardinal Health’s most recent survey, more than two-thirds of participating oncologists said RWE is necessary to inform treatment decisions, consistent with the results of the 2017 survey.
In Cardinal Health’s most recent survey, more than two-thirds of participating oncologists said RWE is necessary to inform treatment decisions, consistent with the results of the 2017 survey.
In terms of prior authorizations, Genentech’s report finds although their use remains high, use of other utilization management tools such as peer-to-peer treatment review has decreased.
Genentech also found that the majority of MCOs — 59% — are integrating coverage decision-making across pharmacy and medical benefits and the vast majority (74%) of employers are now integrating reports for medical and pharmacy benefits, an increase compared with 67% in 2017.
MCO use of guidelines (when making both medical and pharmacy benefit decisions) continues to grow, with the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology being the most used guidelines among MCOs. In addition to using guidelines to develop policy, the majority of MCOs involve oncologists; however, compared with last year, fewer MCOs are using specialists for assistance in coverage decisions, with the exception of genetic testing and counseling.
Genentech’s survey also found increased use of value-based incentives and payments for pathway adherence. In fact, 22% of MCOs already have outcomes-based payments, while an additional 30% would like to have them and have the capabilities to do so. Overall, 90% are interested in having them, but 38% lack the capabilities to do so.
“In the oncology market, the concept of paying for value and negotiating based on value is something we’ve been seeing more of over the past decade and across major developed markets," Mr. Kleinrock says. “We expect to see more of it in the future."
A recent survey of payers by Trinity found that novel contracting agreements may not be enough to address the high costs of cell and gene therapies. Trinity found interest in such innovative arrangements does not exist beyond national payers — for example, United, Anthem, Aetna, Cigna, and Humana — and even among these payers interest is limited for cell and gene therapies.
In fact, no payers within Trinity’s sample had, or were building, functional capabilities to enact such contracting agreements with manufacturers. Annuity- and outcomes-based payment models have generated much attention, yet the majority of payers preferred to pay a single lump sum due to the small treated patient populations and low resulting impact to a plan’s budget.
Trinity’s interviews found annualized payment models garnered little enthusiasm among payers; nine of 10 interviewed medical directors flatly rejected this option. Payers realize that annualized models will not discount cell and gene therapies’ costs and thus won’t impact their bottom line.
Medical directors are not interested in paying for a patient’s therapy after the person has died or left the plan. This contracting would require universal coverage decisions and unrealistic coordination between plans that would include the sharing of sensitive information with direct competitors.(PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
World Cancer Day: Feb. 4, 2020
World Cancer Day is the one singular initiative under which the entire world can unite together in the fight against the global cancer epidemic. World Cancer Day aims to save millions of preventable deaths each year by raising awareness and education about cancer, and pressing governments and individuals across the world to take action against the disease.
2020 marks the midway point of the three-year “I Am and I Will" campaign. I Am and I Will is an empowering call-to-action urging for personal commitment and represents the power of individual action taken now to impact the future.
This year alone, almost 9.6 million people will die of cancer, and left unchecked, the number of deaths will increase to 13.2 million per year by 2030. Many of these deaths can be avoided with increased governmental support and funding for prevention, detection, and treatment programs. Unless urgent action is taken to raise awareness about cancer and develop practical strategies to address the disease, millions of people will continue to die.
Sadly, the greatest increase in cancer is projected to hit low- and middle-income countries, those least equipped to cope with the social and economic impact of the disease.
There is an urgent need for action to raise awareness of the disease and to develop practical strategies to reduce the burden of cancer. World Cancer Day is the perfect opportunity to spread the word and create a global awareness of the disease in international media and global health and development programs.
For more information, visit https://www.worldcancerday.org.
To engage in the social media campaign, use the hashtag: #IamAndIWill.
~~~~~~~~~~~~~~~~~~~~~~~~~
Trends Driving Change in Oncology Clinical Development
Digital health technologies are expected to enable trial participants to receive rapid physician support for adverse events thus improving patient safety, decreasing attrition, and extending life.
Increased focus on patient reported outcomes (PROs) will shed new light on patient outcomes (PROMs) and experience (PREMs) outside the clinical setting or at home, as well as track performance status, to inform ongoing clinical decisions, serve as secondary endpoints, influence labeling, and accelerate trial times.
Real-world data is expected to speed trials by aiding in investigator/site selection, help optimize trial design, including right-sizing trials for treatment effect, and enable new trial designs.
Predictive analytics and AI will identify new clinical hypotheses to test, reduce trial design risks, speed enrollment by identifying protocol-ready patients, and help narrow trial patient populations to predefined subgroups (i.e., precision medicine).
Shifts in drug types include the development of targeted therapies, immunotherapies, cancer vaccines, oncolytic viruses, bispecific monoclonal antibodies, next-generation
biotherapeutics, and combinations, that will improve efficacy and success rates overall and lead to trials for new indications lacking current options.
Increased availability and ease of biomarker testing will help enrollment of patient populations, increasing study efficiency and enable tissue agnostic approvals such as anti-PD-1 for microsatellite instability-high tumors.
Availability of pools of prescreened patients and direct-to-patient recruitment will facilitate trial recruitment and help sites/trials hit accrual targets. As providers/vendors conduct more diagnostic tests and record demographics and prior treatment, trials targeting defined patient subsets will find it easier to recruit.
Changes in the regulatory landscape will encourage the use of biomarkers and further precision medicine, drive use of novel trial designs and endpoints, and may minimize work burden through the use of risk-based monitoring, electronic records, and electronic signatures. It will also speed drug approvals in oncology by increasing the use of real-world data to expedite drug development, especially for drugs pursuing an unmet medical need indication.


















