 Researchers and drug developers are gaining a better understanding of the immune system and how to better target cancer; as a result, immuno-oncology continues to be a key area of anticancer drug development.
Researchers and drug developers are gaining a better understanding of the immune system and how to better target cancer; as a result, immuno-oncology continues to be a key area of anticancer drug development.
Researchers over the last few years have found that the immune system has the capability to detect and destroy abnormal cells, but cancer cells are sometimes able to avoid detection and destruction. For example, cancer cells may reduce the expression of tumor antigens on their surface, making it harder for the immune system to detect them. They may express proteins on their surface that lead to immune cell to be inactivated. Or they may lead cells surrounding the tumor to release substances that suppress immune responses.
The advancing field of cancer immunology has produced several therapies for treating cancer that increase the strength of immune responses against tumors. There are several areas of research, including checkpoint inhibitors, chimeric antigen receptor t-cell therapies (CAR Ts), and novel therapeutic vaccines.
Checkpoint inhibitors have gained the most attention with Merck’s Keytruda and Bristol-Myers Squibb’s Opdivo being the leading  products available in this category. The most recent approved checkpoint inhibitor is Genentech’s Tecentriq, approved in October 2016 to treat metastatic non-small cell lung cancer and metastatic urothelial cancer.
products available in this category. The most recent approved checkpoint inhibitor is Genentech’s Tecentriq, approved in October 2016 to treat metastatic non-small cell lung cancer and metastatic urothelial cancer.
Recent research has shown that cancer cells often use immune checkpoint molecules to suppress an immune system attack.
Checkpoint inhibitors block these normal proteins on cancer cells, enabling the T-cells that respond to them.
According to BioPharm Insight, Opdivo, which received its first approval in December 2014, is forecast to have global sales of more than $12 billion by 2022. Keytruda is predicted to have global sales of $7.7 billion during the same year. Keytruda’s first approval was in September 2014.
The cancer immunotherapies space is expected to grow to $75.8 billion by 2022, representing a CAGR of 23.9%, according to business intelligence provider GBI Research.Established immunotherapies, particularly Revlimid, Opdivo, Keytruda, and Gazyva, are set to witness rapid sales growth.
BioPharm Insight, however, expects more modest sales, with the entire immuno-oncology market generating peak sales of $35 billion by 2022.
Experts agree that immune therapies will be an important part of the oncology treatment arsenal in the future. They say the next  generation of immune therapeutics — CAR Ts, bispecific antibodies, therapeutic vaccines, and antibody drug conjugates (ADCs) — will be will be the focus on continued research, as will the research of combination immune therapies.
generation of immune therapeutics — CAR Ts, bispecific antibodies, therapeutic vaccines, and antibody drug conjugates (ADCs) — will be will be the focus on continued research, as will the research of combination immune therapies.
“Immunotherapy will become the backbone of all cancer treatment regimens," says Robert Petit, Ph.D., chief science officer, Advaxis.
In the next five years, Dr. Petit says researchers will look at combinations of immunotherapies, as well as where immunotherapies fit in best with other treatment regimens, such as radiation, chemotherapy, and targeted agents.
“Oncologists have a lot of tools in the toolbox, and they all have been shown to be effective in some form," he says. “Now, immunotherapy has given us a whole new class of tools to add to the toolbox, and we need to figure out the smartest way to use them all together to benefit patients."
Richard Gregory, Ph.D., executive VP and chief scientific officer, ImmunoGen, anticipates that there will be many more combination studies involving drug conjugates and immunotherapies in general.
“Almost all oncology drugs are used in combination, and sponsors are working to define what the best combinations are to deliver optimal benefit for patients at an acceptable risk."
Immune Oncology Research Challenges
GBI’s latest report states that there are currently 2,037 products in active development in the cancer immunotherapy pipeline, equating to 37% of the entire oncology pipeline, which is strong reflection of the interest in this class of therapy.
About one-third of this research is in either therapeutic cancer vaccines or checkpoint inhibitors, according to Thomson Reuters’ Cortellis Clinical Trials Intelligence report. Although therapeutic cancer vaccine trials have steadily trended downward in the past five years, particularly in the noncommercial space, checkpoint inhibitors experienced a 25-fold increase in the number of commercially relevant trials started in 2015 as compared with 2010.
GBI analysts point out that a significant proportion of the immunotherapy pipeline consists of early-stage products.
Trying to harness the immune system to fight cancer is more complicated than first thought and severe side effects that can lead to death have been seen recently in clinical trials.
In November 2016, two patients in Juno Therapeutics’ trial of JCAR015 for adult patients with relapsed or refractory B-cell acute lymphoblastic leukemia died from severe brain swelling. This was the second time Juno’s trials were stopped. In the summer of 2016, three patients died as part of the same trial.
Also in November, Bristol-Myers Squibb announced that two patients in a trial assessing a combined Opdivo and Yervoy regimen died from myocarditis, inflammation of the heart muscle.
 Because checkpoint inhibitors fundamentally block a mechanism of immune tolerance across the entire body of the patient, there is potential for side effects that resemble autoimmune symptoms, Dr. Petit says.
Because checkpoint inhibitors fundamentally block a mechanism of immune tolerance across the entire body of the patient, there is potential for side effects that resemble autoimmune symptoms, Dr. Petit says.
Phil Gotwals, Ph.D., executive director of exploratory immuno-oncology, Novartis, says cytokine release syndrome — where T-cells release huge amounts of chemicals that can lead to side effects — is an issue developers are aware of and are working to manage the toxicities, just as they would in any development program.
He says checkpoint inhibitors act by re-engaging the immune system and that’s not specific to the cancer cell.
“This reaction can happen in peripheral tissues," Dr. Gotwals says. “This result hasn’t been unexpected. The question is whether there would be a therapeutic window and there does seem to be that. Checkpoint inhibitors, including the anti-PD-1, anti-PD-L1, and anti-CTLA-4 inhibitors can generate autoimmune-like toxicities, which also is not unexpected given their mechanism of action. These side effect will be managed the way toxicities are managed in any development program."
Additionally, treating late-stage cancers with immune therapies can be challenging, Dr. Petit says.
“As the cancer continues to progress, the immune system of the patient can become weaker and tolerance of the cancer more entrenched," he says. “So it’s really difficult for individual immunotherapies to catch hold in later stages of rapidly growing cancers. Immunotherapies rely on the patient’s immune cells to fight cancer so they can take more time to work than chemotherapy. Single immunotherapies tend to be more effective in earlier disease, where the cancer is less entrenched and the patient’s immune system is stronger, but there can be exceptions."
Jeff Abbey, CEO of Argos Therapeutics, says it’s important for developers to be smart about clinical trial design.
“A lot of companies are combining different approaches without necessarily thinking through what makes sense," he says. “It’s important to understand the mechanism of action of the different agents that are available and conduct the preclinical work to make sure the different therapies are compatible, and, if not synergistic, at least additive."
Andrew Zupnick, Ph.D., VP, oncology division, Novella Clinical, says the increase in clinical research of immunotherapies has created an ironic challenge.
 “Developers are facing a landscape that is increasing in complexity and shifting rapidly," he says. “For example, it is becoming more difficult to design a protocol for a specific indication, particular one that includes an investigational drug in combination with a checkpoint inhibitor due to the pace of new approvals and how quickly the standard of care is changing. For example, consider the recent surprise sBLA filing by Merck to market pembrolizumab in combination with chemo in first-line NSCLC, regardless of PD-L1 status; with multiple checkpoint inhibitors vying for market dominance. Which should one pick in designing a forward-looking protocol?"
“Developers are facing a landscape that is increasing in complexity and shifting rapidly," he says. “For example, it is becoming more difficult to design a protocol for a specific indication, particular one that includes an investigational drug in combination with a checkpoint inhibitor due to the pace of new approvals and how quickly the standard of care is changing. For example, consider the recent surprise sBLA filing by Merck to market pembrolizumab in combination with chemo in first-line NSCLC, regardless of PD-L1 status; with multiple checkpoint inhibitors vying for market dominance. Which should one pick in designing a forward-looking protocol?"
Dr. Zupnick suggests developers create options that are open-ended and remain flexible given the unknowns and remain open to protocol amendments based on shifts in the landscape.
“We just don’t know how things may evolve, and of course there are always new competitive entries coming into the marketplace to consider," he says.
Another challenge is that protocols involving immunotherapies have increased in complexity over the last two years.
“Protocols in early drug development look for different means to determine an optimal dose, such as target engagement or immune reactions instead of classical measures of toxicity, and are building in different combinations, multiple arms to test a range of options at once," Dr. Zupnick says. “‘Basket’ and ‘mono/combo’ designs are becoming a more common protocol design. This is when a range of patients with different indications are enrolled based on a given genetic profile, or the investigational agent is tested as a monotherapy and then in combination with an immunotherapy all in the same protocol. From there, the monotherapy and combination can have expansion arms to test multiple indications at the same time, each looking at different indications and lines of therapy, which can bring a substantial level of complexity to the trial’s implementation."
Selected Current Pharma Research
Novartis has a portfolio of immunotherapies that includes several approaches, including CAR T technology, checkpoint inhibitors, STING (Stimulator of Interferon Genes) pathway, T-cell engaging bispecific antibody programs, and myeloid cell targeting agents.
In the CAR T area, Novartis’ CTL019 is being developed to treat relapsed/refractory pediatric and young adult patients with B-cell acute lymphoblastic leukemia. CTL019, licensed from the University of Pennsylvania, is directed specifically against what are called CD19-expressing B cell hematological malignancies.
Dr. Gotwals says this involves a patient’s own T-cells being engineered to engage the tumor. The cells are infused back into the patient to seek out the tumor.
A global Phase II trial of CTL019 found that 82% (41 of 50) of patients achieved complete remission or complete remission with incomplete blood count recovery. This pivotal data, as well as data from a U.S. multicenter trial, will be the basis of the first global CAR T-cell trial and the basis of a biologics licensing application (BLA) to the FDA in early 2017. The company plans to file with the European Medicines Agency (EMA) later in 2017. The investigational therapy received a PRIME (PRIority MEdicines) designation from the EMA earlier this year.
Novartis also has a collaboration with Xencor that uses the bispecific antibody approach where antibodies are directed against the tumor and also against the T-cell. In June 2016, Novartis received the right to develop four additional bispecific antibodies and to use other Xencor proprietary antibody engineering technology for up to 10 additional biotherapeutic programs across the Novartis R&D portfolio.
In addition, the companies will collaborate to co-develop Xencor’s two bispecific T-cell engaging antibodies targeting CD3xCD123 and CD3xCD20 for the treatment of acute myeloid leukemia and B-cell malignancies. According to Dr. Gotwals, these programs are just entering the clinic.
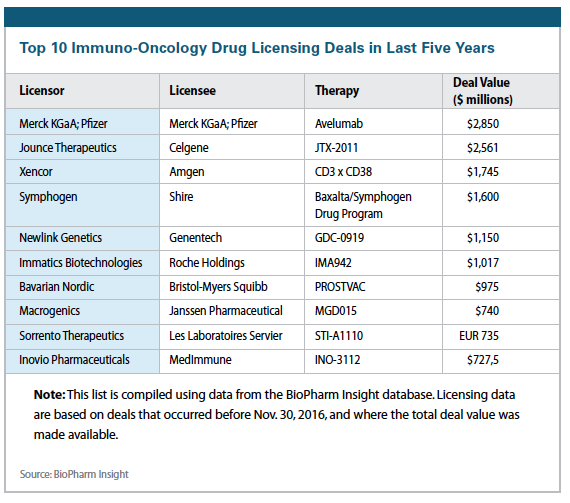 In March 2015, Novartis entered into a collaboration with Aduro focused on the discovery and development of next-generation cancer immunotherapies targeting the STING pathway. Aduro’s novel small molecule cyclic dinucleotides (CDNs) have shown in preclinical models to generate an immune response that specifically attacks tumor cells. This is in Phase I trials.
In March 2015, Novartis entered into a collaboration with Aduro focused on the discovery and development of next-generation cancer immunotherapies targeting the STING pathway. Aduro’s novel small molecule cyclic dinucleotides (CDNs) have shown in preclinical models to generate an immune response that specifically attacks tumor cells. This is in Phase I trials.
“The most exciting program to induce an innate immune response we have going on right now is the STING agonist," Dr. Gotwals says. “These are small molecules that are directly injected into a lesion that we hope will drive a very robust innate immune response. Direct stimulation of the immune system in a tumor cell is a very active field of research. This has the potential to turn cold tumors, that is tumors that do not have an active immune infiltrate, into so-called hot tumors or ones that do have an infiltrate. Our STING program is aimed in that therapeutic area."
ImmunoGen is pursuing multiple combinations for its lead program, mirvetuximab soravtansine, including PARP inhibitors and immuno-oncology drugs such as Merck’s Keytruda and expand into earlier lines of treatment, increasing the addressable patient population.
Mirvetuximab soravtansine is a first-in-class folate receptor alpha (FR)-targeting antibody drug conjugate. It is in a Phase III trial as a single agent treatment for platinum-resistant ovarian cancer.
“We develop antibodies that specifically recognize those proteins on the surface of tumor cells and then use them to introduce a drug into the tumor cell to kill it," Dr. Gregory says.
The technology behind the product is expected to have broad applicability.
Mirvetuximab also is being assessed in combination regimens with Keytruda, an immune checkpoint inhibitor, as well as Doxil, carboplatin, and Avastin for both platinum-resistant and platinum-sensitive ovarian cancer in the Phase Ib/II FORWARD II trial.
Preclinical data demonstrate the potential for enhanced activity when combining mirvetuximab soravtansine with immune checkpoint inhibition.
“In the combination studies, my major concern would be inadvertent immune responses against non-tumor tissues that could occur because of the checkpoint inhibitor and the lack of specificity," Dr. Gregory says.
Vaccines and Oncolytic Viruses
Two burgeoning areas of cutting-edge clinical oncology research include vaccines and oncolytic viruses.
Thus far only one cancer vaccine and one cancer virus have been approved by the FDA. In 2010, Dendreon’s Provenge, a cancer vaccine, was given marketing clearance to treat prostate cancer. In October 2015, Amgen received approval for Imlygic (talimogene laherparepvec), the first genetically modified oncolytic viral therapy. Imlygic is for the local treatment of unresectable cutaneous, subcutaneous, and nodal lesions in patients with melanoma recurrent after initial surgery. Designed to replicate within tumors and produce an immunostimulatory protein, cGM-CSF, Imlygic releases tumor-derived antigens that may promote an anti-tumor immune response.
In addition, companies such as Advaxis and Argos Therapeutics are exploring vaccines to treat cancer.
Advaxis’ lead immunotherapy, axalimogene filolisbac, or Axal, uses the company’s Lm Technology, and is in Phase III clinical trials for three potential indications: invasive cervical cancer, head and neck cancer, and anal cancer. The company initiated a Phase III trial in 2016 in late-stage cancer patients whose current treatments have failed for recurrent refractory cervical cancer, and the company is initiating a second Phase III trial in patients with metastatic cervical cancer.
Advaxis’ Lm Technology uses live, attenuated bioengineered Listeria monocytogenes (Lm) bacteria to stimulate the immune system to view tumor cells as potentially bacterial-infected cells and target them for elimination.
Researchers start with a live, attenuated strain of Listeria, then add multiple copies of a plasmid that encodes a fusion protein sequence, including a fragment of the LLO (listeriolysin O) molecule joined to the antigen of interest. This fusion protein is secreted by the Listeria inside antigen-presenting cells, which has been shown to result in a stimulation of both the innate and adaptive arms of the immune system. The LLO fragment in the fusion protein has been shown to reduce tumor defense mechanisms, making it easier for the immune system to attack and destroy the cancer cells.
“We give these live, weakened bacteria to patients through an IV infusion and the patient’s immune system gobbles them up greedily the same way it does when any bacteria is present," he says. “In the process of responding to what the immune system perceives as a potential infection from this bacteria, it picks up the tumor antigen target as well and starts hunting down cells that express the tumor antigen target along with any cells that have potential signs of this bacteria."
Dr. Petit says side effects associated with the infusion appear to be mild, transient, and flu-like with no off-target toxicities.
The FDA has granted axalimogene filolisbac orphan drug designation in each of indications being studied, as well as a Special Protocol Assessment for the Phase III AIM2CERV trial in cervical cancer, and fast track designation. Axalimogene filolisbacis currently being evaluated in eight clinical trials for HPV-associated cancers.
Additionally, Advaxis has a global collaboration with Amgen to develop ADXS-NEO under its MINE (My Immunotherapy Neo-Epitopes) program, which is designed to activate a patient’s immune system to respond against the unique mutation-associated targets, or neoepitopes, contained in each individual patient’s tumor identified through massively parallel DNA sequencing. ADXS-NEO is directed at patient-specific mutations. The company recently joined the Parker Institute for Immunotherapy and the Cancer Research Institute to further the research of neoepitope-based immunotherapies.
Argos Therapeutics is developing rocapuldencel-T, an autologous dendritic cell-based immunotherapy for metastatic renal cell carcinoma (mRCC), which is in a pivotal Phase III trial with 462 patients. The company’s platform technology, Arcelis, is a precision immunotherapy technology that captures the spectrum of mutated and variant antigens that are specific to each patient’s individual disease.
Mr. Abbey says the therapy involves taking a sample of the patient’s tumor and isolating the messenger RNA. Argos then uses a proprietary method to program the patient’s own matured immune cells, called dendritic cells, with messenger RNA isolated from the patient’s tumor. These activated dendritic cells are then put back into the patient by intradermal injection.
“The result is that our product induces a patient-specific, tumor-specific immune response directed at the patient’s tumor and only at the patient’s tumor," Mr. Abbey says. “All of the patients who we’re treating still have metastatic disease; our product generates T-cells that kill the tumors that remain. Because it’s individualized for each patient, rocapuldencel-T captures all of the cancer mutations, which is key to treating that patient."
A Phase II trial included 21 patients with advanced, metastatic kidney cancer who had their primary tumor removed by surgery and then were treated with standard of care targeted therapy combined with Argos’ therapy. In the 21 patients, the median overall survival was 30 months. Mr. Abbey points out that with the standard of care in the particular patient population, median survival was only expected to be 15 months.
Mr. Abbey says a number of patients in the Phase II trial have survived more than five years and a couple of patients are now out past seven years.
“For those patients, we do a second white blood cell collection and then produce more T-cells using the same RNA," he says. “Two patients are still in remission — approaching eight years — using the exact same RNA with new white blood cells."
He says the company anticipates top-line survial data from the Phase III trial later this year, and expects to file rocapuldencel-T with regulatory authorities in the first half of 2018.
In preparation for longer-term commercial uptake, the company is building a new 125,000-square-foot manufacturing facility in North Carolina that will be able to manufacture the company’s therapy for up to 10,000 patients a year.
At launch, Argos will use its current facility, which can be expanded to manufacture for up to 2,400 patients a year.
The Argos therapy is also being studied in investigator-initiated Phase II to treat non-small cell lung cancer and bladder cancer. (PV)


















