A New Way to Treat Severe Asthma
Dr. Bing Yao, Respiratory, Inflammation & Autoimmunity Head at MedImmune, talks about the company’s efforts to develop a next-generation asthma therapy.
 Severe, uncontrolled asthma is a debilitating and potentially fatal form of the disease. Uncontrolled asthma can lead to a dependence on oral corticosteroids, which have serious and irreversible adverse effects, including osteoporosis, anxiety, depression, weight gain, glaucoma, and diabetes.
Severe, uncontrolled asthma is a debilitating and potentially fatal form of the disease. Uncontrolled asthma can lead to a dependence on oral corticosteroids, which have serious and irreversible adverse effects, including osteoporosis, anxiety, depression, weight gain, glaucoma, and diabetes.
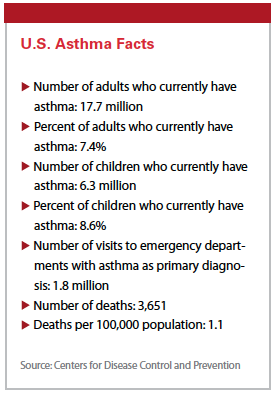
Asthma currently affects the health and day-to-day lifestyles of 315 million individuals worldwide, and by 2020 the numbers are expected to increase to as many as 400 million people. Up to 10% of asthma cases are severe, of which about 40% remain uncontrolled on the current standard of care medicine.
MedImmune, AstraZeneca’s global biologics research and development arm, is researching an alternative to currently available monoclonal antibodies. Benralizumab has a unique way of working in patients with severe asthma. It targets eosinophils, white blood cells that are a key trigger in inflammatory respiratory diseases in about half of all asthma patients.
“What we and others have found is the higher the eosinophil counts in the blood, the more severe the disease," says Dr. Bing Yao, respiratory, inflammation and autoimmunity head at MedImmune. “Around half of severe asthma patients have elevated eosinophils."
Benralizumab is an antibody that targets a receptor on the membrane of the eosinophils. Benralizumab was engineered with a special property to this antibody that, once it binds to the receptor of the eosinophils, will bring in another cell type — a Natural Killer cell — essentially removing the eosinophils.
Benralizumab is the first biologic to target the interleukin-5 (IL-5) receptor, unlike GlaxoSmithKline’s 2015-approved Nucala (mepolizumab) and Teva’s 2016-approved Cinqair (reslizumab), which target the IL-5 ligand.
“Targeting the IL-5 receptor results in a more complete depletion of the eosinophils," Dr. Yao says.
 Phase III studies show benralizumab has led to a significant decrease in the frequency of asthma exacerbations, cutting the rate of exacerbations by a third to a half compared with placebo among people with the most severe form of asthma.
Phase III studies show benralizumab has led to a significant decrease in the frequency of asthma exacerbations, cutting the rate of exacerbations by a third to a half compared with placebo among people with the most severe form of asthma.
Data also suggest that benralizumab’s mechanism of action results in a faster onset of action than current monoclonal antibodies. A Phase I/II study showed that benralizumab is able to rapidly deplete eosinophils, with an onset of action within 24 hours.
Dr. Yao says the clinical studies have shown efficacy across different parameters. There was exacerbation reduction, the primary endpoint, and there was improvement in lung function.
“We’ve also seen a reduction of the symptoms,“ he says. “The dosing frequency — our antibody has the potential for every eight weeks — has good efficacy, and the antibody also is well tolerated."
Dr. Yao says the initial indication for benralizumab is expected to be as an add-on therapy for severe eosinophilic asthma.
The company plans to submit a BLA to regulatory authorities by the end of 2016, Dr. Yao says.
Benralizumab is also in development (Phase III) for the treatment of COPD. The company expects to file with regulatory authorities in 2018 for this indication.
AstraZeneca and MedImmune are committed to the respiratory area, with a strategy focused on targeting subsets of the asthma market.
“By targeting patient subsets, we have a better chance of success and can provide patients the maximum benefit from therapies," Dr. Yao say. “We call it calibrating to meet the needs of the patients."
Another AstraZeneca/MedImmume pipeline product for severe asthma is tralokinumab, which is in Phase III development. Tralokinumab is an anti-interleukin-13 (IL-13).
Another product, tezepelumab, which is Phase II, is a monoclonal antibody that targets a molecule called TSLP, which Mr. Yao says sits upstream in the disease pathway. He says allergens, viruses, or even industry pollution can trigger the production of TSLP in lung epithelium. TSLP activates multiple pathways that are involved in asthma induction, persistence, and flares. By targeting the upstream TSLP with Tezepelumab, it may have efficacy in broader patient population. (PV)










