For smaller companies, the ability to find funding and form alliances is the key to continuing research on innovative new medicines.
For other companies, venture capital plays a huge role in financing. But some companies are also looking at alternative ways to fund their research, including public-private partnerships, grants, and risk-sharing arrangements with academia.
Emerging companies have a robust pipeline, with more than 3,400 drug indication programs under development, based on recent analysis by the Biotechnology Innovation Organization of the BioMedTracker database. This accounts for a full 69% of the entire global industry pipeline. Roughly 43% of these programs involve partnerships, demonstrating the importance of licensing and alliance development in the industry.
 In 2015, $10.1 billion was invested in 783 deals in the life-sciences industry, making the life-sciences industry the second-most invested destination after software, in terms of dollars, according to the MoneyTree Report from PricewaterhouseCoopers (PwC) and the National Venture Capital Association (NVCA). For the full year, deal volume increased by 12% compared with 2014, despite a 3% decline in deal activity, resulting in an increase in average deal size by 15%.
In 2015, $10.1 billion was invested in 783 deals in the life-sciences industry, making the life-sciences industry the second-most invested destination after software, in terms of dollars, according to the MoneyTree Report from PricewaterhouseCoopers (PwC) and the National Venture Capital Association (NVCA). For the full year, deal volume increased by 12% compared with 2014, despite a 3% decline in deal activity, resulting in an increase in average deal size by 15%.
More than half of all deals in 2015 went to seed and early-stage companies, with more than 1,400 companies raising venture capital for the first time.
U.S. venture capital funding declined 27% by value and 15% by volume, on a year-over-year basis during the fourth quarter of 2015. Uncertain global economic conditions made investors wary, say executives at PwC. This impacted IPOs as well as venture capital investments during the fourth quarter.
Public-Private Partnerships
Experts say greater cooperation with academic and government researchers, as well as with other companies, is needed to create collaborations that push innovation forward. There is a growing consensus that the industry must team up with public, private, and government organizations to create collaborations to bring about new and innovative therapies. There is also a growing movement toward open collaboration and pre-competitive agreements with the public sector and even with other companies.
Partnerships with academic institutions and the public sector have always played a significant role in research. In fact, successful private-public partnerships have led to private sector growth and innovation, job creation, and a strengthened national economy. Public-private partnerships are not new; what is new is a focus on precompetitive research.
In the past, public-private partnerships were largely a funding vehicle. Now, the partnerships that are successful have a genuine recognition of the strength of all parties.
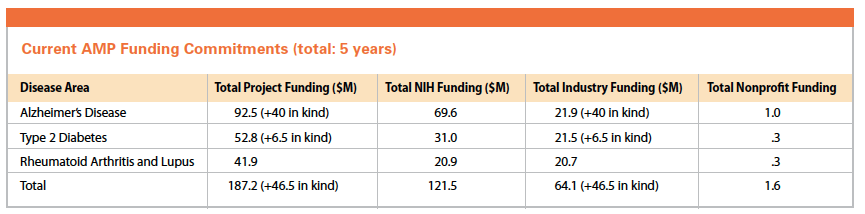
One such public-private partnership is the Accelerating Medicines Partnership (AMP), which was launched in February 2014. The initiative is a venture between the National Institutes of Health, 10 biopharmaceutical companies — AbbVie, Biogen, Bristol-Myers Squibb, GlaxoSmithKline, Johnson & Johnson, Lilly, Merck, Pfizer, Sanofi, and Takeda — and several nonprofit organizations to jointly identify and validate promising biological targets of disease.
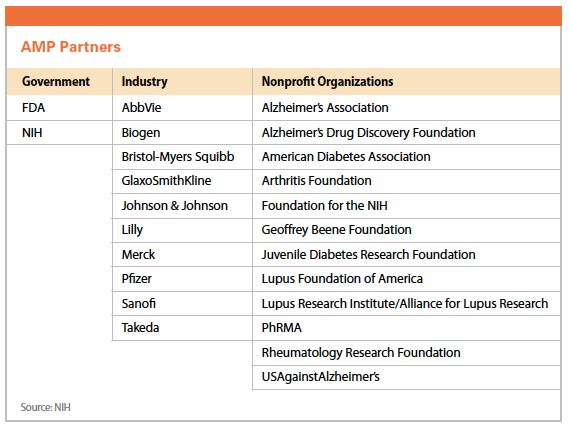 Through this partnership, which is managed through the Foundation for the NIH (FNIH), NIH and industry partners are contributing a total of $230 million over five years.
Through this partnership, which is managed through the Foundation for the NIH (FNIH), NIH and industry partners are contributing a total of $230 million over five years.
AMP has begun three-to-five year pilot projects in three disease areas: Alzheimer’s disease, type 2 diabetes, and autoimmune disorders of rheumatoid arthritis and lupus.
Another important public-private partnership is the Discovering New Therapeutic Uses for Existing Molecules pilot through the NIH’s National Center for Advancing Translational Sciences. This program, launched in May 2012, matches researchers with a selection of molecular compounds from the industry to test ideas for new therapeutic uses, with the goal of identifying promising new treatments for patients.
Eight companies — AbbVie, AstraZeneca, Bristol-Myers Squibb, Lilly, GlaxoSmithKline, Janssen Research & Development, Pfizer, and Sanofi — provided 58 compounds for the pilot program.
In Europe, the Innovative Medicines Initiative (IMI), Europe’s largest public-private initiative, has a budget of €3.3 billion for the period 2014 to 2024. IMI was launched in 2008 and currently has more than 50 projects, with more in the pipeline. Some focus on specific health issues such as neurological conditions (Alzheimer’s disease, schizophrenia, depression, chronic pain, and autism), diabetes, lung disease, oncology, inflammation and infection, tuberculosis, and obesity. Others focus on broader challenges in drug development such as safety, knowledge management, the sustainability of chemical drug production, the use of stem cells for drug discovery, drug behavior in the body, the creation of a European platform to discover novel medicines, and antimicrobial resistance.
In Japan, the government created the GHIT Fund, an international nonprofit funded jointly by the Japanese government, Japanese pharmaceutical companies, Bill & Melinda Gates Foundation, Wellcome Trust, and United Nations Development Program. GHIT promotes the development of drugs, vaccines, and diagnostics as part of the fight against infectious disease primarily prevalent in low- and middle-income countries.
In the GHIT Fund’s first year, FY 2013, it invested more than $17.5 million in more than 20 development partnerships between Japanese and international research institutions. In the last two years, it has spent $43 million to develop innovative tools for fighting diseases worldwide.
Funding Innovation
 One company with nontraditional financing is Oncoceutics, a clinical-stage biopharmaceutical company engaged in the discovery and development of highly differentiated treatments for difficult cancers. The company receives non-dilutive grants and private investments and has alliances and collaboration with academic centers around the world to help fund its research, as well as grants through the National Cancer Institute to fund a family of compounds.
One company with nontraditional financing is Oncoceutics, a clinical-stage biopharmaceutical company engaged in the discovery and development of highly differentiated treatments for difficult cancers. The company receives non-dilutive grants and private investments and has alliances and collaboration with academic centers around the world to help fund its research, as well as grants through the National Cancer Institute to fund a family of compounds.
Additionally, the company has a unique risk-sharing alliance agreement with the MD Anderson Cancer Center. MD Anderson is conducting two clinical trials at a no-cash cost in exchange for some restricted exclusivity on clinical development. MD Anderson is the primary site for trials for certain indications for a period of time and will receive some financial reward upon FDA approval of the drug.
Denmark-based LEO Pharma, a well-established pharmaceutical company with a centered focus on skin care conditions, has announced a $65 million international initiative to take its already successful operation and put an effort into beyond-the-pill solutions.
LEO has established a separate unit, the LEO Innovation Lab, which will build and test applications, wearables, and digital platforms with a specific focus on people living with psoriasis. Some of the areas they are looking to address, which medication-only treatment overlooks are diet, as well as mental health and social well-being.
The Innovation Lab is looking to collaborate with other partners in the organization’s hub cities, which are London, Copenhagen, Paris, and Toronto. According to the company, it is willing to invest and give grants to startups that it believes will set the agenda for the future of healthcare in this area.
“With LEO Innovation Lab, the company is doubling down on innovating beyond the pill products and services," explains Catalina Cernica, head of Innovation Lab UK. “We can build and iterate solutions at speeds unimaginable until now. The development cycle for each minimum viable product is limited to 100 days, which will include design, prototype and feedback from the people using them." (PV)