 As new drug launches proliferate in the increasingly complex specialty therapeutic space, biopharma is finding that success is often pre-determined by actions that take place very early in the development and commercialization cycle. In fact, the earlier companies start thinking through their launch strategy, the better. In particular, emerging companies that are pouring in millions of dollars into clinical research and development simply cannot afford a false start. In many cases commercial input is often overlooked. A vital driver of success is the quality and depth of interactions with key influencers including clinicians, patients and equally as important, payers. Done right, this preparation can help a new medicine outperform at market launch as well as facilitate prompt uptake by healthcare professionals and payers.
As new drug launches proliferate in the increasingly complex specialty therapeutic space, biopharma is finding that success is often pre-determined by actions that take place very early in the development and commercialization cycle. In fact, the earlier companies start thinking through their launch strategy, the better. In particular, emerging companies that are pouring in millions of dollars into clinical research and development simply cannot afford a false start. In many cases commercial input is often overlooked. A vital driver of success is the quality and depth of interactions with key influencers including clinicians, patients and equally as important, payers. Done right, this preparation can help a new medicine outperform at market launch as well as facilitate prompt uptake by healthcare professionals and payers.
Since cracking the human genome code, life-science companies have been working on innovative ways to apply genomics to drug development for personalized medicines. Remarkably, personalized medicines now represent some 42% of drugs in the pipeline, according to a survey by the Tufts Center for the Study of Drug Development1. While highly specialized drugs produce better outcomes for patients, they present a tougher challenge for marketers, which must be considered early on.
Personalized drugs also require a fundamentally different pricing model. Companies’ commercial teams must convince both payers and healthcare providers that their product not only produces better outcomes but also reduces the cost of treatment. The education and messaging required to communicate the value of precision medicines is significantly more complicated. Further, the market landscape today is incredibly diverse, replacing the traditional physician relationship with a vast, interconnected network of stakeholders who can each influence product success differently.
 A recent benchmarking survey by Quintiles aimed to understand how companies are integrating the payer voice into clinical development, and then validated these insights with payers. Three key themes emerged — the need to secure leadership buy-in for reimbursement and regulatory approval success; to develop and maintain transparent internal processes; and to link incentives to the financial success of products.
A recent benchmarking survey by Quintiles aimed to understand how companies are integrating the payer voice into clinical development, and then validated these insights with payers. Three key themes emerged — the need to secure leadership buy-in for reimbursement and regulatory approval success; to develop and maintain transparent internal processes; and to link incentives to the financial success of products.
Secure Leadership Buy-in for Reimbursement and Regulatory Approval Success
Top performing manufacturers have switched to a business of creating drug products that are both reimbursable and approvable, not just ones that meet regulatory requirements. It is critical for companies to understand the potential financial downside of launching a poorly differentiated product that fails to demonstrate value to a broader set of stakeholders, particularly payers. As payer controls continue to increase, successful product development will require a cross-functional approach starting as early as pre-clinical research. For example, the manufacturers surveyed had established portfolio management boards chaired by representatives of R&D and commercial leadership. These boards helped manufacturers incorporate a blend of regulatory and reimbursement feedback into investment, approval, and strategic cross-therapeutic area milestone decisions to a greater degree than all other manufacturers.2
Develop and Maintain Transparent Internal Processes
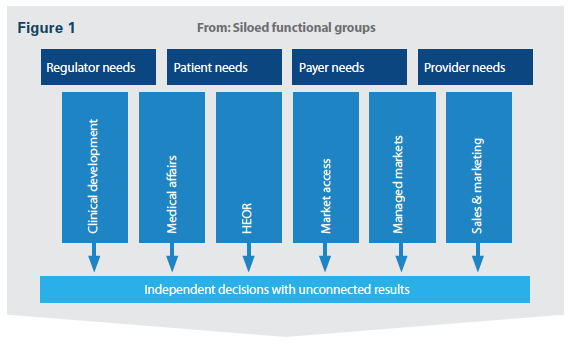
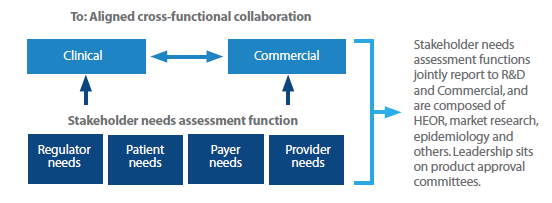
Early and integrated market access input is critical to evidence-based decisions and evidence-informed trials. Companies should move from the traditional, siloed functional groups addressing stakeholder needs separately, to an aligned system with cross-functional collaboration (Figure 1). New functions that have input to both clinical and commercial decisions can help improve cross-functional collaboration and set the stage to demonstrate value to broad sets of stakeholders.
 There is also a clear cross-functional need for better integration of commercial and market access input; the question is how to optimize this within current constraints. New paradigms that prioritize pricing and reimbursement first are continually evolving, but clinical investment should always be made in those products likely to achieve financial success due to addressing the unmet needs of patients, payers and providers.
There is also a clear cross-functional need for better integration of commercial and market access input; the question is how to optimize this within current constraints. New paradigms that prioritize pricing and reimbursement first are continually evolving, but clinical investment should always be made in those products likely to achieve financial success due to addressing the unmet needs of patients, payers and providers.
Understanding payer pressure points earlier in the clinical development process can ease access concerns at product launch.
Link Incentives to Financial Success of Products
Clinical and commercial teams together should be accountable for product approvability and reimbursement. All functional areas should hold some accountability for manufacturer profitability, but should also have a say in investment decisions. The survey indicates that top performing manufacturers base compensation and performance metrics on drug launch financial success, not just on regulatory approval milestones, as is often the case. The most successful approach involves an evolution from the typical incentive approach, where clinical and commercial incentives may not have been historically aligned, to linked functional area incentives. Grouped incentive structures align clinical and commercial teams to be incentivized on many similar product attributes; variable ‘weighting’ by therapeutic area or product class accounts for products where the payer voice does not have such a major influence at launch.
In the survey, all manufacturers recalled disputes and difficult decisions throughout development and launch; some believed that a strong global franchise group could help alleviate these disputes.
Simply put, plans must be refined over time. No single manufacturer has identified the “optimal" solution to aligning incentives across clinical and commercial teams.
Finally, simply commissioning market research studies early in clinical development is not enough. Clear incorporation of this insight into each development stage-gate is imperative, as this is an iterative process. Recent product launches confirm that payers are increasingly requiring significant rebates for access parity, with products that fail to add value over incumbents struggling to gain access.
Measure Twice, Cut Once
In this environment, companies must deliver tailored information to healthcare providers based on very specific patient use cases. The education of each of the healthcare stakeholders must be more detailed, widely accessible and bi-directional to support ongoing engagement. To start, biopharma companies should conduct far-reaching and innovative market research and use the results to inform a smart go-to-market strategy.
All key stakeholders should also be surveyed, including healthcare professionals both in group practices and in hospitals or healthcare systems, administrators, payers, patients, and those who influence regulatory approval. Alternative therapies from other companies should be analyzed as part of the mix. These collective insights will guide the development of the product’s value proposition and targeting strategy. All of this data will also enable companies to anticipate product uptake and support pricing negotiations with payers. All told, this will serve as the backbone for commercial success.
In conclusion, companies must recognize that the world has changed and adjust their commercialization plans accordingly by starting early and taking an integrated, holistic approach. A product launch plan must be comprehensive, extending beyond commercialization to consider manufacturing capabilities, clinical expectations, economic impact, and more. This launch planning process should start as early as possible, sometimes three years ahead of the anticipated launch enabling executives to take necessary time to “measure twice" (or thrice) and cut just once. Launch excellence — the culmination of an iterative process that unifies clinical and commercial teams along with key stakeholder objectives — is needed to deliver an optimized launch in an increasingly complex healthcare marketplace. (PV)
Editor’s Notes:
1. Source: www.phrma.org/sites/default/files/pdf/pmc-tufts-backgrounder.pdf
2. Source: Internal Quintiles expert interviews and manufacturer benchmarking studies
Quintiles has supported more than 220 product launches in 20 countries since 2009. Guided by your priorities, we help you drive successful commercialization with a complete lineup of services. From upfront strategic planning to tactical execution, we can accurately assess the gaps in your capabilities, then rapidly fill them with highly skilled marketers, medics, regulatory experts and sales leaders.
Benefits of a commercialization partnership with Quintiles include:
Retaining full strategic control over your product.
Keeping internal resources focused on your core portfolio.
Maximizing the value of your product to your company.
Retaining the flexibility for future business opportunities.
Reduced operational risk.


















