 It’s been 16 years since tragedy struck in the clinical research world. A patient in an experimental gene therapy trial died just days after receiving the therapy. In 1999, Jesse Gelsinger came to the University of Pennsylvania with a rare metabolic disorder that prevents the body from breaking down ammonia. In this trial, a weakened adeno virus, a cold virus, was used as a vector to delivery a corrective gene. But after he received the therapy, Jesse’s body began to shut down and he died a few days later.
It’s been 16 years since tragedy struck in the clinical research world. A patient in an experimental gene therapy trial died just days after receiving the therapy. In 1999, Jesse Gelsinger came to the University of Pennsylvania with a rare metabolic disorder that prevents the body from breaking down ammonia. In this trial, a weakened adeno virus, a cold virus, was used as a vector to delivery a corrective gene. But after he received the therapy, Jesse’s body began to shut down and he died a few days later.
Jesse’s death sparked new regulatory controls in gene therapy trials. Early in 2000, the Food and Drug Administration and the National Institutes of Health enhanced patient protection through two new programs: the Gene Therapy Clinical Trial Monitoring Plan and the Gene Transfer Safety Symposia.
This death also had an impact on gene therapy research, with investors and larger companies pulling out of many projects. But academic researchers continued to look for safer vectors and new ways to address genetic diseases. And they began developing new technologies and platforms.
Now, gene therapy, which can be broadly defined as the transfer of defined genetic material to specific target cells of a patient for the ultimate purpose of preventing or altering a particular disease state, is a growing area of research that is again attracting investors interested in start-up companies.
 Large pharma companies are also getting into the arena, collaborating and conducting their own research.
Large pharma companies are also getting into the arena, collaborating and conducting their own research.
As of July 2015, more than 2,200 gene therapy trials were being conducted or completed worldwide, according to The Journal of Gene Medicine. The majority of these, 1,386, have been in the United States.
Venture firms have invested more money into gene therapies since 2010, $715.8 million, than they did all of last decade, $653.6 million, according to VentureSource, which is owned by Dow Jones & Co. Investors see the initial opportunity in neurological, vision, and blood disorders. Investments in gene therapies for the brain have amounted to $503.5 million since 1999, while about $224.8 million has been invested in blood-related gene therapies, and $211.6 million in gene therapies for the eyes.
In 2014, there was a dramatic increase in both the number and value of gene therapy deals when compared with historical levels, according to GlobalData. Gene therapy deals increased from 16 in 2013 to 36 in 2014, while total deal value soared from $122.8 million in 2013 to nearly $5 billion in 2014.
Additionally, GlobalData researchers say between 2014 and 2015, nearly $1.5 billion was raised in investment funding, which is indicative of renewed investor confidence and could lead to higher-than-expected deal activity for gene therapies in the future.
Part of this increase in investment dollars, according to Usman Azam, M.D., global head of the cell and gene unit at Novartis, is because the basic fundamental science has changed over the last 15 years.
“There is now an understanding of the implications, of how to manage genomic changes and gene editing, and there is a fundamental acceleration around viral vector platforms," he says. “This new understanding and knowledge are transformative to the industry."
 Olivier Danos, Ph.D., senior VP of gene therapy at Biogen, says in the past 15 years the research community has produced novel technologies that may not fully solve gene-based challenges, but in some cases they have made some situations retractable.
Olivier Danos, Ph.D., senior VP of gene therapy at Biogen, says in the past 15 years the research community has produced novel technologies that may not fully solve gene-based challenges, but in some cases they have made some situations retractable.
“Fifteen years ago, there were a lot of theories around gene-based therapies and the technology was not validated enough or even efficient enough to be able to translate the findings into the real world," he says.
“Today, some of these data can be translated. Hopefully in another 10 years or 15 years we’ll have a landscape of possible applications in gene therapy that will be even larger than what it is today."
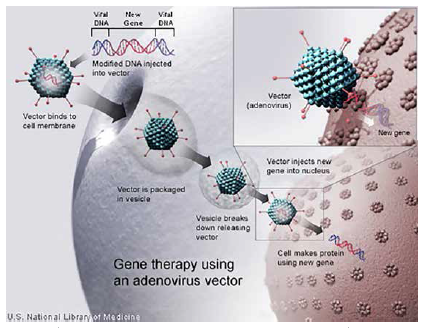
Gene therapy is a very complex way of bringing a therapy to the body.
“In essence, gene therapy involves using a virus as a vector, which can enter a cell, delivering a new functioning gene with the goal that the cell will start expressing proteins that before had been missing," explains Joern Aldag, CEO of uniQure.
 Mr. Aldag says what has changed over the last 15 years is the ability of researchers and scientists to manufacture safe and effective viral vectors. In the past, the manufacturing process wasn’t able to completely render the virus harmless, which created an immune reaction in the body.
Mr. Aldag says what has changed over the last 15 years is the ability of researchers and scientists to manufacture safe and effective viral vectors. In the past, the manufacturing process wasn’t able to completely render the virus harmless, which created an immune reaction in the body.
That was the case with Jesse, who experienced a severe immune reaction to the viral vector in the gene therapy.
“There were events that occurred with systemic gene therapy that frankly put the field back many years as a result of fatalities," Dr. Azam says. “Some of these events occurred at the University of Pennsylvania and it is nice to see that they are part of the renaissance again of this research."
Novartis is working with the University of Pennsylvania to create a Center for Advanced Cellular Therapeutics (CACT) on the Penn Medicine campus in Philadelphia. This will be the center of research using chimeric antigen receptor (CAR) technology, which enables a patient’s T cells to be reprogrammed outside of the body so when they are re-infused into the patient, the T cells have the ability to hunt and destroy cancer cells.
The CACT, which will be funded in part through a $20 million investment from Novartis, will be devoted to the discovery, development, and manufacturing of these personalized cellular cancer therapies, through a joint research and development program led by scientists and clinicians from Penn and Novartis. The new facility is expected to be completed in 2016.
Dr. Azam says manufacturing has moved from a cottage industry into a more mature model of cell manufacturing.
“The ability to have GMP scale up of what is a very different model from small molecules and biologics has given both regulators and the industry confidence that these are viable for patients," he says. “There is confidence that this can be managed in a correct way and potentially introduce a new business model."
Research Leads to New Therapies
Scientists carried on trying to find a vector that would not trigger the immune system. They began looking seriously at adeno-associated viruses, which didn’t appear to trigger the immune system.
It is one of these adeno-associated viruses that was used in the first gene therapy — Glybera — which received regulatory approval in Europe in 2012. Glybera has not yet been commercially used in Europe.
uniQure’s commercialization partner Chiesi Farmaceutici is in the process of launch but Mr. Aldag expects it to be available later this year in Germany as its first market. In the United States, uniQure is working with the FDA and will likely do an additional trial for its U.S. submission.
Mr. Aldag says Glybera is a single intervention with a long-term effect.
“Patients were dosed with Glybera six years ago and we have demonstrated that it is still in effect," he says.
Glybera is indicated for the treatment of adult patients diagnosed with familial lipoprotein lipase deficiency and suffering from severe or multiple pancreatitis attacks.
“If there is a gene mutation in the muscle cells, then the protein it produces is a nonsense protein that doesn’t perform its function of processing fat in the muscle," he says. “The consequence is that excess fat travels through the blood stream and obstructs capillaries in the pancreas. This leads to pancreatitis."
 Glybera provides a healthy gene into muscles so that these muscle cells can then express the lipoprotein lipase function properly. The mechanism gets repaired and the person can have significantly less risk of getting pancreatitis.
Glybera provides a healthy gene into muscles so that these muscle cells can then express the lipoprotein lipase function properly. The mechanism gets repaired and the person can have significantly less risk of getting pancreatitis.
GlobalData analysts believe the approval of Glybera laid the foundation for increased business development in the gene therapy field, including deals activity, and new financing from investors.
“There is now a broad level of support for adeno-associated virus therapies that can deliver the correct functioning gene to multiple target tissue," says Tim Miller, Ph.D., CEO, Abeona Therapeutics. “The key with gene therapy is matching the right delivery vehicle for the targeted tissue. 2016 is set up to be a very exciting year with multiple gene therapy companies reporting clinical data. Next year, we’ll be talking about significant insights into the promise of multiple approaches to gene therapies in targeting tissues and correcting underlying disease, many of which have no standard of care treatment."
Abeona’s lead program is an adeno-associated virus-based gene therapy for Sanfilippo syndrome (MPS IIIA and IIIB), , a metabolism disorder. Trials for Sanfilippo types A and B are expected to begin toward the end of 2015 in the United States and in Europe in 2016.
The company recently licensed an AAV-based gene therapy program in juvenile Batten disease, a fatal autosomal recessive neurodegenerative disorder, from the University of Nebraska Medical Center, as well as an AAV-based gene therapy program for Fanconi anemia disorder, a rare blood disorder. Dr. Miller anticipates the company will begin clinical trials in the second half of 2016 for the juvenile Batten program.
Biogen is another company working with adeno-associated vectors. In July, Biogen entered in an agreement with AGTC to develop gene-based therapies for multiple ophthalmic diseases.
The collaboration focuses on the development of a portfolio of AGTC’s therapeutic programs, including both a clinical-stage candidate and a preclinical candidate for orphan diseases of the retina that can lead to blindness in children and adults.
Biogen has made an up-front payment of $124 million to AGTC, which includes a $30 million equity investment in AGTC.
The lead development programs in the collaboration, both of which use an adeno-associated vector, include a clinical candidate for X-linked retinoschisis, a disease affecting boys during the teenage years and that can lead to serious complications such as vitreous hemorrhage or retinal detachment during adulthood.
Another program is a preclinical candidate for the treatment of X-linked retinitis pigmentosa, which usually causes night blindness by the age of 10 and progresses to legal blindness by the time a person is in his or her early 40s.
“These are rare genetic diseases of the eye," Dr. Danos says. “Potentially, we will be able bring therapies to patients who have absolutely no treatment available. Based on what we’ve learned in our previous research and preclinical studies, with gene transfer we can restore vision."
Immunotherapies with Viral Vectors
Researchers from Kalorama Information says gene therapies for cancer are ideal and potentially much more effective for certain cancer types when using a targeted approach and recruiting the patient’s own immune system.
Kalorama estimates a $100 billion market for cancer treatments, which the firm says is fueling new solutions. Gene therapies for cancer that have progressed through clinical trials predominantly use a two-pronged approach in attacking cancer: the use of engineered viruses and the introduction of genetic material that enables immune system recognition of tumor cells.
One such company using this approach is Novartis. The company has a number of therapies within its chimeric antigen receptor (CAR) T cell therapy program. The lead product is CTL019, which is in an ongoing Phase II study in pediatric patients with relapsed/relapsed acute lymphoblastic leukemia. The product has received breakthrough therapy designation from the FDA, which provides expedited review of new medicines that treat serious or life-threatening conditions.
The University of Pennsylvania is also conducting a Phase II clinical study of CTL019 in the treatment of hard-to-treat non-Hodgkin lymphoma. The study is being conducted by the University of Pennsylvania’s Perelman School of Medicine in adults with large B-cell lymphoma and follicular lymphoma.
This treatment works by extracting the leukocytes, the white blood cells, from a patient. Then the T cells are harvested and reprogrammed through an eight- to 10-day process at Novartis’ Morris Plains, N.J., facility. During this process, a viral vector that has been genetically modified is introduced to the T cells and they begin to express a CAR T molecule known as CTL019. CTL019 can hone in on a cancer tumor biomarker known as CD19, which is expressed on B cells.
“In effect what we are doing is reprograming the patients natural T cells to become hunter cells and multiply in the patient to destroy the cancer," Dr. Azam says. “These are potentially curative therapies, which is different from other types of therapies, such as a small molecule or biologic. We believe these are going to be a definitive pillar for the practice of medicine over the coming years."
Novartis is working toward filing CTL019 with the FDA in 2016 for the treatment of pediatric acute lymphoblastic leukemia with a potential launch in 2017.
Dr. Azam says in the pediatric indication, they have seen more than a 90% complete response rates for patients given this therapy over three years. In the lymphoma arena, 55% to 60% of patients are achieving a complete response and in certain subtypes of lymphomas there is close to 100% response.
This therapy, however, is not without risks. The main side effect is the release of cytokines.
“When we see cytokine, a toxin, release occur, it can be a marker that the body is fighting," Dr. Azam says.
“The trick here is to ensure that we don’t let the cytokine release get out of hand. There are medications that can be given to help manage these patients in terms of their cytokine release."
Novartis also has a program with Regenerex using the company’s facilitating cell therapy (FCRx) platform in transplants. The program aims to use the donor’s cells to help the person receiving an organ be more receptive, hopefully avoiding the use of immunosuppressants, which can lead kidney damage. So far, 24 people have been treated as part of the program and Dr. Azam says the company is scaling up for larger studies next year.
Gene Editing Programs
Gene editing technologies aim to use “molecular scissors" to make a cut in the genome and then DNA can be inserted, removed, or replaced. There are currently four families of engineered nucleases being used: zinc finger nucleases (ZFNs), transcription Activator-like effector nucleases (TALENs), the CRISPR-Cas system, and engineered meganuclease re-engineered homing endonucleases.
Zinc fingers are the most well-known. In order to create a double-strand break at a specific location, two individual ZFNs are introduced into the cell at the same time and they each bind to opposite strands of the DNA to cause the cut.
The CRISPR-Cas9 system is the newest genome editing approach and it uses a protein-RNA complex that has been designed to recognize a particular DNA sequence. It enables surgically efficient knock-out, knock-down, or selective editing of defective genes and has the potential to allow safer, more precise gene modification.
Biogen has a collaboration with Sangamo BioSciences in the area of zinc finger nucleases related to hemoglobinopathies, targeting sickle cell disease and beta-thalassemia. The beta-thalassemia program was initiated with a BCL11a knockout strategy, and the sickle cell program already employs the BCL11A enhancer approach.
Both beta-thalassemia and sickle cell manifest several months after birth, when patients’ cells switch from producing functional fetal globin to a mutant form of adult beta-globin, which causes their condition. The development program uses ZFN-mediated genome editing of a patient’s own hematopoietic stem and progenitor cells (HSPCs) to increase production of fetal globin in cells that will ultimately become red blood cells.
Sangamo and Biogen have developed two related but distinct ZFN-mediated genome editing approaches to disrupt critical aspects of the regulatory pathway that, in early infancy, lead to the switch in production from fetal to adult globin.
Initially, the companies developed a strategy for beta-thalassemia that specifically knocked out the gene encoding for the BCL11A transcription factor, a critical regulator of the switch from fetal to adult globin production.
A second approach was initiated for the sickle cell program, which involved the disruption of the more recently described erythroid-specific enhancer of BCL11A expression, a regulatory DNA sequence in the genome that is essential for expression of BCL11A but that is functional exclusively in cells destined to become RBCs.
Dr. Danos says clinical trials for both programs are expected to begin in 2016.
Novartis also is doing research in gene editing. The company has a collaboration with Intellia Therapeutics and Caribou Biosciences to explore making medicines and drug discovery tools with CRISPR genome editing technology.
Research and development activities will focus on using CRISPR ex vivo for engineering chimeric antigen receptor T cells and hematopoietic stem cells (HSCs).
The collaboration and licensing agreement with Caribou is focused on using Caribou’s foundational CRISPR platform and intellectual property as a research tool for drug discovery.
Under the terms of the agreement with Intellia, Novartis receives rights to develop all collaboration programs focused on engineered CARs and the right to develop an undisclosed number of targets for ex vivo editing of HSCs. In addition, Novartis receives non-exclusive rights for limited in vivo therapeutic applications of CRISPR systems.
“There is still much work to be done, and it is very early days, but there is a lot of excitement around the CRISPR-Cas9," Dr. Azam says. “We still have to prove in clinical settings how these gene modifying products and platforms can work."
Abeona is another company using the CRISPR-Cas9 gene editing technology, albeit with a unique approach targeted at rare blood diseases. The company is working on Fanconi anemia and rare blood diseases using the CRISPR-Cas9 gene editing system licensed from the University of Minnesota. The company has demonstrated proof of concept in mutiple models and is currently conducting IND enabling preclinical studies.
“Our particular approach to using that gene editing system works in vivo and goes directly into patient," Dr. Miller says. “Normally, for many of these strategies, it’s necessary to take cells out and purify them, then transduce them with a virus, and characterize them, and reinject them into the patient. Ex vivo approaches are time consuming and costly and only a certain population of cells are produced."
Gene Therapy is Not Just for Genetic Diseases
UniQure also has a collaboration with Bristol-Myers Squibb to develop 10 gene therapies in cardiovascular diseases.
BMS paid uniQure $140 million up front for technology access and equity, with the potential for up to $250 million per program if milestones are met during the development of the 10 programs.
“Gene therapy is not only good for genetic diseases; it’s also good for degenerative diseases where organs have failed performing certain functions," Mr. Aldag says. “For example, patients who have had a myocardial infarction struggle to deliver sufficient oxygen into the extremities of the body. The heart then receives signals to beat faster. Over time, a certain protein that is needed for the heart to contract properly becomes depleted. One way to get more oxygen flowing is to use a gene therapy to deliver the genes that express those proteins."
The collaboration with Bristol-Myers Squibb includes uniQure’s gene therapy program for congestive heart failure that is intended to restore the heart’s ability to synthesize S100A1, a calcium sensor and master regulator of heart function. Thus far, the program has been tested in animal studies in pigs. Myocardial infarction was induced and then the pigs were treated either with gene therapy or placebo. About 90% of pigs treated with a single injection of the therapy were alive after 12 months whereas 90% of the ones that got the placebo died. (PV)

















