CROs’ Use of eTMF Apps Increases: Veeva Survey
Trending Now: A majority of CROs cite speeding study start-up as top driver for eTMF technology adoption.
 A significant number of contract research organizations (CROs) are increasingly using electronic trial master file (eTMF) applications, according to a new survey by Veeva. The global study of TMF owners found that 38% of CROs surveyed now use eTMF applications versus just 21% in 2014. A number of benefits are prompting CROs’ adoption of eTMF technology. More than half (58%) say faster study start-up is a key driver, while 50% cite cost savings.
A significant number of contract research organizations (CROs) are increasingly using electronic trial master file (eTMF) applications, according to a new survey by Veeva. The global study of TMF owners found that 38% of CROs surveyed now use eTMF applications versus just 21% in 2014. A number of benefits are prompting CROs’ adoption of eTMF technology. More than half (58%) say faster study start-up is a key driver, while 50% cite cost savings.
CROs that have implemented an eTMF solution report a broad range of benefits impacting all aspects of the TMF. When it comes to inspection-readiness, particularly, most CROs see improvements in the number of missing documents (92%); misfiled documents (89%); duplicate documents (86%); incomplete documents and/or missing signatures (84%); and expired documents (81%). Additionally, more than half (57%) report improved audit- and inspection-readiness as a result of eTMF adoption.
“Indeed, eTMF applications are the key to fully capitalizing on the wealth of information available to 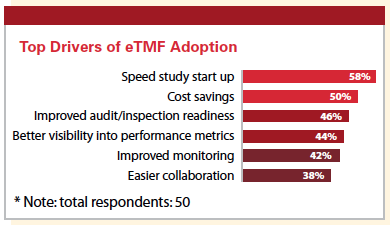 today’s clinical leaders," says Mike Burton, Veeva’s director of CRO alliances. “There is tremendous opportunity for improvement across clinical processes, but it’s often difficult to spot weak points without metrics. Advanced eTMF solutions, in contrast, can quickly reveal common bottlenecks and process inefficiencies to help speed study start-up and, ultimately, improve study quality."
today’s clinical leaders," says Mike Burton, Veeva’s director of CRO alliances. “There is tremendous opportunity for improvement across clinical processes, but it’s often difficult to spot weak points without metrics. Advanced eTMF solutions, in contrast, can quickly reveal common bottlenecks and process inefficiencies to help speed study start-up and, ultimately, improve study quality."
Reimbursement Biggest Challenge for Cancer Patients
 Lack of reimbursement for supportive care services topped the list of challenges facing cancer programs today, with 65% of programs naming this as their biggest challenge, despite the increase in communication with payers on the value of these services, according to the Association of Community Cancer Centers’ (ACCC) sixth annual Trends in Cancer Programs survey.
Lack of reimbursement for supportive care services topped the list of challenges facing cancer programs today, with 65% of programs naming this as their biggest challenge, despite the increase in communication with payers on the value of these services, according to the Association of Community Cancer Centers’ (ACCC) sixth annual Trends in Cancer Programs survey.
The number of patient-centered services provided has continued to grow, but the reimbursement necessary to provide these services is lagging. The majority of cancer programs now offer nurse navigation (89%), psychological counseling (88%) survivorship care (87%), and palliative care (87%).
This expansion of services may have contributed to the 61% of respondents who cited budget restrictions as their second biggest challenge.
“America’s cancer programs are increasingly being asked to do more with less, a trend we are seeing across healthcare," says ACCC President Steven D’Amato. “As these programs strive to provide more patient-centered services, knowing the positive effect these services have on improving patient health outcomes, reimbursement has become a bigger challenge."
Managed Markets Groups Manage Government Payers
In the life-sciences industry, managed markets groups at the majority of surveyed companies are the primary function responsible for communicating with government payers. According to pharmaceutical intelligence firm Cutting Edge Information, these groups are typically trained in discussing health economics data or have the flexibility to engage a medical science liaison (MSL) or health outcomes liaison (HOL).
 Managed markets groups at 57% of companies are tasked with communicating with government payers. MSLs are the primary means of communication at 14% of companies. Meanwhile, 29% of survey respondents indicated dedicated reimbursement or market access-related groups that have similar responsibilities to a managed markets group.
Managed markets groups at 57% of companies are tasked with communicating with government payers. MSLs are the primary means of communication at 14% of companies. Meanwhile, 29% of survey respondents indicated dedicated reimbursement or market access-related groups that have similar responsibilities to a managed markets group.
“An important aspect of government payer relationship management is frequent contact," says Jacob Presson, senior research analyst at Cutting Edge Information. “While managed markets teams typically lead this charge, other groups are often involved with getting products onto formularies."
Americans Want to Know What’s in Their DNA
More than half (53%) of U.S. consumers want to know what’s in their DNA, according to a new survey by health technology company Counsyl. Yet only 7% of respondents say their doctor has discussed genetic screening with them.
“The survey findings speak to the public’s appetite to know what’s in their DNA — something we’re encouraged to see, since early awareness of risk can make a big difference in health outcomes," says Ramji Srinivasan, co-founder and CEO of Counsyl. “There is still much to be done, though, in helping patients and physicians understand how advancements have made genetic screening cost-effective and results delivery easy to navigate, so that potentially life-changing information can be within everyone’s reach."
The survey, conducted by independent research company ORC International of 1,020 adults, explored knowledge of and attitudes toward genetic testing among adult consumers. The strong interest in genetic screening may point to an interest among consumers in using genetic screening to be more proactive about their healthcare. (PV)