 With a population of 38 million people and a healthy, growing economy, Poland is an attractive market for the pharma industry. This is bolstered by the fact that Poland is a member of the European Union, providing it with stability and a predictable regulatory environment.
With a population of 38 million people and a healthy, growing economy, Poland is an attractive market for the pharma industry. This is bolstered by the fact that Poland is a member of the European Union, providing it with stability and a predictable regulatory environment.
Poland underwent many political and economic changes in 1989, when it began to open up to international collaboration to change its economic and political systems. This was also the impetus for pharma collaboration, with Polish companies leveraging scientific and medical exchanges and joining international networks, says Piotr Kolataj, M.D., senior director, clinical operations EMEA East Sub-Region, country manager Poland, Parexel.
Poland is the largest pharma market in Central and Eastern Europe and the sixth largest in Europe. While the value of the pharmaceutical market in Poland lags behind that of most Western Europe nations, it is growing steadily, says Piotr Piotrowski, Ph.D., medical affairs director, KCR, an international CRO.
In 2014, IMS Health valued the Polish pharma market, including pharmacies and hospitals, at 28.7 billion Polish Zloty ($775.2 million).
A Pharma Advantage
Poland’s stability and bright outlook attract most global pharmaceutical companies, says Wojciech Przyby, M.D., VP of business operations and optimization, real-world and late phase research, Quintiles. He adds many pharmaceutical companies have their Central and Eastern European headquarters in Poland.
Reforms over the last 10 years have led to a more mature and transparent environment in healthcare, says Marynika Woroszylska-Sapieha, general manager of Sanofi Poland. Major changes were introduced in the pricing and reimbursement system, with a goal of increasing transparency, reducing drug spending, and providing higher access to innovations. The first of these goals has been achieved, but access to innovation can still be improved, Ms. Woroszylska-Sapieha says.
general manager of Sanofi Poland. Major changes were introduced in the pricing and reimbursement system, with a goal of increasing transparency, reducing drug spending, and providing higher access to innovations. The first of these goals has been achieved, but access to innovation can still be improved, Ms. Woroszylska-Sapieha says.
Magdalena Matusiak, manager, clinical development pharmacovigilance team lead, KCR, says Poland’s location makes it ideal for exporting products to Europe and Asia, its economy is stable, and there are many incentives to bolster the market.
“Authorities have created 14 Special Economic Zones to encourage and facilitate development of new companies and to offer tax incentives along with additional funds," Ms. Matusiak says.
Poland is also the largest beneficiary of European Union funds, offering new investors solid and reliable support in business development, she says. And as one of the fastest-growing sectors in Poland, biotech has easy access to government grants, EU funds, and tax incentives.
“According to an EY report, European Attractiveness Survey, published in 2014, Poland ranks third in all of Europe in terms of new jobs created thanks to direct foreign investment projects," Ms. Woroszylska-Sapieha says.
One of Poland’s strengths is its ability to compete with low-cost countries through stability, economic development, and by investing in potential candidates and professionals, says Antoni Jedrzejowski, M.D., project director, Parexel.
Dr. Piotrowski says Poland has a large number of highly qualified staff, who have broad experience in the pharma market.
“The country boasts a high number of people with university or doctorate degrees working for the pharmaceutical industry, including a significant proportion of M.D.s and pharmacists," he says.
“At Sanofi Poland, we work with universities and regularly run internship programs for graduates at the Rzeszów plant and our Warsaw office," Ms. Woroszylska-Sapieha says.
Poland also has a well-developed manufacturing infrastructure, logistics, and know-how dating from the communist era, as well as a very strong network of scientific institutes.
Foreign investors also recognize other advantages of the market, which include regulatory compliance (particularly with respect to managing intellectual property rights and compliance with quality procedures) and transparent terms of cooperation, Dr. Przybyś says.
Another thing that sets Poland apart is the growing number of business initiatives organized by young people, Ms. Woroszylska-Sapieha says. In December 2014, Forbes published a ranking in which Poland was among the countries with the largest start-up growth potential.
There is a particular focus among Polish pharmaceutical companies to develop new molecules, Dr. Kolataj says.
 “Government-supported infrastructure and effective regulations position the market well for the future," Dr. Kolataj says. “As such, I truly believe that Poland has the potential to develop innovative drugs with international CROs offering consulting and clinical services locally."
“Government-supported infrastructure and effective regulations position the market well for the future," Dr. Kolataj says. “As such, I truly believe that Poland has the potential to develop innovative drugs with international CROs offering consulting and clinical services locally."
In addition, Poland has demonstrated a growing readiness for consolidation, which makes it attractive for acquisitions and mergers, Dr. Przybyś says.
In the first half of 2012 there were more than 21,000 employees in the pharmaceutical production sector, and the estimated total employment rate in the pharmaceutical sector is 31,000 employees with 11,000 of them employed by innovative companies. Retail and wholesale pharmacies employ another 80,000 to 90,000 individuals, and there are about 4,000 employees in the biotech sector, according to a PwC report.
Pharma Activity
The Polish pharmaceutical market has an exceptionally high share of generics drugs, says Monika Stefanczyk, head pharmaceutical market analyst at PMR Ltd.
“The only countries in Central and Eastern Europe where the share is higher are Ukraine and Russia, but those countries do not have a modern reimbursement system," she says.
All in all, according to PMR estimates, the share of generics in the total pharma market (pharmacy and hospital), taking into account manufacturer prices, was about 70%, while in Romania it is 30% and in the Czech Republic, 40%. The reason is a very strict cost-containment reimbursement policy.
While the Polish pharmaceutical market has largely consisted of high-volume generics, a number of Polish companies have started R&D operations focused on novel molecules, innovative therapies, and biosimilar products, Ms. Matusiak says.
 In 2013, there were 191 companies in Poland conducting R&D in biotechnology, Ms. Matusiak says, quoting figures from the Central Office of Statistics, Poland.
In 2013, there were 191 companies in Poland conducting R&D in biotechnology, Ms. Matusiak says, quoting figures from the Central Office of Statistics, Poland.
Innovative pharmaceutical companies are consistently developing their operations in Poland, for example by transferring the manufacturing branch of their business to Poland, Ms. Woroszylska-Sapieha says. National firms are also continuously developing their industrial operations.
“The value of capital expenditure incurred by pharmaceutical companies with manufacturing operations in Poland is largely earmarked to create new production lines," she says. “Many pharmaceutical companies also contribute to the growth of the industrial infrastructure in Poland, for example by building logistics and distribution centers and shared-service centers in areas such as HR, finance, customer service, IT, and warehouses."
Sanofi Poland has a cutting-edge manufacturing plant in Rzeszów in the southeast part of the country. “Compared with Western European countries, we stand out with competitive manufacturing costs, e.g. low support service prices and investment costs," Ms. Woroszylska-Sapieha says. “We provide GMP-compliant manufacturing conditions and offer excellent quality products. What also sets our factory apart from other pharmaceutical plants is the attractive diversification of production. We make medicines, dietary supplements, and dermocosmetics, which is why we can quickly respond to the needs of the ever-more demanding market."
Ms. Woroszylska-Sapieha says Poland boasts a network of more than 110 scientific institutions with almost 3,000 researchers working in the field of biotechnology and molecular biology. Companies’ R&D expenditures in Poland have risen by several hundred percent over the last couple of years.
According to FDI Intelligence, Poland ranked fifth in 2010 on the global ranking list of biotechnology investment projects. In the coming years, further growth of the domestic biotechnology market is expected, stimulated by innovative research projects conducted by Polish biotechnology companies and research units as well as by the flow of foreign investment projects into this sector.
Clinical Trials
Clinical research studies started about 25 years ago, in the 1990s, in Poland, making it a mature and well-established market with a long history of studies conducted per GCP rules, Dr. Przybyś says.
“Polish clinical sites began to participate in international collaborative efforts through local pharma companies," Dr. Kolataj says.
An average of 40,000 patients a year take part in clinical trials, Ms. Woroszylska-Sapieha says.
The key driving force for the domestic clinical trials’ market is the population size, which at 38 million makes it the world’s 34th largest country in terms of population.
country in terms of population.
Poland is the largest clinical trials market in CEE (Central and Eastern Europe)/CIS (Commonwealth of Independent States) and accounts for roughly 20% of clinical trials in Eastern Europe, Dr. Kolataj says, adding that both patient participation and site penetration rates signal potential for growth.
More than 400 studies are registered in Poland every year. According to the Association for Good Clinical Practice in Poland, about 40,000 patients participate in clinical trials each year. There are more than 840 studies with open recruitment according to clinicaltrials.gov, Dr. Przyby says.
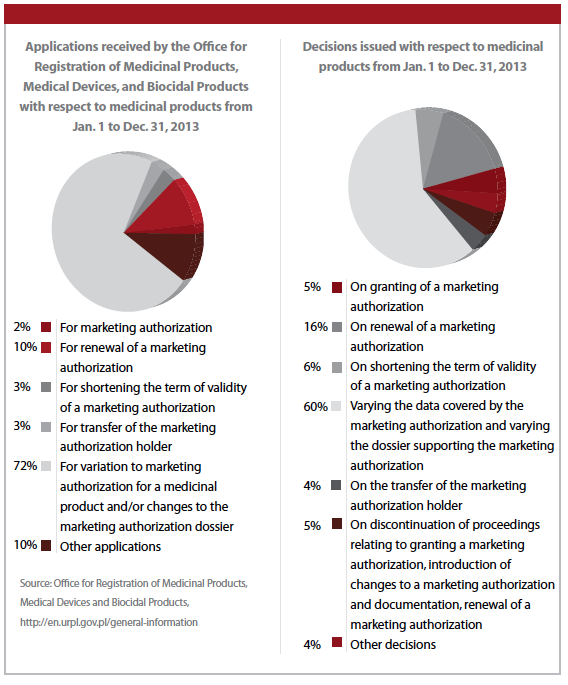
In 2013, there were 407 applications to the Polish regulatory authority to commence a clinical trial for a medicinal product, Ms. Matusiak says.
According to a report from the regulatory authority, the majority of registered clinical trials are Phase III trials (more than 50%) and Phase II (more than 30%) trials.
In 2013, more than 18% of the registered clinical trials of medicinal products were for oncology.
Phase IV studies are also expected to increase, Dr. Przybyś says. Early phase trials are still viewed as being rather underrepresented in Poland.
Poland’s regulatory processes are predictable and transparent, Dr. Piotrowski says.
“Poland does not have major corruption issues, investigators are experienced, logistics are easy and improving, and the costs remain lower than in western European countries or North America," Dr. Piotrowski says. “Although the same is true of neighboring countries, these lack size and scale. So rather than having 10 clinical sites in three small countries, and going through all trial processes, one can find those 10 sites in one country — Poland."
He adds that the regulatory framework in Poland allows for predictability, which is important for companies looking to bring drugs to market.
“Furthermore, Poland has a proper level of support from regulatory agencies that are open to discussing new research plans, as well as an effective ethics committee review process," Dr. Jedrzejowski says.
 Polish clinics and hospitals meet all international standards. Investigators take an active part in many international conferences, not just as passive participants but very often as chairs or presenters, Dr. Przybyś says.
Polish clinics and hospitals meet all international standards. Investigators take an active part in many international conferences, not just as passive participants but very often as chairs or presenters, Dr. Przybyś says.
Poland reflects the global trends in clinical trial research: there were many active cardiovascular and psychiatric clinical studies in the 1990s, later on osteoporosis became very popular. Recently, oncology, neurology, and rheumatology represent more the 50% of the pipeline.
Ms. Stefanczyk says that Poland’s fast patient recruitment rates can be attributed in part to the low share of innovative drugs in the market, which means that patients have limited access to innovative therapies and look for alternative forms of treatment.
“Participation in clinical trials, especially in the case of in-patient testing, very often offers superior quality of service, faster access to treatment — waiting lists do not apply, and the availability of expensive drugs at lower or no cost," Dr. Jedrzejowski says.
Health Costs and Access
Healthcare expenditures account for 7.4% of the Polish GDP, which surpasses the average expenditures in the region, but is still lower than in Western Europe (10%), Dr. Przybyś says.
He adds that according to a forecast, by 2016 healthcare expenditures in Poland will have increased by 50%, exceeding $40 billion. A new report by the finance ministry estimates that by 2017, GDP per capita in Poland will reach 74% of the average in the European Union.
The healthcare system is characterized by heavy out-of-pocket payments, one of the highest in Europe, Ms. Woroszylska-Sapieha says. (PV)



















