 The science of clinical development continually astounds even those of us intimately connected to it. We recognize that our overarching industry mandate — to develop and deliver safe and effective new therapies to fight and cure disease — must be continually informed by new thinking and tools if we are to realize not just incremental, but exponential success. The COVID-19 pandemic has underscored the need for this in an unprecedented way.
The science of clinical development continually astounds even those of us intimately connected to it. We recognize that our overarching industry mandate — to develop and deliver safe and effective new therapies to fight and cure disease — must be continually informed by new thinking and tools if we are to realize not just incremental, but exponential success. The COVID-19 pandemic has underscored the need for this in an unprecedented way.
Likewise, the engineering of artificial intelligence (AI)-powered automation also continues to astound. The pharmaceutical industry, which always has its ear to the ground for ways to improve the clinical trial process, is beginning to embrace AI to a greater degree than ever before. COVID-19 has highlighted the astonishing applications AI offers biopharma, and its almost limitless potential, while at the same time underscoring the industry’s overwhelming need to adopt and integrate AI to accelerate clinical trials. However, in order to fully leverage AI for clinical development the industry must trust AI, and we aren’t quite there yet. It is only through the collaboration of the great scientific minds of biopharma and the great engineering minds of tech that such trust can be established, thereby enabling the life sciences industry to effectively leverage AI to slingshot safe and effective drugs from lab to market in record time.
Connecting Scientific and Engineering Minds Thru AI
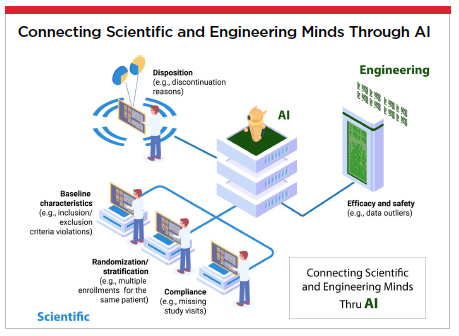 Just as drug development begins in the scientific lab, AI development begins in the engineering lab. Though seemingly separate and distinct, the rigorous research that serves as the foundation for both is actually parallel and complementary.
Just as drug development begins in the scientific lab, AI development begins in the engineering lab. Though seemingly separate and distinct, the rigorous research that serves as the foundation for both is actually parallel and complementary.
Unfortunately, at the early stages there is usually no line of sight from one to the other.
Leading software-as-a-service (SaaS) product companies have created AI research laboratories that focus on deep learning to optimize clinical trial technology and accelerate the drug discovery and development process. The AI algorithms and solutions born of the advanced engineering research conducted at these facilities bring immense value to clinical trial data review and management, enabling the industry to rapidly map raw data to STDM and expedite it for analysis.
By working together, biopharma and SaaS product companies can develop and deploy AI-powered analytical tools to clear many of the obstacles faced by study data managers and monitors. Combining AI-powered tech platforms with biopharma’s clinical data and domain knowledge facilitates the creation of deep learning models trained to make predictive analytics with unprecedented accuracy.
Machine learning models, trained to predict data discrepancies and manage queries using historical clinical data, can offer a significant process improvement that reduces time to database lock. Sponsors and CROs can use current clinical and scientific data from electronic data capture (EDC) systems and third-party sources (labs, biomarkers, PK/PD) to almost instantly identify discrepancies and generate query text related to:
Baseline characteristics (e.g., inclusion/exclusion criteria violations)
Randomization/stratification (e.g., multiple enrollments for the same patient)
Compliance (e.g., missing study visits)
Disposition (e.g., discontinuation reasons)
Efficacy and safety (e.g., data outliers)
Data managers and medical monitors are able to identify data quality issues more efficiently and enable continuous improvement as machine learning models grow even smarter. AI as a tool can empower the industry to better understand its clinical trial models, helping to speed up trials and get drugs to the market more quickly.
AI During COVID-19
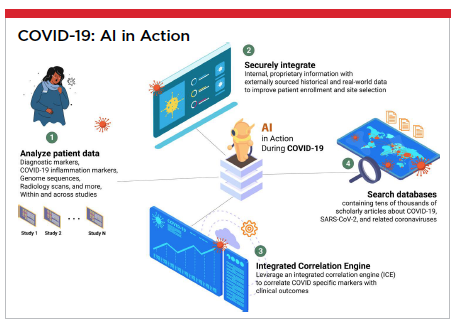 A current real-world example involves a state-of-the-art AI automation platform being leveraged to ensure data quality and management for a COVID-19 vaccine trial involving over 40,000 people. Because of the AI-powered solution being utilized, data entry to data cleaning took less than 24 hours. Natural language understanding (NLU) and machine learning enabled these powerful results.
A current real-world example involves a state-of-the-art AI automation platform being leveraged to ensure data quality and management for a COVID-19 vaccine trial involving over 40,000 people. Because of the AI-powered solution being utilized, data entry to data cleaning took less than 24 hours. Natural language understanding (NLU) and machine learning enabled these powerful results.
In a more broad application to the industry during the pandemic, AI is now also being built into command center solutions targeted at COVID-19 to combine the power of precision medicine with large-scale clinical trial operations. With unprecedented visibility into multiomics and clinical data sets — along with historical data from past trials — sponsors and CROs can improve inclusion/exclusion criteria, monitor individual patients, and track trends in patient cohorts, all while ensuring patient safety and drug efficacy. Such AI-powered command centers enable biopharma to:
Analyze patient data, including diagnostic markers, COVID-19 inflammation markers, genome sequences, radiology scans, and more, within and across studies
Securely integrate internal, proprietary information with externally sourced historical and real-world data to improve patient enrollment and site selection
Leverage an integrated correlation engine (ICE) to correlate COVID-specific markers with clinical outcomes
Search databases containing tens of thousands of scholarly articles about COVID-19, SARS-CoV-2, and related coronaviruses
Such command center tools also have broad applicability to other therapeutic areas, beyond COVID-19, and offer the ability to reshape clinical research processes and timing.
Explainability Key to AI Trust
 Success, however, is built on a foundation of trust. Critical to the industry’s willingness to and effectiveness at deploying AI in clinical research is achieving a comfort level for entrusting valued clinical development efforts to this amazing technology. This is where AI explainability comes in. Explainable AI describes the processes and methods used to apply AI, so that the results of an AI-generated solution can be understood by humans. It hinges on clear articulation of how the various techniques involved in machine learning models generate their outputs, thereby helping end users trust that the AI is making good decisions. Explainable AI is the antithesis of machine learning as a mysterious “black box," for which sometimes even tech engineers can’t explain the decisions produced.
Success, however, is built on a foundation of trust. Critical to the industry’s willingness to and effectiveness at deploying AI in clinical research is achieving a comfort level for entrusting valued clinical development efforts to this amazing technology. This is where AI explainability comes in. Explainable AI describes the processes and methods used to apply AI, so that the results of an AI-generated solution can be understood by humans. It hinges on clear articulation of how the various techniques involved in machine learning models generate their outputs, thereby helping end users trust that the AI is making good decisions. Explainable AI is the antithesis of machine learning as a mysterious “black box," for which sometimes even tech engineers can’t explain the decisions produced.
The focus of explainable AI is transparency and providing direct line-of-sight to understanding the predictions at which AI arrives. It is crucial that AI tech partners to life-science companies ensure explainable AI in order to establish and enable its continued, effective deployment, as well as the ability to build upon and expand today’s successes into tomorrow.
The Next Frontier of AI
The road to precision medicine of the future, where a person’s genetic make-up, lifestyle, and environment inform how researchers develop targeted therapies, is paved with AI. The one-size-fits-all approach to clinical development will be cast aside in favor of a more personal approach to disease treatment and prevention.
Existing and future generations of AI platforms can and will continue to facilitate dynamic visualization, analysis, and interrogation of data across clinical research programs to further realize the promise of precision medicine. The seamless integration they offer can be leveraged to curate and animate clinical trial data for more actionable insights and faster, more reliable decision making.
The great scientific minds of the pharmaceutical industry can utilize these advanced AI platforms, developed by the great engineering minds of the tech industry, to do their jobs more efficiently than ever before. By learning to trust AI and leveraging it in clinical development, safe and effective new therapies can be slingshot from lab to market for the people who need them, both during and after the COVID-19 pandemic.(PV)
Saama Technologies is the No. 1 AI clinical analytics platform company, enabling the life sciences industry to conduct faster and safer clinical development and regulatory programs.
For more information, visit saama.com.





















