Turning on the Switch
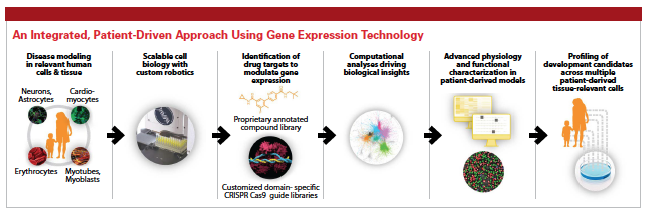
Fulcrum’s proprietary product engine identifies drug targets that can modulate gene expression to treat the known root cause of gene mis-expression.
 Bringing more than 30 years of R&D experience to his role as president and CEO of Fulcrum Therapeutics, Robert Gould, Ph.D., joined the company at its launch in 2016. In a very short time, he has helped drive the clinical-stage biopharmaceutical company toward a possible solution to modulating gene expression to treat the root cause of genetically defined diseases. Fulcrum Therapeutics works on unlocking gene control mechanisms to develop small-molecule therapies that modulate the on/off control mechanisms that regulate genes. Focused on genetically defined rare diseases in areas of high unmet medical need, Fulcrum’s proprietary product engine identifies drug targets that can modulate gene expression to treat gene misexpression.
Bringing more than 30 years of R&D experience to his role as president and CEO of Fulcrum Therapeutics, Robert Gould, Ph.D., joined the company at its launch in 2016. In a very short time, he has helped drive the clinical-stage biopharmaceutical company toward a possible solution to modulating gene expression to treat the root cause of genetically defined diseases. Fulcrum Therapeutics works on unlocking gene control mechanisms to develop small-molecule therapies that modulate the on/off control mechanisms that regulate genes. Focused on genetically defined rare diseases in areas of high unmet medical need, Fulcrum’s proprietary product engine identifies drug targets that can modulate gene expression to treat gene misexpression.
One of its treatments includes losmapimod, which was previously studied extensively by GSK as a treatment for COPD, with no clinical success. In 2016, GSK shelved the product, and three years later Fulcrum bought it and began researching its viability as a treatment for a type of muscular dystrophy, facioscapulohumeral or FSHD. In the early stages, FSHD affects muscles in the face or facio, which often presents as loss of ability to whistle or smile or other communication deficits associated with facial effects; the disease then typically will move into the muscles of the upper shoulder and shoulder girdle — the scapular muscles into the upper muscles of the arm — and eventually the trunk.
In fewer than two years, the drug has advanced to Phase IIb clinical trials. Losmapimod, which had been tested in more than 3,500 people during prior clinical research, had an established safety profile.
“Due to the previous work done by GSK, we were able to in-license the compound and jumpstart our FSHD program, which enabled us to move very quickly into a Phase II study in FSHD patients," Dr. Gould says.
“What was attractive and interesting to us about this particular disease is that the genetic cause is very well-known."
FSHD is caused by the aberrant expression of the DUX4 gene in skeletal muscle, resulting in the inappropriate presence of DUX4 protein.
Normally, DUX4-driven gene expression is limited to early embryonic development, after which time the DUX4 gene is silenced. In patients with FSHD, the DUX4 gene is unsilenced as a result of a genetic mutation. The result is muscle death replaced by fat, resulting in skeletal muscle weakness and progressive disability presenting as a dystrophy.
There are currently no approved therapies for FSHD and no other industry-sponsored programs in clinical development.
“We were able to obtain skeletal muscle cells from patients and through our proprietary platform we found the broken switch that’s causing DUX4 protein to be expressed when it shouldn’t be," he says. “It may be overly simplistic, but here is a useful analogy: in FSHD there’s a light that’s permanently on. As a result, DUX4 is permanently being expressed in the cell, when the switch should have it turned off."
The broken switch is a protein that through its enzymatic activity is causing DUX4 to be expressed. “When we inhibited that protein, we could turn DUX4 off and stop the muscle cells from dying in vitro," Dr. Gould says. “That was really exciting to us because we linked two pieces of information. FSHD is caused by a deletion in the genetic code and that deletion causes DUX4 to be turned on. In between that deletion and DUX4 being turned on is a switch called p38 that is contributing to the DUX4 being turned on."
Fulcrum is using its expertise in gene regulation to advance additional development programs for other rare diseases, such as sickle cell disease and beta thalassemia.
“Additionally, we’re focusing on those diseases where we think small molecule therapies are the most relevant," Dr. Gould says. “Our platform is set up to identify mechanisms that are amenable to small molecule inhibition or activation to regulate gene expression."
Fulcrum is also partnering with Acceleron on the identification of therapeutic targets and small molecule drug candidates for respiratory and pulmonary disease.
“We are excited that we’re in muscular dystrophy, hematology with sickle cell, and now respiratory disease with our partnership with Acceleron," Dr. Gould says. “Our product engine is applicable across a number of these genetically defined diseases, so we’re really excited about that potential." (PV)