 In 2020, the phrase “clinical trial solutions" takes on a new — and more relevant — meaning than ever before. The world needs a solution to the global COVID-19 pandemic, and we in the life-sciences industry can contribute to that solution through innovative, state-of-the-art technology that enables clinical trials to not only proceed, but flourish during these unprecedented times. AI-powered remote monitoring capabilities enable all clinical trials, COVID-19-related and otherwise, to continue as the world battles the novel coronavirus.
In 2020, the phrase “clinical trial solutions" takes on a new — and more relevant — meaning than ever before. The world needs a solution to the global COVID-19 pandemic, and we in the life-sciences industry can contribute to that solution through innovative, state-of-the-art technology that enables clinical trials to not only proceed, but flourish during these unprecedented times. AI-powered remote monitoring capabilities enable all clinical trials, COVID-19-related and otherwise, to continue as the world battles the novel coronavirus.
The life-sciences community has responded impressively to the dire need to investigate new therapies that prevent and treat the novel coronavirus. Biopharma and its partners have pivoted with lightning speed to expedite clinical operations that explore the potential use of existing drugs, as well as identify and test promising new therapeutic options for COVID-19.
Throughout this time, the industry has also been faced with the challenge of continuing trials for other disease states that were in progress before the global pandemic, and that both require and deserve uncompromised focus. The public health threat and resulting stay-at-home restrictions and social distancing guidelines ladder up to what, at first glance, might seem to be insurmountable obstacles to these goals. But, to the credit of the strong partnership between the life sciences and technology industries, this is not the case. In March 2020, as COVID-19 took hold of the world, the FDA issued guidelines recommending remote monitoring to 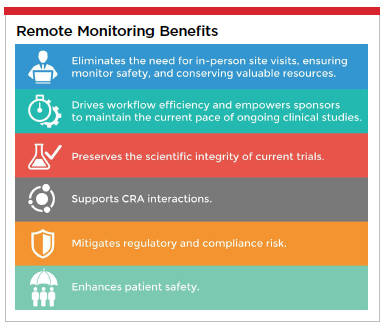 maintain oversight of clinical studies during the pandemic. This direction dovetailed with earlier guidance, updated in 2016, on the importance of these capabilities to help detect data anomalies in real-time and quickly correct any identified deficiencies in clinical trial processes.
maintain oversight of clinical studies during the pandemic. This direction dovetailed with earlier guidance, updated in 2016, on the importance of these capabilities to help detect data anomalies in real-time and quickly correct any identified deficiencies in clinical trial processes.
Clinical trials have continued to grow in complexity and scope during recent years. The global partnerships born of the pandemic to uncover COVID-19 treatments and cures surpassed the scope of previous collaborations, and have provided an added layer of intricacy to the clinical research process. Traditional site visits by monitors to review and confirm case report form (CRF) data, capture adverse events, and ensure appropriate protocol deployment frequently entailed 100% source data verification. This was a cumbersome, time consuming and resource-draining proposition in the best of times, and nearly impossible to achieve during a global pandemic.
Enter Remote Monitoring
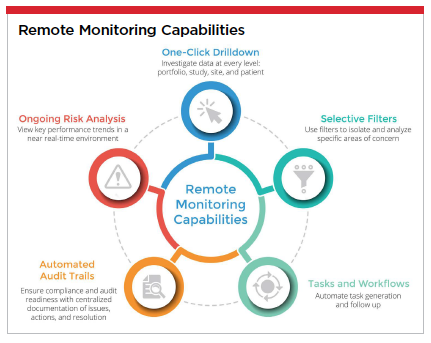 Today, more than ever before, state-of-the-art data analytics can and are being successfully leveraged by sponsors worldwide to manage studies and sites remotely through AI-powered clinical applications that aggregate data from electronic data capture (EDC) and clinical trial management systems (CTMS), enabling key performance indicators (KPIs) and key risk indicators (KRIs) surrounding vital pressure points in clinical trials.
Today, more than ever before, state-of-the-art data analytics can and are being successfully leveraged by sponsors worldwide to manage studies and sites remotely through AI-powered clinical applications that aggregate data from electronic data capture (EDC) and clinical trial management systems (CTMS), enabling key performance indicators (KPIs) and key risk indicators (KRIs) surrounding vital pressure points in clinical trials.
Remote monitoring, increasingly proven in recent years to be a beneficial and viable supplement for clinical studies, has been catalyzed by the pandemic to become a vital necessity for today’s drug research efforts. Remote monitoring offers numerous benefits that let sponsors work smarter and safer.
Many sponsors, organizations, and institutions have been leveraging the advantages of remote monitoring in their clinical research efforts for years. The Leukemia & Lymphoma Society (LLS) embraced remote monitoring and faster data acquisition in its Beat AML (BAML) Master Clinical Trial to help get medical treatments developed more quickly. BAML manages complex oncology trials that run more than 11 protocols simultaneously at 15-plus major academic institutions, with very little dependence on on-site visits. Recently Len Rosenberg, Ph.D., R.Ph., Head of Clinical Operations at Beat AML, LLC, a division of LLS, said:
“BAML was focused on efficiency and speed of operations, not on the coronavirus outbreak or pandemics in general. Yet, the model and technologies used by BAML are applicable for biopharma sponsors who want to adapt their clinical operations … while preparing for another outbreak or weathering the current crisis."1
LLS’s Beat AML Master Clinical Trial is an excellent example of remote monitoring done successfully and effectively, and a model worthy of adoption by other biopharma sponsors, CROs, and non-profit organizations. Although there is no one-size-fits-all solution, data analytics companies can help develop and customize programs that meet the varied needs of individual organizations. With a risk-based management approach, sponsors have a better chance at ensuring site productivity and responding to site-specific issues before they become major problems, during or after the pandemic.
Many of the new collaborations founded for the express purpose of identifying therapies to prevent and treat COVID-19 count remote monitoring capabilities as part of the state-of-the-art data analytics platforms that are integral to their efforts. One example is the EndPandemic National Data Consortium, an organization with the mission to integrate data from all ongoing and future clinical studies to dramatically accelerate analysis on COVID-19 and SARS-CoV-2 research to reduce the time to find a cure. The consortium utilizes a unique platform that allows researchers to dynamically and remotely visualize, analyze, and interrogate data across all available programs.
The life-sciences industry, in collaboration with its data analytics partners, has proven its ability to, as the British said during World War II, “Keep calm and carry on" in the fight against the novel coronavirus. COVID-19 has and will continue to change the model for everything we know, in many cases accelerating the adoption of new guidelines and parameters that have been talked about for years. The global office-based workforce has been transformed, and will likely remain altered to a great degree; systems of mass transportation, their use, and associated protocols will continue to evolve; and the norms related to social gatherings and events will come under increased scrutiny and regulation. Though the pandemic also has the potential to slow clinical research, going digital is enabling sites and sponsors to share information and achieve successful virtual collaboration, not letting COVID-19 slow them down at all, while at the same time working to architect its obliteration.
The life-sciences industry has undergone a transformation, one that will ultimately benefit humankind. The remarkable ability and agility that has been demonstrated by biopharma in pursuit of COVID-19 treatments and cures, all while maintaining critical clinical research for other conditions, can be attributed in great measure to increasingly nimble adoption of data analytics capabilities like remote monitoring that enable sponsors to move forward without missing a beat. Life science companies investing in and adopting remote monitoring capabilities to transition from traditional, onsite monitoring are making a wise and strategic investment that will accelerate outcomes, reduce risk, improve data quality, increase patient safety, save money and ensure the future viability of their clinical research programs.
Although hastened by the pandemic, it is likely that this “new normal" of remote trial monitoring is here to stay, and its overarching benefits will fortunately continue to be realized as it drives clinical trial efficiency and safety, now and into the future. As it turns out, what’s critical during a pandemic is good practice all of the time.(PV)
Editor’s Note:
1 https://www.clinicalleader.com/doc/clinical-operations-in-the-age-of-a-pandemic-0001
Saama is the #1 AI clinical analytics platform company, enabling the life sciences industry to conduct faster and safer clinical development and regulatory programs. Saama’s award-winning Life Science Analytics Cloud (LSAC) platform facilitates an unprecedented, authoritative oversight of comprehensive clinical research data, enabling companies to file New Drug Applications (NDAs) more efficiently and bring drugs to market faster.
For more information, go to www.saama.com and follow Saama @SaamaTechInc.





















