Nemaura Medical Repurposes CGM for Temp Monitoring
Trend Watch: Health technology aids in the diagnosis and monitoring of COVID-19
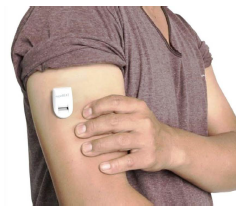 Nemaura Medical, a medical technology company focused on developing micro-systems-based wearable diagnostic devices, is repurposing its non-invasive and flexible continuous glucose monitor (CGM) sugarBEAT as a continuous temperature monitor (CTM) since its built-in temperature sensor is capable of continuously tracking body temperature through the skin.
Nemaura Medical, a medical technology company focused on developing micro-systems-based wearable diagnostic devices, is repurposing its non-invasive and flexible continuous glucose monitor (CGM) sugarBEAT as a continuous temperature monitor (CTM) since its built-in temperature sensor is capable of continuously tracking body temperature through the skin.
CTMs have been recognized as potentially having a key role to play in battling COVID-19, with Google’s Verily announcing plans to develop a small body worn temperature patch that transmits data to a phone application to provide timely notification of fever and support earlier diagnosis and treatment of viral infections like the flu or coronavirus.
Nemaura’s body-worn skin patch designed to monitor glucose levels on the skin at five-minute intervals also contains a thermistor, which measures skin temperature as an integral part of the device. The Nemaura device could be repurposed for this standalone function.
Nemaura is considering various options to expedite the launch of a CTM, including potential partnerships and direct-to-consumer sales. The CGM device is CE approved in Europe and is in commercial production in the UK.
“We see a significant commercial opportunity to utilize our BEAT platform technology specifically for CTM purposes with data being automatically sent by low energy Bluetooth to a mobile phone app from which it can then be reviewed by the user and/or sent to a family member or caregiver," says Dr. Faz Chowdhury, CEO of Nemaura. “We believe that there are parallels between family members and caregivers remotely monitoring glucose fluctuations in a person, and monitoring fever caused by viral infections, providing a means of remote tracking and intervention. In the current climate we see this as an opportunity to contribute to general wellbeing and patient management."
Orion Health Launches Coronavirus Outbreak Monitoring Platform Globally
Orion Health has developed the first comprehensive coronavirus pandemic outbreak monitoring platform to alleviate the demand on health systems and reduce the risk of further spread of COVID-19. The Orion Health software that supports this will be offered at no cost to existing and future customers.
The core functionality of the solution includes the ability to remotely monitor and engage patients in their homes, facilitating communication between quarantined people and the healthcare service, as well as maintaining visibility of those recently discharged. The intention is to alleviate spikes in demand on health systems, reducing the risk of further spread of the virus and flattening the epidemiological curve.
Over time the solution will increasingly use AI to allow providers to identify patients at risk of deterioration and optimize their care. This will allow resources to be allocated to those most in need.
A globally available website has been developed to support the different clinical protocols that have been produced by countries and regions to support the unique aspects of their local health systems and the current progression of COVID-19 within their jurisdiction. This site provides a regionally configurable online symptom checker with a background calculation of patient risk based on a number of factors. From this website, the public will also be able to connect into remote patient monitoring functionality in regions where this is available.
Another aspect of the solution is the ability to remotely monitor, at home, health workers who have had exposure or fallen ill. This functionality can also be used to monitor patients that may have been discharged to the home early to free up hospital capacity.
Pear Therapeutics Earns FDA Approval for First Rx Digital therapeutic
 The FDA granted authorization for Somryst, made by Pear Therapeutics, as the first prescription digital therapeutic intended for use in the treatment of patients 22 years of age and older with chronic insomnia.
The FDA granted authorization for Somryst, made by Pear Therapeutics, as the first prescription digital therapeutic intended for use in the treatment of patients 22 years of age and older with chronic insomnia.
Somryst is the first product submitted through FDA’s traditional 510(k) pathway while simultaneously reviewed as part of FDA’s Software Precertification Pilot Program to help build and test FDA’s Digital Health Precertification Working Model 1.0.
Somryst is intended to provide tailored neurobehavioral interventions, specifically cognitive behavioral therapy for insomnia (CBTi) and sleep restriction driven by algorithms designed to improve the symptoms of insomnia. Somryst includes features that allow for personalization, including the patient’s ability to set a sleep window. The American Academy of Sleep Medicine and the American College of Physicians clinical guidelines recommend CBTi as first-line treatment for people with chronic insomnia.
“More than 30 million adults suffer from chronic insomnia, which can seriously impact one’s quality of life and can lead to depression, suicidality, hypertension and even heart attacks. Treatment options for chronic insomnia are limited as most available sleep medications are only recommended for short term use, can lead to problems with tolerance, and can have unwanted side-effects, including next-day cognitive impairments," says Dr. Charles M. Morin, PhD, Professor of Psychology, Director Sleep Research Centre at Université Laval.
The FDA submission was supported by two randomized controlled trials evaluating the effectiveness of the therapeutic. Together, these studies enrolled more than 1,400 adults with chronic insomnia. In a trial of 303 patients with chronic insomnia, those on treatment demonstrated clinically meaningful improvements in insomnia severity at the end of treatment, as well as at six and 12 months follow-up compared with active control. Study results were published in JAMA Psychiatry.
Swittons Debuts First Fully Customizable IoT Powered Solution to Improve Pharma to Physician Communication
Swittons, a P360 company, has launched the first fully customizable IoT powered solution for pharmaceutical commercial organizations and physicians. Swittons enhances the pharma-to-physician relationship by helping representatives cover more territory, and by breaking down communication barriers to office visits, professional consultations, and product orders. The smart device can also be configured for biotech and medical device sales organizations.
“At Swittons, we have reimagined the world of pharma-to-physician communication by fundamentally changing the way marketers and sales organizations interact with customers and prospects," says Swittons CEO and Founder Anupam Nandwana. “Our solution not only saves organizations time and money it gives them a new digital channel of engagement for meaningful interactions. From dashboard to devices to data, Swittons powers seamless engagement."
Swittons is designed to be compatible with existing commercial infrastructure and integrates seamlessly with leading CRM and ERP systems. The device, which can be custom branded, comes programmed to execute up to four different predefined functions. Bringing critical data from a device to the enterprise has never been easier, because Swittons takes care of all the complex technical work.
Physicians appreciate Swittons because the device enables them to be in better control of their schedule. At the click of a button, they can request samples, schedule sales visits, book medical science liaison (MSL) consultations, and access important medical information. And since the four-inch device takes up very little desk space, it is conveniently available and integrates seamlessly with physician workstyles. The smart devices can be preprogrammed with individualized functionality as well.
For physicians who receive Swittons, the device comes out-of-the-box ready and automatically connects through a Wi-Fi or GSM cellular connection.
Biofourmis and Hong Kong’s Dept. of Health Partner on Remote Monitoring Platform to Fight Coronavirus
Boston-based Biofourmis is being leveraged in a remote monitoring and disease surveillance program in Hong Kong involving patients who have been diagnosed or are suspected to have COVID-19. The national disease-monitoring program — administered by The University of Hong Kong’s Department of Health — also includes Hong Kong-based Harmony Medical Inc., which is Biofourmis’ joint-venture partner for the China region.
Diagnosed and potentially COVID-19-infected patients are being monitored with Biofourmis’ Biovitals Sentinel platform, a turn-key solution that the company built in less than two weeks specifically for this initiative by customizing its FDA-cleared AI-powered biovitals analytics platform. Biovitals Sentinel’s 24/7 remote monitoring technology and analytics are providing clinicians involved in the COVID-19 program with clinical decision support for early identification of any physiological changes that could indicate deterioration, to enable earlier interventions for better outcomes.
To measure body temperature and numerous other biomarkers, patients are wearing Everion, which has several medical-grade sensors, including optical, temperature, electrodermal, accelerometer, and barometer.
Everion can be comfortably worn for 24 hours a day for several days and has much higher-than-average patient satisfaction and adherence rates.
AI-powered Biovitals Analytics will derive more than 20 physiological signals from the sensor data, including temperature, heart rate, blood pulse wave, heart rate variability, respiration rate, inter-beat-interval, and others. These signals are then fed through advanced AI and machine-learning techniques to flag key physiological changes that could indicate disease progression.
The Biovitals Sentinel solution also includes a smartphone app to collect qualitative data reported by patients as they respond to AI-generated “smart nuggets." Treating physicians, through the Biovitals Sentinel clinician-facing web dashboard, will observe and be alerted to any significant physiological changes and adverse events, so they can intervene when necessary.
Biofourmis is in conversations with government agencies in other regions about applying its technology to help even more patients and fight against the global spread of COVID-19 so that it can be treated more effectively.
Micro-OCT Lets Docs Spot Tumors Below Tissue Surface
A collaboration between researchers at Nanyang Technological University, Harvard Medical School, and University of Alabama has led to the development of a prototype device capable of imaging through tissues at resolutions down to one micrometer. The micro-OCT imager takes advantage of optical coherence tomography (OCT) at wavelengths between 700 and 950 nanometers. At these wavelengths, the near-infrared light can penetrate a few millimeters below the skin, as well as other soft tissues, to elucidate the structure of individual cells below.
The technology doesn’t rely on expensive equipment, such as CT and MRI, and doesn’t involve ionizing radiation, which should make it appropriate for screening and diagnosis of a number of cancers.
To allow the micro-OCT to produce 3D images, the device emits infrared light and measures the bounce back from the tissues. A computer algorithm converts these data, coming from individual 2D cross-section slices, into a colorized 3D representation of the cellular structures being imaged.
The technology is easy enough for just about anyone to use by simply applying the imager against the tissue of interest and activating it. It works in a matter of minutes and the results can be analyzed by non-pathologist physicians.
The device has already been tested at Wuhan University for the detection of colon polyps, back before the COVID-19 emergence, and it allowed non-pathologists to be in 95% agreement, regarding malignancy, with senior pathologists analyzing the same tissues.
SF Hospitals Test mHealth Wearables for Coronavirus Monitoring
A project launched by UCSF will allow hospital officials to monitor front-line care workers through an mHealth wearable device that measures heart and respiration rate as well as changes in body temperature. In a project developed by the University of California at San Francisco, emergency medical workers at UCSF and Zuckerberg San Francisco General Hospital are wearing an Oura Ring, which through an app monitors heart and respiratory rates and changes in body temperature. The digital health device is being given to staff who have recently come in contact with people infected by COVID-19 and will be worn for three months.(PV)




















