Boehringer-Ingelheim Launches Patient-Centric MyStudyWindow Powered by Carebox
Trend Watch: HealthTech Innovations Continue to Transform Patient Care
 Boehringer Ingelheim and Carebox Healthcare Solutions have launched MyStudyWindow, a digital platform empowering patients, families, caregivers, and doctors to learn about Boehringer Ingelheim’s studies by offering information in an easy to access and understandable way. The collaboration brings together Carebox’s expertise in study matching based on patient eligibility criteria and Boehringer Ingelheim’s extensive scientific capabilities to pursue innovation on behalf of patients around the world.
Boehringer Ingelheim and Carebox Healthcare Solutions have launched MyStudyWindow, a digital platform empowering patients, families, caregivers, and doctors to learn about Boehringer Ingelheim’s studies by offering information in an easy to access and understandable way. The collaboration brings together Carebox’s expertise in study matching based on patient eligibility criteria and Boehringer Ingelheim’s extensive scientific capabilities to pursue innovation on behalf of patients around the world.
The digital platform MyStudyWindow follows a patient-centric approach using lay friendly language, educational content, and user-friendly features that provide guidance: Users can select from a range of condition-specific questionnaires to anonymously answer questions about a patient’s diagnosis, disease stage, and treatment history. MyStudyWindow instantly matches the user to potentially relevant clinical studies from the Boehringer Ingelheim portfolio, suggests nearby study site locations, and provides guidance on contacting the site for next steps. For studies that Boehringer Ingelheim would conduct in the future, patients will have the opportunity to provide feedback on their research experience to further stepping closer towards clinical trial democratization. The functions are facilitated by AI-powered automation which enables Carebox to continuously curate and structure eligibility.
Electronic Bandage Delivers Drugs, Leaves No Scar
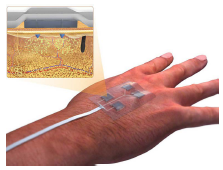 Researchers from University of Nebraska-Lincoln, Harvard Medical School, and University of Connecticut have developed an electronic bandage that can deliver multiple drugs deep into a wound when programmed to do so.
Researchers from University of Nebraska-Lincoln, Harvard Medical School, and University of Connecticut have developed an electronic bandage that can deliver multiple drugs deep into a wound when programmed to do so.
Wounds require different drugs at different stages of progression, but simply placing topical medication on the wound is not optimal. The new bandage has tiny needles that penetrate into the wounded tissue and electronically controlled drug chambers can be activated to push drugs through the needles.
No bandage changes are necessary during drug administration, so the wound is allowed to stay covered for longer.
The bandage is wirelessly controlled and can be operated using a smartphone, allowing clinicians or even patients themselves to regulate the drug release.
The system has already been tried in diabetic mice with skin injuries and the device helped the mice to heal nearly completely, and even minimized scar formation at the wound sites.
There’s already a patent application filed for the new bandage.
Smartphone with Laser and Thermal Cameras to Screen Passersby for Fever
 Singapore’s Integrated Health Information Systems (IHIS) and AI startup KlonKare is testing a fever screening system that uses thermal and laser cameras, coupled with a smartphone, to measure people’s temperature at a distance. The technology can help to speed travelers through transit hubs, hospital entrances, and other places, while alleviating the need for large numbers of screeners to manually measure everyone’s temperature.
Singapore’s Integrated Health Information Systems (IHIS) and AI startup KlonKare is testing a fever screening system that uses thermal and laser cameras, coupled with a smartphone, to measure people’s temperature at a distance. The technology can help to speed travelers through transit hubs, hospital entrances, and other places, while alleviating the need for large numbers of screeners to manually measure everyone’s temperature.
The device accepts a smartphone within a built-in cradle and the combo can be placed on a tripod near a door or anywhere else there are people passing by that have to be screened. Using artificial intelligence, the technology is even capable of measuring the temperature of people that are wearing hats, glasses, face masks, and other head-wear. It does this by recognizing the forehead of those passing in front of the cameras and compensates accordingly, including for the ambient temperature.
The system has been undergoing testing in Singapore:
St. Andrew’s Community Hospital is partnering with IHIS to be a pilot site for iThermo to see if it can be a viable and cost-efficient solution for the screening of temperature of community hospital visitors. iThermo has the potential to cut down on the time taken to check each visitor’s temperature compared to the manual method.
The device, which can measure temperature from up to a distance of 3 meters away, can be used to monitor people standing in line, and will generate an alert when someone with a fever walks by it, prompting hospital staff or screeners to carry out secondary checks on those people using a standard method.
First Databank’s Authoritative Drug Information Helps Answer Customer Questions on Amazon Alexa
 Thanks to First Databank, provider of drug and medical device knowledge that helps healthcare professionals make precise decisions, consumers now have access to a wide variety of drug information from FDB through simple queries via Alexa.
Thanks to First Databank, provider of drug and medical device knowledge that helps healthcare professionals make precise decisions, consumers now have access to a wide variety of drug information from FDB through simple queries via Alexa.
This initiative will leverage concise, clinical drug information authored specifically for Alexa by FDB clinicians, based on their review of the most relevant content from the company’s proprietary consumer drug information monographs. Additionally, the project provides custom content that allows consumers to ask about a drug’s effects such as drug interactions, side effects, precautions, and the drug’s class. The custom data file will provide information for Alexa customers in both English and Spanish and will be updated on a regular basis.
A common medication-related query might be: “Alexa, what is Tylenol?"
Smart Jumpsuit to Spontaneously Measure Infants Movements
 In a first, researchers have developed a smart jumpsuit that accurately measures the spontaneous and voluntary movement of infants from the age of five months, an advance that may aid in the early diagnosis of neurological disorders affecting mobility.
In a first, researchers have developed a smart jumpsuit that accurately measures the spontaneous and voluntary movement of infants from the age of five months, an advance that may aid in the early diagnosis of neurological disorders affecting mobility.
The study, published in the journal Scientific Reports, noted that the assessment of spontaneous and voluntary movements is part of the neurological examination of infants.
According to the researchers, including those from the University of Helsinki in Finland, the quantitative tracking of children’s motility in their natural environment has not been possible. They said until now children have been primarily qualitatively assessed at the physician’s or physiotherapist’s practice.
Researchers say the data obtained from the smart jumpsuit is valuable, since the detection of abnormalities in the neurological development of infants at an early stage enables early support.
They said the ability of the human brain to form new nerve connections and facilitate new learning is at its strongest in early childhood. These brain functions are benefited by supporting child development, which are targeted at recurring intervals.
SmartTab Wireless Pill for Targeted Drug Delivery
 Colorado-based Velóce Digital Health is developing the SmartTab, an ingestible capsule that can be wirelessly controlled via a smartphone to release its contents at precise locations within the gastrointestinal system.
Colorado-based Velóce Digital Health is developing the SmartTab, an ingestible capsule that can be wirelessly controlled via a smartphone to release its contents at precise locations within the gastrointestinal system.
SmartTab works using several key technological features. The capsule itself is powered by inductive charging. Once swallowed, the capsule is tracked electronically using a small adhesive patch worn on the skin near the target area. When the capsule approaches the desired spot for administration, the contents can be released automatically or by the user pushing some buttons on the accompanying iPhone app.
Of all the potential drugs that SmartTab could deliver, the company hopes to start with medications for Crohn’s disease. Other uses could include long term antibiotics, heart medications, or epilepsy treatments.
In 2019, SmartTab completed animal pre-clinical studies and demonstrated that the pill could successfully release its contents at the targeted area upon receiving the wireless signal. Human clinical studies will begin in 2020 towards the goal of FDA approval. From there, the company will continue down the FDA approval track, with a two- to three-year submission period ahead of them.
The idea of SmartTab was born when founder and CEO Robert Niichel, after a career in the pharmaceutical space, saw an opportunity to mix wireless technology with the antiquated practices still associated with drug delivery.
Starting in 2017, the company began developing the SmartTab. The device has no onboard batteries, is powered conductively, and contains a small microprocessor. Mr. Niichel says he anticipates that a pharmaceutical company will look to acquire the solution after FDA approval.
“The idea is that with the smart capsule, you will have precision medicine delivered to a very targeted area," Mr. Niichel says. This approach could reduce drug dosages, adverse effects, and improve patient outcomes.(PV)




















