The World’s First Virtual Caregiver Unveiled
Trend Watch: Meeting Patient Needs Through Technology
SameDay Security, a provider of automated home care solutions and safety devices, unveiled the world’s first virtual caregiver, Addison Care, earlier this year at the annual Computer Electronic Show (CES).
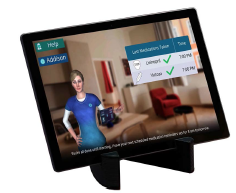 Addison Care is named after its ambient augmented reality virtual caregiver, Addison. She’s a state-of-the-art, 3D-animated caregiver designed to engage aging and chronically ill clients at home to supplement care and through various health and safety features. Appearing on 15-inch monitors strategically placed throughout the residence, Addison carries on two-way conversations, and she is programed for a user’s personal needs and plans of care.
Addison Care is named after its ambient augmented reality virtual caregiver, Addison. She’s a state-of-the-art, 3D-animated caregiver designed to engage aging and chronically ill clients at home to supplement care and through various health and safety features. Appearing on 15-inch monitors strategically placed throughout the residence, Addison carries on two-way conversations, and she is programed for a user’s personal needs and plans of care.
Addison’s capabilities include 24/7 in-home checkups monitoring a person’s activity, including providing medication reminders and verifying adherence; the tool provides real-time assessments if a client develops evidence of increased risk of falling or health decline. She measures health performance, rewards patients for making progress, collects vitals, and conducts in-home examinations.
The company says Addison requires a complex array of devices, configured for instant and easy implementation, involving technologies such as Bluetooth, cellular, Internet, cloud computing, edge machine learning processing, complex visual sensing, clinical interfaces, conversational speech, augmented reality, robust security, and a dynamic user interface.
SDS is developing contracts with hundreds of home-care partners across America that will participate in Addison Care marketing to their clients. New clinical trials are scheduled with G60 Trauma of Phoenix Arizona involving 500 patients over three years to determine the impact on patient outcomes, cost reduction, lower hospitalization, chronic disease management, and long-term care objectives.
Otsuka and Click Develop Digital Therapeutics for Patients With Major Depressive Disorder
 Otsuka America and Click Therapeutics are collaborating to develop and commercialize a prescription digital therapeutic for treatment of major depressive disorder (MDD), with the intent to address unmet medical needs among this patient population and to improve outcomes.
Otsuka America and Click Therapeutics are collaborating to develop and commercialize a prescription digital therapeutic for treatment of major depressive disorder (MDD), with the intent to address unmet medical needs among this patient population and to improve outcomes.
This collaboration leverages Click’s ability to discover and validate a software application, with Otsuka’s expertise in developing approved prescription therapies for patients with serious mental illnesses, including Otsuka’s established development and commercialization capabilities.
“This collaboration signals Otsuka’s commitment to meet patients’ unmet medical needs by developing solutions far beyond medication," says Kabir Nath, president and CEO, Otsuka North America Pharmaceutical Business Division, Otsuka America Inc. “Our goal is to deliver evidence-based cognitive therapies to a broader population of patients with MDD than is currently feasible, due to the challenges of a shortage of mental health professionals and limited time for them to conduct cognitive therapy.
We are proud to be one of the few pharmaceutical companies that continue to invest in developing medicinal and digital products for the treatment of mental illnesses, and we are doing so by breaking down barriers and collaborating with leading therapeutic technology companies, such as Click, which share our vision."
Otsuka has agreed to commit capital to fully fund development of Click’s novel mobile application CT-152 for MDD. Otsuka will pay Click up to $10 million in upfront and regulatory milestone payments, along with an estimated $20 million in development funding. An additional $272 million in commercial milestone payments are contingent upon regulatory approvals. In addition, Click will receive tiered, double-digit royalties on global sales of the software and the digital therapeutic applications that result.
J&J and Apple Study Use of Apple Watches in Health Monitoring
Janssen Pharmaceuticals entered into a research study in collaboration with Apple to investigate whether a new heart health program using an app from Johnson & Johnson in combination with Apple Watch’s irregular rhythm notifications and ECG app can accelerate the diagnosis and improve health outcomes of the 33 million people worldwide living with atrial fibrillation (AFib), a condition that can lead to stroke and other potentially devastating complications.
The study aims to analyze the impact of Apple Watch on the early detection and diagnosis of AFib, and the potential to improve outcomes including the prevention of stroke. A multi-year research program will be launched later in 2019. This large-scale program will occur in the United States only, and will be designed as a pragmatic randomized controlled research study for individuals age 65 years or older.
Johnson & Johnson’s recent mSTOPs (mHealth Screening to Prevent Strokes) study demonstrated that earlier screening leads to increased AFib detection. “Utilizing wristwatch-based optical heart sensor and ECG monitoring is a logical evolution of this research and may also lead to increased AFib diagnosis and improved clinical outcomes for patients," says Paul Burton, M.D., Ph.D., VP, medical affairs, internal medicine, Janssen Scientific Affairs.
“We’re excited about the potential of common, wearable technology to aid in the earlier detection and prevention of a frequent cause of stroke," says Paul Stoffels, M.D., vice chair of the executive committee and chief scientific officer, Johnson & Johnson. “Based on the insights generated through this research program, we may be able to develop new ways to detect other health conditions earlier in the future that also exhibit measurable physiological symptoms."
In other news, Apple has been in talks with at least three private Medicare plans about subsidizing the Apple Watch for people older than 65 to use as a health tracker. The insurers are exploring ways to subsidize the cost of the device for those who can’t afford the $279 price tag, which is the starting cost of an older model. The latest version of the device, which includes the most extensive health features including fall detection and an electrocardiogram to measure the heart’s rhythm, retails for a minimum of $399, which many seniors could benefit from but can’t afford.
Bayer Partners with ZappRx to Improve Access to Specialty Medications
 ZappRx, a digital health company that streamlines the complex processes associated with prescribing specialty medications, is working with Bayer to provide a more efficient approach to the accessing of specialty medications and associated support programs.
ZappRx, a digital health company that streamlines the complex processes associated with prescribing specialty medications, is working with Bayer to provide a more efficient approach to the accessing of specialty medications and associated support programs.
ZappRx will support patients living with two specific types of pulmonary hypertension (PH) and patients with relapsing remitting multiple sclerosis (RRMS), conditions within Bayer’s Specialty Medicines portfolio.
Subsequent to the integration with ZappRx’s platform, Bayer will also gain access to unique data assets and meaningful insights around the treatment of these patient populations.
This agreement constitutes another meaningful step toward transforming how specialty medications are prescribed and accessed.
Specialty prescribing and prior authorizations are traditionally manual, paper-based processes heavily dependent on fax machines. ZappRx provides physicians with a comprehensive digital solution for specialty prescribing and prior authorization.
Providers using the ZappRx platform can improve their prescription approval rate from weeks to just a matter of days, even when dealing with the most complex cases.
NIH Integrates Fitbit Data into “All of Us" Research Program
 Participants in the NIH All of Us research program can now volunteer their Fitbit data to better inform the national population health research initiative.
Participants in the NIH All of Us research program can now volunteer their Fitbit data to better inform the national population health research initiative.
Digital health technologies, including Internet of Things (IoT) devices such as wearable fitness trackers, can add critical insight into everyday activities and physical fitness levels.
Participants in All of Us can already contribute their electronic health record data and biosamples in addition to answering surveys online.
Adding everyday information such as heart rate and step count can supplement researchers’ understanding of habits and routines that may contribute to health outcomes. The data will be linked to other contributed datasets, and securely protected in the same manner.
The program will be launching additional digital health partnerships with Fitbit and other companies in the future.
Proteus Digital Health Supports ChemoTherapy via Technology
Silicon Valley startup Proteus Digital Health has partnered with Fairview Health Services and the University of Minnesota Health system to provide its digital medicine technology to cancer patients to support chemotherapy treatments.
Data gathered from digital oral oncolytics will enable cancer drugs and treatment regimens to be optimized to work their best for each individual patient, something not possible until now. The company believes using the digital medicine will enable oncology patients to stay on their therapy longer, avoid hospital admissions and respond better to therapy overall.
Proteus’ new oral oncology drug is a digital version of capecitabine, a common chemotherapy medication often used to treat breast and gastrointestinal cancers. In this case, the company’s technology will be prescribed to Fairview Health Services and the University of Minnesota Health patients with stage 3 and 4 colorectal cancer.
Essentially what the digital pill does is helps to optimize treatment regimens by measuring and collecting information about medication usage and dosage, along with activity metrics. With patient consent, this data can be shared to the person’s physician, caretaker or pharmacist to help gauge and adjust treatment.
The digital oral chemotherapy pill was developed in concern with Fairview Ventures, the innovation arm of the health system, as well as oncology medication experts from Fairview Pharmacy Services and the University of Minnesota.
To build a stronger clinical evidence base, Proteus is also launching a digital oral oncolytic medication registry, which will collect data from multiple sites around the country testing the digital version of capecitabine as a treatment.
“Proteus’ expansion of support for digital medicines into the oncology treatment area is not only important for patients and providers, it will be a game changer for the industry developing therapies intended to one day eradicate cancer," Olivia Ware, Proteus’ senior VP of U.S. markets and franchise development, said in a statement.
Some of the company’s previous partnerships include a collaboration with Otsuka on Abilify MyCite, a digital sensor inside a pill that records medication adherence for the anti-psychotic medication. (PV)





















