 Decades of progress in information technology reveal, however, that as individuals and in the collective, we constantly struggle to find balance between the benefits of sharing and our desire to keep control.
Decades of progress in information technology reveal, however, that as individuals and in the collective, we constantly struggle to find balance between the benefits of sharing and our desire to keep control.
The mainframe computer was cheered as it enabled a corporation to share information that had previously been locked in metal file cabinets lining its hallways; but later we fell in love with the “personal" computer because we could once again do things our own way. When the benefits of sharing became sorely missed a few years later, the local area network was the solution, connecting departments of PC users, which were then connected into wide-area networks of departmental servers to prevent us from operating in silos.
Sharing Is Now Here to Stay
Once the Internet became widely available, the benefits of sharing overpowered the need for control within the boundaries of a corporation. Collaborations were formed and standards were negotiated to codify the ability to share, enabling data and applications to become accessible from any computer around the world. This commitment to collaboration and sharing has provided more independence, broader choices, and greater efficiencies than we could have ever imagined.
Collaboration, efficiency, and choice have been the guiding principles of TransCelerate Biopharma Inc.’s initiatives since its formation in 2012. It’s no accident that the concept of sharing is baked into the name of one of its largest initiatives — the Shared Investigator Platform — because the sponsor companies adopting this initiative understand the value that a shared common platform brings to each and every sponsor and clinical site, as well as to clinical research as a whole.
Shared objectives brought to fruition by the Shared Investigator Platform
When it comes to relationships with sites, sponsors of all sizes share these common objectives:
To find the most appropriate and competent sites for a given study
To initiate and execute studies as rapidly and economically as possible
To make participation in trials as easy as possible for site staff and patients
To continually expand the available clinical research pool of investigators
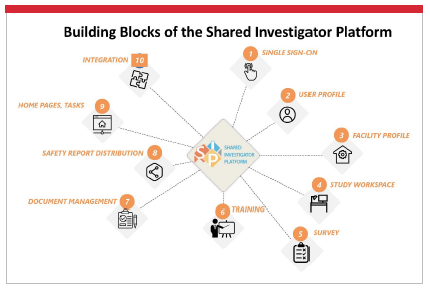 The Shared Investigator Platform consists of building blocks that will enable each and every sponsor to achieve these objectives. As a multi-sponsor platform, it provides 360-degree collaboration across trial resources — to sites as well as to sponsors — and unites existing clinical systems from multiple vendors in an open architecture.
The Shared Investigator Platform consists of building blocks that will enable each and every sponsor to achieve these objectives. As a multi-sponsor platform, it provides 360-degree collaboration across trial resources — to sites as well as to sponsors — and unites existing clinical systems from multiple vendors in an open architecture.
Vision of the Shared Investigator Platform: An Open, Collaborative Platform for All Stakeholders in Clinical Research
Solving the problem of proliferation
The digital age has delivered on its promise of paperless trials. Ironically, the benefits gained are being erased at the site level by the proliferation of digital systems. Sites consistently report that the number of systems and interfaces they need to manage per sponsor reduces their willingness to work with new sponsors.
This growing problem limits access to patients and delays the start-up of trials, and it cannot be solved by individual sponsor solutions. Collaboration and harmonization across sponsors is the only way to transform our industry from the inside out.
Investigator portals promoted by individual sponsors as one-stop access to all the resources needed to participate in clinical trials is a claim that doesn’t ring true for sites.In the eyes of site personnel, every pharmaceutical company-specific portal represents a myopic view of that sponsor’s universe, with redundant log-ins, GCP and system training, user and facility profile requests, and a host of clinical applications from a variety of vendors.
Collaboration and harmonization across sponsors is the only way to transform our industry from the inside out.
A multi-sponsor solution gives sites visibility
The Shared Investigator Platform is a multi-sponsor solution that elevates the site perspective. It provides sites with the exposure and visibility needed for the industry to make appropriate matches between the supply of investigators and the demand for clinical trials, at greater speed and larger scale. The results are increased engagement and efficiencies across clinical development resources:
Study start-up is accelerated, because both sponsors and sites contribute and have access to a single repository for profiles, CVs, and mutually recognized training courses and records.
Administration burden is reduced, because processes to develop and distribute feasibility studies, clinical documents, and safety notifications are shared.
Site personnel have more time to spend with patients and provide higher quality data, because they have the ability to manage tasks and workload across multiple sponsors.
By joining the Shared Investigator Platform, sponsors eliminate the expense of developing, maintaining, and supporting company-specific portals. They also reduce their own administrative burden associated with the existence of disparate processes and tools used by sites to manage multiple trials and multiple sponsors.
A single sign-on accesses a multi-vendor environment
Single sign-on sounds like a simple concept, but it is actually a complex function to achieve across the clinical research ecosystem. The Shared Investigator Platform provides identity, credentialing, and access management. With single sign-on access into all core functionality, as well as study-specific, sponsor-specific clinical systems, as enabled by each sponsor, the need for site personnel to create and remember dozens of user ID/password combinations for multiple sponsor portals and systems is eliminated.
Staying true to the vision of TransCelerate, the Shared Investigator Platform makes it easy for individual sponsors to connect and enable the use of established or new study-specific systems from their vendor of choice, including those for electronic data capture (EDC), interactive response technology (IRT), electronic patient-reported outcomes (ePRO), drug safety systems, and others. The Study Workspace provides secure, compliant, one-click access to all appropriate resources, significantly reducing the administrative burden on sites and the support requests made to sponsors.
Reversing unintended consequences
Is it time for your company to help reverse the unintended consequences of system proliferation and trial fatigue at investigative sites? The Shared Investigator Platform is gaining scale and is the only solution that will reduce the administrative burden on sites, speed study start-up, and increase efficiencies for all clinical trial stakeholders.(PV)
Editor’s Note: Cognizant was chosen by
TransCelerate in 2014 to develop, host, maintain, and support the Shared Investigator Platform.
Cognizant (Nasdaq-100: CTSH) is one of the world’s leading professional services companies, transforming clients’ business, operating and technology models for the digital era.
For more information, visit cognizant.com.





















