Amicus Therapeutics Launches Healing Beyond Disease
 Amicus Therapeutics has launched Healing Beyond Disease, a global, employee-driven initiative inspired by people living with rare diseases and their families. Healing Beyond Disease is designed to enhance and expand the company’s support for patients, families, and advocacy organizations beyond the company’s research and development of novel medicines. Healing Beyond Disease is based on five key pillars and leverages Amicus’ varied internal resources, existing philanthropic endeavors and drug development expertise to bring a different level of support to the rare disease community.
Amicus Therapeutics has launched Healing Beyond Disease, a global, employee-driven initiative inspired by people living with rare diseases and their families. Healing Beyond Disease is designed to enhance and expand the company’s support for patients, families, and advocacy organizations beyond the company’s research and development of novel medicines. Healing Beyond Disease is based on five key pillars and leverages Amicus’ varied internal resources, existing philanthropic endeavors and drug development expertise to bring a different level of support to the rare disease community.
The State of Innovation in Pain
While many promising treatment approaches are in development, lower investment and pipeline breadth in pain and addiction relative to other disease areas with high societal health burdens demonstrates the need for incentivizing more research and clinical development in these areas, according to a new report by Biotechnology Innovation Organization (BIO).
Key takeaways:
There have been only two novel chemical entities FDA approved to treat pain over the past decade.
The industrywide pain pipeline consists of 215 clinical-stage drug programs, with 125 of these testing novel chemical entities in the clinic, 87% of which are for non-opioid receptors. Over the past decade, the biopharmaceutical industry has been working to develop abuse deterrent formulations, with 142 clinical trials initiated and 12 FDA approvals for abuse deterrent pain products.
Clinical success in pain drug development has been extremely difficult for novel drugs, with only a 2% probability of FDA approval from Phase I compared with an overall 10% success rate across all diseases.
Private company investment, as measured by venture capital into U.S. companies with lead-stage programs in pain, is 3.6% of total drug development venture funding. For venture funding of novel R&D, pain has received 17 times less venture capital than oncology over the last decade.
Venture investment for addiction drug R&D is nearly nonexistent.
Health Spending Expected to Grow
Under current law, national health spending is projected to grow 5.5% annually on average in between 2017 and 2026 and to represent 19.7% of the economy in 2026, according to a report in the February issue of Health Affairs.
Projected national health spending and enrollment growth over the next decade is largely driven by fundamental economic and demographic factors: changes in projected income growth, increases in prices for medical goods and services, and enrollment shifts from private health insurance to Medicare that are related to the aging of the population.
The recent enactment of tax legislation that eliminated the individual mandate is expected to result in only a small reduction to insurance coverage trends.(PV)
~~~~~~~~~~~~~~~~~~~~~~~~~
Awards
ERT Recognized for Advanced Central Trial Analytics and Patient Engagement
ERT’s clinical trial technology solutions have been shortlisted for two Informa Clinical and Research Excellence (CARE) Awards. Informa, a business intelligence and academic publishing company, conducts the annual CARE Awards to showcase significant accomplishments and highlight innovation across the industry. ERT is being recognized in the category of Best Sponsor-focused Clinical Development for its Centralized Clinical Trial Analytics Solution and in the category of Best Patient-focused Clinical Development for its Voice Assistance Data Capture solution.
Johnson & Johnson Named to Fortune’s Most Admired List
Johnson & Johnson as been named to Fortune’s Most Admired Company list, making this the 16th year the company has appeared in the top 20. The company is being recognized for its commitment to employees, innovation, and the health and well-being of people around the world. Johnson & Johnson also placed No. 1 in the pharmaceutical category worldwide for the fifth consecutive year — making it the highest-ranked healthcare company to appear on the top companies list in 2018.
Klick Earns Best Workplace for Women Distinction
Klick was named a Best Workplace for Women for the third consecutive year after thorough and independent analysis conducted by Great Place to Work.
“We are proud of our legacy as a Best Workplace for Women and stand alongside other leaders and companies that are committed to ensuring more progress is made on diversity, inclusion, and equality at work," says Klick President Lori Grant, a recipient of Women’s Executive Network’s (WXN) Top 100 Most Powerful Women. Ms. Grant is also a PharmaVOICE 100 honoree — 2016.
Parexel Receives
Two Industry Recognitions
Parexel International is being recognized for its diverse and flexible workplace by Forbes and FlexJobs. The company has been named to Forbes’ new America’s Best Employers for Diversity list, which recognizes the top 250
employers across all industries. The company was also recognized by FlexJobs as a Top 100 Company to Watch for Remote Jobs in 2018, its fifth consecutive time on the list. The list recognizes companies that are remote-friendly, allowing for employees to telecommute either on a full- or part-time basis.
Pharma Companies Named
Top for Executive Women
Bristol-Myers Squibb and Roche Diagnostics have been named as 2018 NAFE Top Companies for Executive Women by the National Association for Female Executives. This honor recognizes American corporations where women have significant clout to make the decisions that affect their companies’ futures and bottom lines.
“We’re very proud of the progress we’ve made over the last several years and we will continue to strive for even more representation of women at all levels of the company through additional training, mentoring and sponsorship opportunities," says Bridget Boyle, VP and site head, human resources, Roche Diagnostics.
PPD Recognized for
Employee Development
Pharmaceutical Product Development (PPD) has been honored for employee learning and development programs that help the global CRO deliver service excellence to its pharmaceutical and biotechnology clients. For the seventh consecutive year, Training magazine named PPD to its Training Top 125 list of businesses that excel in employee learning and development.
Symphony Clinical Research Wins BIG Innovation Award
Symphony Clinical Research has been named a winner in the 2018 BIG Innovation Awards presented by the Business Intelligence Group.
~~~~~~~~~~~~~~~~~~~~~~~~~
PV 100 News
DrugDev Promotes Elisa Cascade
 Elisa Cascade has been promoted to chief product officer at DrugDev. She is responsible for the overall development and growth of the DrugDev Spark clinical operations suite.
Elisa Cascade has been promoted to chief product officer at DrugDev. She is responsible for the overall development and growth of the DrugDev Spark clinical operations suite.
In addition, Ms. Cascade served as architect of the DrugDev Golden Number, the industry’s universal identifier for global site facilities and personnel used by the Investigator Databank, the TransCelerate Investigator Registry, and leading pharmaceutical companies and CROs.
Ms. Cascade is a PharmaVOICE 100 honoree — 2016.
Michael Parisi Named CEO at Guidemark Health
 Guidemark Health, a healthcare communications agency, has appointed Michael Parisi as CEO. Mr. Parisi is a 27-year healthcare communications veteran. In this new role, Mr. Parisi is responsible for all Guidemark Health offices and is leading the agency’s advertising/marketing/communications, medical communications, and learning businesses.
Guidemark Health, a healthcare communications agency, has appointed Michael Parisi as CEO. Mr. Parisi is a 27-year healthcare communications veteran. In this new role, Mr. Parisi is responsible for all Guidemark Health offices and is leading the agency’s advertising/marketing/communications, medical communications, and learning businesses.
Mr. Parisi was a managing partner at Ogilvy CommonHealth Worldwide. He joined Ogilvy in 2000 and steadily earned increasing leadership roles and responsibility.
He sits on several nonprofit boards and currently serves as board president for CancerCare, a national organization that provides free professional support for people with cancer.
Mr. Parisi is a PharmaVOICE 100 honoree — 2012.
~~~~~~~~~~~~~~~~~~~~~~~~~
Innovation Corner
Amgen Provides Golden Tickets to LabCentral
Amgen and LabCentral have chosen Kernal Biologics and QurAlis to receive Amgen Golden Tickets at LabCentral. The Golden Ticket represents one year of bench space for one scientist, including benefit of LabCentral’s shared infrastructure and services. Amgen scientists also provide informal mentoring. LabCentral is a shared laboratory space designed as a launchpad for high-potential life sciences and biotech startups.
Astellas Oncology Awards $100,000 in C3 Prize
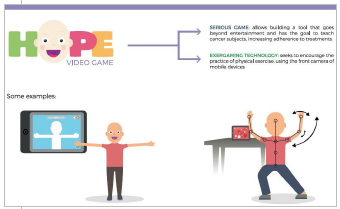 Astellas Oncology has named Hernâni Oliveira of Porto, Portugal, the Grand Prize Winner of the second annual C3 Prize, a global challenge that acknowledges and supports nonmedicine innovations aimed at improving the cancer care experience for patients, caregivers, and their loved ones.
Astellas Oncology has named Hernâni Oliveira of Porto, Portugal, the Grand Prize Winner of the second annual C3 Prize, a global challenge that acknowledges and supports nonmedicine innovations aimed at improving the cancer care experience for patients, caregivers, and their loved ones.
Mr. Oliveira received $50,000 for his entry, Hope Project, which consists of a two-part app developed to help pediatric cancer patients and their parents solve issues related to medication adherence and the sedentary lifestyle of children who are diagnosed with cancer, as well as help parents further understand and explain complex cancer treatment procedures. Four runners-up each receive a $12,500 grant.
Sage Bionetworks Announces Winners in Digital Biomarker Challenge
Sage Bionetworks, in collaboration with The Michael J. Fox Foundation, has announced the results of the Parkinson’s Disease Digital Biomarker DREAM challenge, an open crowd-sourced research project designed to benchmark the use of remote sensors to diagnose and track Parkinson’s disease. The first sub-challenge was won by Yuanfang Guan and Marlena Duda from the University of Michigan, Ann Arbor, who developed a deep learning convolutional neural network.
In the second sub-challenge, three different groups won for their features predicting different Parkinson’s symptoms. Bálint Ármin Pataki from Eötvös Loránd University in Hungary took the honors in building features for tremor severity.
Jennifer Schaff from Elder Research, used statistical methods to derive features and feature selection to develop the top performing submission in predicting dyskinesia severity. Team Vision from Columbia University consisting of Yuanjia Wang and Ming Sun used spectral decomposition to build features that outperformed all other teams in predicting bradykinesia.
Takeda and the New York Academy of Sciences Announce Innovators in Science Award
 Takeda Pharmaceutical Company and the New York Academy of Sciences have named the honorees of the inaugural Innovators in Science Award for their commitment to and excellence in neuroscience research.
Takeda Pharmaceutical Company and the New York Academy of Sciences have named the honorees of the inaugural Innovators in Science Award for their commitment to and excellence in neuroscience research.
The winner of the Senior Scientist Award is Shigetada Nakanishi, M.D., Ph.D., director, Suntory Foundation for Life Sciences Bioorganic Research Institute in Japan. Dr. Nakanishi is developing cloning strategies for membrane embedded transmitter receptors and subsequent identification of functional genes encoding NMDA and G-protein coupled glutamate receptors.
The winner of the Early-Career Scientist Award is Viviana Gradinaru, Ph.D., assistant professor of biology and biological engineering at the California Institute of Technology. Dr. Gradinaru is working to develop novel tools for neuroscience and using them to probe circuits underlying locomotion, reward, and sleep.
Also recognized for discoveries ranging from neural mechanisms underlying cognitive function and emotional and social behaviors to the role of astrocytes at synapses in health and disease, and ion channels that enable somatosensation and pain perception are Senior Scientist Finalists: Ben Barres, M.D., Ph.D., professor of neurobiology, Stanford University School of Medicine and David Julius, Ph.D., professor and chair of physiology, UC San Francisco. Michael Halassa, M.D., Ph.D., (pictured) assistant professor of neuroscience, New York University, and Kay Tye, Ph.D., Whitehead Career Development assistant professor of neuroscience, as early-career scientist finalists.
~~~~~~~~~~~~~~~~~~~~~~~~~
eBook
Reducing the Burden of Data Collection in Oncology Trials
Provided by: CRF Health
Podcast Plus Video
Death of the Campaign Factory: Why Advertising Agencies Must Evolve
Sponsored by: precisioneffect
Webinars
Driving Improved Access and Compliance Through Direct-to-Physician Sampling
Sponsored by: Cardinal Health 3PL
Improving Patient Engagement Through Design Psychology Principles
Sponsored by: Mad*Pow
How to Challenge the Status Quo in Highly Competitive Markets
Sponsored by: Quantum Learning
White Papers
Breaking Through the Barrier: Accessing Hard To Reach Physicians
Provided by: BulletinHealthcare



















