Using Electric Fields to Treat Cancer
Bill Doyle, Executive Chairman of Novocure, talks about the company’s treatment that uses electric fields to treat glioblastoma, a deadly brain cancer, and other cancers.
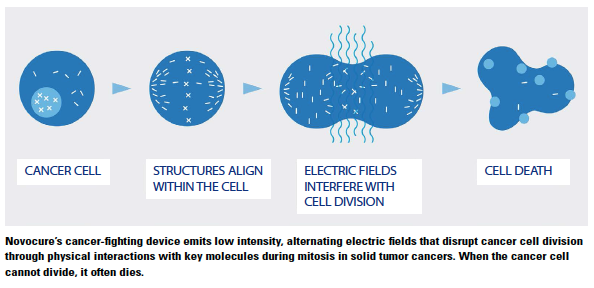
 Novocure’s approach to treating cancer may look like pseudo science, but the results are real. Novocure is using low intensity, alternating electric fields to disrupt cell division with no known systemic side effects to treat solid tumor cancers.
Novocure’s approach to treating cancer may look like pseudo science, but the results are real. Novocure is using low intensity, alternating electric fields to disrupt cell division with no known systemic side effects to treat solid tumor cancers.
In 2000, founder Professor Yoram Palti sought to leverage his expertise in biophysics to develop a new way to treat cancer. The result of this research has created a different way to treat cancer: Tumor Treating Fields (TTFields), the use of electric fields tuned to specific frequencies to disrupt solid tumor cancer cell division.
An electric field is analogous to the gravitational field that holds us to the earth’s surface, explains Bill Doyle, executive chairman of Novocure.
“We can’t see it or touch it, but we know that it’s what is keeping us in our chair instead of floating up into space," he says. “We also have a good sense for what a magnetic field is — the invisible force field that will pull a plate with a positive charge and a particle with a negative charge together.
An electric field can impact the proteins involved in cell division. These proteins make up the spindle apparatus that allows for DNA to be split in cell division, and when this happens it creates an electric charge.
Novocure’s product, Optune, is a wearable, portable device that delivers these electric fields to the location of tumors. It includes adhesive patches, called transducer arrays, which are connected to a battery. Optune is approved for the treatment of adult patients with glioblastoma, the most common and deadliest type of primary brain cancer. It was approved in October 2015 for first-line glioblastoma and in 2011 for recurrent glioblastoma.
Because the electric field does not enter the bloodstream like a drug, it does not cause many of the side effects of chemotherapy, such as infections, nausea, vomiting, diarrhea, and lowered blood counts. The most common adverse events associated with Optune were scalp irritation and headache.
For patients with newly diagnosed glioblastoma, Optune is used with the chemotherapy temozolomide (TMZ) after surgery and during radiation with TMZ. In a large clinical study, Optune demonstrated a five-year survival advantage. Analyses showed 2.5 times greater survival at five years with Optune plus TMZ (13%) compared with TMZ alone (5%). Nearly half, or 43%, of the people who used Optune with TMZ were alive at two years or longer, compared with 31% who were on TMZ alone.
“We’ve had to do a tremendous amount of fundamental science to provide data so people can wrap their heads around how the technology works," Mr. Doyle says. “It’s different from immunotherapy, which people have some understanding. In our case, we have to start with the basic physics. At every step of the way we had to invent new techniques and technologies to conduct the science and then bring it to market."
As of June 2017, there were 1,460 active patients worldwide using Optune. Of these, 1,083 are in the United States.
The Optune therapy is regulated as a device, and the company used the PMA process through the Center for Devices and Radiological Health. Because this was a new type of therapy when Optune received its approval, the agency required a tremendous amount of data.
 In May, the delivery system received a humanitarian use device (HUD) designation from the FDA for treatment of pleural mesothelioma, an initial step toward a Humanitarian Device Exemption (HDE) approval in the United States.
In May, the delivery system received a humanitarian use device (HUD) designation from the FDA for treatment of pleural mesothelioma, an initial step toward a Humanitarian Device Exemption (HDE) approval in the United States.
Mr. Doyle says over the last 15 years the company made a strategic decision to do more studies than would be needed to show that the science is real.
Additionally, Novocure provides the therapy free to Medicare beneficiaries until the time the government approves coverage. In Germany, Optune reimbursement is on a case-by-case basis, and the company is working with government payers in the United States, Switzerland, and Japan. In August, Novocure received reimbursement in Austria.
“We’re not denying access to anyone in the United States," Mr. Doyle says.
He says the company is doing trials to investigate Optune with other chemotherapies to determine if survival rates can be increased. Novocure has a collaboration with Celgene to study marizomib and temozolomide in combination with Optune for the treatment of glioblastoma.
“We’re also doing a lot of work to combine Optune with radiation and there’s strong preclinical data to suggest when used with radiation therapy the results would be even better," he says.
Novocure has ongoing or completed clinical trials investigating TTFields in brain metastases, non-small cell lung cancer, pancreatic cancer, ovarian cancer, and mesothelioma.
The company plans to begin a Phase III pivotal trial in locally advanced pancreatic cancer in the second half of 2017. A Phase II trial found median progression free survival in the TTFields-treated group was 12.7 months compared with 5.5 months in nab-paclitaxel plus gemcitabine historical controls. Pancreatic cancer is the fourth-leading cause of cancer death in the United States; five-year survival among patients with metastatic pancreatic cancer is 2%.
The company also plans to begin a Phase III pivotal trial of Optune in recurrent ovarian cancer in 2018. Novocure is also conducting Phase III trials of Optune in brain metastases and non-small cell lung cancer, as well as a Phase II trial in mesothelioma. (PV)



















