Clinical study sites are transforming their technology infrastructures with the adoption of electronic medical records (EMRs). But while EMRs improve treatment-related tasks in healthcare settings, they have not helped to reduce the burden of clinical trial studies on the sites.
 A May 2017 survey shows that study sites remain burdened with mundane administrative tasks, such as creating and then transcribing paper source forms into electronic data capture (EDC) during study execution. The survey was conducted by the Society for Clinical Research Sites, CenterWatch, and Clinical Ink.
A May 2017 survey shows that study sites remain burdened with mundane administrative tasks, such as creating and then transcribing paper source forms into electronic data capture (EDC) during study execution. The survey was conducted by the Society for Clinical Research Sites, CenterWatch, and Clinical Ink.
The survey found that while EMR adoption rates at sites are becoming more prominent with 64% of study sites adopting EMRs, EMRs had minimal impact on reducing clinical trial site burdens, as 90% of sites still create study-specific source forms. Of those sites, 96% still use paper-based approaches when creating source forms and collecting source data.
Additionally, sites that have adopted EMR technology rely on it to access information for clinical trials for confirmation of inclusion/exclusion criteria, medications, medical history, and detecting adverse events. Paper source forms are also created to ensure that all study data are captured.
Even for sites that do have EMR systems, there is no connection between EMR and EDC systems used for clinical trials.
Sites reported that EMR inaccuracies are a major issue that drives sites toward collecting study related information onto paper, as well as the convenience of having the forms with them to transcribe directly onto, says Christine Pierre, president, Society of Clinical Research Sites.
“As we know, EMRs were developed to meet very specific market drivers, and research was not the main consideration in how they were designed," she says.
 There is still a lot of reliance on paper given the limited technology options currently available to sites, says Jason Tibbs, senior VP of operations, general partner, PRA Health Sciences.
There is still a lot of reliance on paper given the limited technology options currently available to sites, says Jason Tibbs, senior VP of operations, general partner, PRA Health Sciences.
“There are a number of challenges," he says. “Certainly there’s the high cost of implementation of some of the systems, both financial and from a resource perspective for sites. Many of the sites that we work with, especially some of the smaller ones, lack the infrastructure or the staff support to implement technology solutions. Many sites also have a desire for one system that checks all of the boxes and are waiting for technology that is all-inclusive. And like in any industry, there’s a general hesitancy to change and a fear of changing what works."
Many sites aren’t seeing an ROI on technology adoption because they have no say in the process, says Nicholas Slack, president of ePharmaSolutions (ePS) and chief growth officer at WIRB Copernicus Group.
“Given the gravity of the sites’ mission and the rather meager profit that sites generate, their top priority certainly isn’t adopting technology to collect data," he says. “Sites will proactively adopt technology when solutions support, rather than hinder, them.
Solutions must be affordable, must give them more time to help patients, allowing them to better manage their patients’ experiences, and sponsors and CROs need to limit how many different technologies they throw at them."
Mr. Slack says many sponsors and CROs haven’t chosen site-friendly technology solutions that make the work flow easier for sites and their patients.
“The sponsors and CROs that do equip sites often don’t provide the proper support and onboarding of those technologies," he says. “Lastly, many of the companies developing technologies are focused only on the sponsor and CRO experience but not necessarily the site experience. Sites are on an island disconnected from the process."
“We’ve seen an 18% increase in conversion from paper to electronic clinical outcome assessment (COA) data collection over the 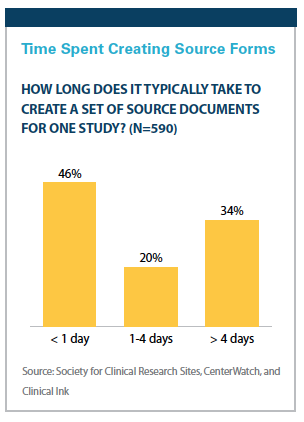 last few years, which reflects the enhanced value that electronic offers over paper methods," says Ron Sullivan, executive VP of eCOA, ERT. “Although some clinical sites continue to use paper for clinical outcomes reporting, we expect eCOA usage to continue increasing as sponsors and site personnel recognize its advantages and become more comfortable embracing eCOA technology during clinical development."
last few years, which reflects the enhanced value that electronic offers over paper methods," says Ron Sullivan, executive VP of eCOA, ERT. “Although some clinical sites continue to use paper for clinical outcomes reporting, we expect eCOA usage to continue increasing as sponsors and site personnel recognize its advantages and become more comfortable embracing eCOA technology during clinical development."
There are several reasons why this is, he says. First, it takes time for sponsors to recommend or mandate digitalization and to modify their clinical processes. Second, the transition from paper to electronic requires clinical site personnel to adapt and embrace new ways of collecting data — and both movements are currently under way.
Jim Murphy, CEO, Greenphire, points out that very few academic clinical institutions do only clinical trials.
“They’re first and foremost treating patients and then they have this investigative element of their mission where they’re trying to advance human health in some way," he says. “The patients participating in clinical studies would only represent a very small percentage of the total patients they see."
What Sites Need
A survey in 2016 by CenterWatch found that less than 10% of investigative sites believe that clinical trial technology solutions provided by sponsors and CROs are meeting their operating needs.
Investigative sites worldwide are being inundated with a growing number of technology solutions that are difficult to use and are not compatible, the CenterWatch survey uncovered. On average, the typical investigative site is working with 12 different systems to collect clinical research data. The majority of investigative sites feel strongly that there are too many user names and passwords that they must manage.
From a site’s perspective, there is inefficiency because of duplicative systems that sites have to use for each clinical trial.
Kim Potts, manager of clinical operations at PMG Research, an integrated network of clinical research facilities, says for each  pharmaceutical company that PMG conducts a trial for, there are different systems for EDC, IVRS systems, electronic diaries, and EKG.
pharmaceutical company that PMG conducts a trial for, there are different systems for EDC, IVRS systems, electronic diaries, and EKG.
“There are even multiple systems that require training for each study for not only the coordinators, but also for the investigators, which makes it time consuming for the investigators," she says. “It’s redundant training but it’s required for this trial for this sponsor."
Sites also are required to use different portals for each study for lab and safety reports.
Mr. Murphy acknowledges that different clinical trials use different technologies because there are so many different options available.
“We need to develop and apply technology specifically for the benefit of the site, not just for the benefit of the sponsor’s goals," he says. “There is an opportunity for vendors to look for opportunities to make things a little simpler for the site. For example, allowing an investigative site or a representative for an investigative site to have one user ID and password that he or she can use across all of the clinical studies that are being done regardless of the sponsor or organization would simplify things."
Mr. Murphy says implementing technologies to streamline payment processes is another way to eliminate burdens for sites, by not only saving them money, but delivering more time for them to focus on the research. With an average savings of 20 minutes per patient per visit, the benefits can quickly become significant.
Additionally, double data entering — having to record information for source documentation and into EDC systems — is a huge burden on sites.
“Time is the biggest burden in collecting data," Ms. Pierre says. “With 90% of sites reporting that they create paper source documents, 54% spend more than a day for every study creating source documents and 34% spend more than four days. The cost of $800 to $1,000 per study to create those forms doesn’t capture that at the same time study patients aren’t being seen and the site isn’t collecting revenue."
Ms. Pierre says 85% of all sites reported it would be helpful or extremely helpful and more than adequate as a form of documentation if data were entered directly onto an electronic case report form.
“Not only would it save time for sites, but also would reduce the number of queries and time spent by all, because in many instances queries are generated by transcription errors," she says.
 Pharma still wants source verification to mirror whatever is in the EDC, says Tonya Ward-Kiser, senior director of operations at PMG Research.
Pharma still wants source verification to mirror whatever is in the EDC, says Tonya Ward-Kiser, senior director of operations at PMG Research.
“If we’re going to input this information into a source document then it needs to be on iPads or tablets, something that could be mobile and be carried in the room with the patient volunteers," she says.
Ms. Ward-Kiser says coordinators would be more effective and efficient if there was a single system for all studies.
“When studies weren’t so complex, coordinators spent 75% of their time seeing patients," she says. “Now 25% of their time is seeing patients and the other 75% of the time is spent working with multiple EDC systems, multiple IVRS, multiple lab and IRB systems. Enrollment takes a backseat to trying to meet all the timelines with all of these different portals and systems."
What is needed from a site perspective, Ms. Ward-Kiser says, is a true eSource solution that links to EDC and other systems that house eConsent and portals for lab and safety reports.
“This would streamline the whole process, not only for sites, but for pharma as well," she says.
Mr. Tibbs says site start up is another area of challenge.
“Our vision is to leverage technology to aggressively cut timelines and to do so by recreating an electronic experience of site startup," he says. “We estimate that about 75% of the information that’s needed on these various forms that we ask as part of start up to be duplicated. Our goal, from a technology perspective, is to look at the opportunity to create site profiles electronically so that we can pre-populate these forms and expedite the whole process in an electronic environment."
Ms. Pierre says sponsors and CROs can look at how they can use their economy-of-scale to foster the development of technology that helps sites.
“For instance, there are many options sponsors and CROs have that could provide a variety of technology services to ease the sites’ burden from central repository of regulatory documents, contract negotiation, and site payments just to name a few," she says.
“Most sites have less than three months of operating cash — they’re not going to be able to make the capital expenditures required to make these technologies a reality at their site. However, when sponsors and CROs can bring efficiencies in with the study, that’s when these technologies can be used to improve the entire process of clinical trials, and ultimately serve the patient better."
The industry needs to start looking at the site as a major stakeholder in the process, Mr. Slack says.
“Sites are essentially forced to adopt a new technology for each study they are selected for," he says. “In clinical research, sites have very little control over the technology they adopt. They need to be brought into the process. They can’t continue to have technology pushed down on them such that they are essentially stuck with it regardless of how little it fits their operating model."
 If sites are not brought into the process, costs of trials are going to continue to soar because adoption of the right tools will continue to lag, Mr. Slack says.
If sites are not brought into the process, costs of trials are going to continue to soar because adoption of the right tools will continue to lag, Mr. Slack says.
“Trial timelines won’t meaningfully improve," he says.
Mr. Tibbs agrees, adding: “If we aren’t able to partner with sites to get them to adopt technology to a greater degree, then we limit our ability to optimize the efforts to expedite trials and trial timelines. That’s only as productive as getting the sites to use the technology in the first place."
Data Integration
Study sites have been working to align their own infrastructures to align with healthcare initiatives, specifically as a result of the 2009 Obama American Recovery and Reinvestment Act. This incentivized healthcare organizations to adopt EMR technology.
In fact, the Society for Clinical Research Sites, CenterWatch, and Clinical Ink survey found that 64% of sites have adopted EMRs, and the academic and hospital-based have the highest rates of adoption.
But there is a gap between what EMRs currently offer and what is needed for clinical research. At the same time, the FDA has suggested the industry move to electronic source data as a way to enhance trial efficiency.
Some companies are working to bridge that gap between what EMRs currently offer and what research sites need. Integrated technology solutions would provide an opportunity to increase efficiency at the site and also to improve communication between sites and sponsors and CROs.
 Mr. Tibbs says an integrated clinical trials solution is not yet available, but there are options today that incorporate “bolt-on" technologies.
Mr. Tibbs says an integrated clinical trials solution is not yet available, but there are options today that incorporate “bolt-on" technologies.
“We recognize that system integration is an area of need for sites," he says. “About a year and half ago, we acquired Nextrials, a company that brings an E2E software solution technology for exactly that, for working with electronic health records and being able to integrate them into the EDC from multiple different types of site medical record platforms."
“The ability to integrate not only reduces the workload for sites, but it clearly decreases the number and duration of site monitoring visits and generates faster, cleaner data, and provides significant cost-saving abilities, bypassing the need for SDV, which is traditionally one of the biggest expense areas of our study budgets," he says. “In future states, I see an opportunity to avoid the need for EDC altogether and go straight from an electronic health record environment into a data source system and have the insights and analytical decision-making done on that data warehouse."
Mr. Sullivan says a central portal for clinical trials is a vision that is realistic but not feasible today.
“We have to make sure that sponsors would buy in to this type of approach, so there are some barriers there," he says. “The industry will need to come together to focus on sites. I’d like to see a couple of industry groups get together and collectively work to make it easy on the sites to be able to collect the data. There would have to be some agreement at the sponsor level to take all of their studies and put them onto one central location."(PV)



















