Patients with end-stage renal disease need life-sustaining dialysis, an artificial process that removes waste and water from the blood. Dialysis, however, often requires access to blood vessels either through fistulas — a connection between the vein and artery — grafts, or catheters, which can lead to infection and other complications.
 Humacyte, a privately held biotech company, has developed and is doing clinical research on a new way to provide vascular access for patients with end-stage renal disease. Humacyl is a bioengineered blood vessel that is being evaluated as a potential alternative to synthetic polytetrafluoroethylene (PTFE) grafts in hemodialysis patients who are not candidates for fistula placement.
Humacyte, a privately held biotech company, has developed and is doing clinical research on a new way to provide vascular access for patients with end-stage renal disease. Humacyl is a bioengineered blood vessel that is being evaluated as a potential alternative to synthetic polytetrafluoroethylene (PTFE) grafts in hemodialysis patients who are not candidates for fistula placement.
Humacyte’s technology platform isolates and grows human acellular vessels (HAV) that have the potential to provide stable, “off-the-shelf" human tissue replacements. The company is researching several applications, with the first being in vascular access for dialysis.
In May 2016, Humacyte began a Phase III trial to compare Humacyl with ePTFE grafts, the current standard of care. The trial is being conducted at about 35 sites in the United States, Europe, and Israel with 350 patients — the largest study of any bioengineered vascular tissue to date. Humacyl received fast track status from the FDA in 2014.
Jeffrey Lawson, M.D., Ph.D., chief medical officer, of Humacyte, says the base technology is formed initially with a biodegradable mesh scaffold.
“The mesh looks like the sleeve of a sweater," he says. “The mesh is formed in a tube that’s the size of the blood vessel we want to reproduce.
Then in a closed bioreactor chamber, we fill in around the mesh with cell culture media, and we seed vascular cells into the meshwork."
As the cells begin to grow in the meshwork, the mesh melts away. By the time the mesh melts, all of the cells have stuck together to form a blood vessel. Then the blood vessel is put through a process called decellularization, which kills all of the live cells and then washes them away.
 “The cells have antigens that the immune system would recognize, and the body might reject the mesh like a transplant," Dr. Lawson explains.
“The cells have antigens that the immune system would recognize, and the body might reject the mesh like a transplant," Dr. Lawson explains.
“When we kill all the cells, what is left behind is the scaffold, which we call a human extracellular matrix. This is the glue that is between cells in all parts of the body. In this case, it’s the glue that was made by vascular cells that are used in making blood vessels."
This process of decellularization creates a protein structure that is comprised of human types I and III collagen, which does not trigger an immune response. The antigenic property is an important safety benchmark for the Phase II clinical trials and is something the company will be monitoring in Phase III trials.
“To date, we have now implanted more than a 100 vessels in more than 100 individuals in both our Phase II and Phase III programs, and we have not seen any signs of rejection or what we would call immune recognition," Dr. Lawson says.
Preclinical and clinical data suggest Humacyte’s blood vessels may have the potential to extend patency — the ability of blood vessels to remain open and functional over time — with the possibility of some reduction in inflammation and some reduction in clotting.
In May 2016, the company published findings from a Phase II study of hemodialysis in The Lancet that showed patients had very good enduring patency. The trial was not a head-to-head comparison, but based on comparisons in the literature, acellular vessels appear to last significantly longer than a conventional dialysis or plastic dialysis graft, have almost no infections, and no sign of rejection.This could be a transformative technology.
“If we can provide someone with a durable blood vessel for long-term use that has a low chance of becoming infected, this will certainly make his or her life better," he says. “We eventually want to help all dialysis patients."
The company is also looking at military applicability.
“If we are able to develop a blood vessel that a military surgeon can use immediately in the battlefield for an injured soldier to save a limb, for 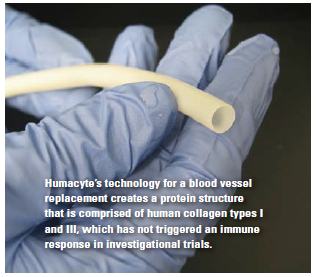 example, this would be meaningful to me personally," Dr. Lawson says. “Someday, we hope to have blood vessels that a surgeon can use to reconstruct vessels anywhere in the body. We are working on the fundamental technologies that can be used to make other more complex tissues well into the future.
example, this would be meaningful to me personally," Dr. Lawson says. “Someday, we hope to have blood vessels that a surgeon can use to reconstruct vessels anywhere in the body. We are working on the fundamental technologies that can be used to make other more complex tissues well into the future.
“If our researchers are successful with the remainder of our clinical development program, at some point thereafter, we could start looking at potential therapies that impact lower extremity vascular disease and arterial trauma," Dr. Lawson concludes.
In October 2015, the company raised $150 million committed Series B preferred stock financing, which is being used to fund the Phase III clinical trial. (PV)




















