 The pharmaceutical industry faces major challenges; patent expiry, generic competition, weak pipelines, spiralling R&D costs, high attrition rates, and falling drug approval rates. In response, the drug development landscape is set for major changes in the coming five to 10 years.
The pharmaceutical industry faces major challenges; patent expiry, generic competition, weak pipelines, spiralling R&D costs, high attrition rates, and falling drug approval rates. In response, the drug development landscape is set for major changes in the coming five to 10 years.
Most pharmaceutical companies, and smaller biotech companies, have already embraced a biomarker approach to drug development, particularly in areas such as oncology, cardiovascular, metabolic, infectious, and inflammatory diseases.
Both the FDA, with its critical path initiative (CPI), and the NIH with its biomarkers consortium foundation, recognize the importance of this area of research and support and encourage the need to continuously develop and validate new and advanced biomarkers, utilizing new platforms and tools that can be used not only as surrogate endpoints for registration approvals, but also for internal decision making during drug development.
It’s clear that the continuing changes in how we develop drugs will see a marked evolution in the role of biomarkers and pave the way for an increase in companion diagnostics (CDX).
A True Global Delivery of Service
 In 2013, the FDA published, “Paving the way for personalized medicine," where it outlined its thoughts on the importance of biomarkers and the role this new and evolving technologies will play, whilst stating the need for a quality approach, standardization, and regulatory control. To achieve success, requires a balance between harnessing new innovations, platforms, and techniques, and delivering a quality-driven and consistent truly global delivery of service. This is set to be one of the most significant challenges the pharmaceutical and CRO industry faces as it sets out to work in partnership to achieve the common goal of developing smarter and more personalized medicines.
In 2013, the FDA published, “Paving the way for personalized medicine," where it outlined its thoughts on the importance of biomarkers and the role this new and evolving technologies will play, whilst stating the need for a quality approach, standardization, and regulatory control. To achieve success, requires a balance between harnessing new innovations, platforms, and techniques, and delivering a quality-driven and consistent truly global delivery of service. This is set to be one of the most significant challenges the pharmaceutical and CRO industry faces as it sets out to work in partnership to achieve the common goal of developing smarter and more personalized medicines.
Recent feedback from pharmaceutical clients indicates that currently “a very fragmented, inflexible biomarker service offering is provided from the large, (CROs) and global central labs." Numerous, academic, specialist, and niche providers are available to bolster the resources available, but they are often not global, or capable of scaling up beyond Phase I. Furthermore, many do not have the quality resources to operate to GCP or have CAP/CLIA-based quality standards in place. This fragmented biomarker service offering is sub optimum, requires significant resource, and proves expensive as clinical teams have to individually approach, qualify, and contract with multiple niche or academic vendors to fulfill their biomarker and development portfolio needs. A review of the current transactional and niche approaches offered by CROs is presented, see Table — A Transactional Approach to Biomarker Services Supporting Drug Development Projects.
The Growing Market for Biomarkers
According to published research , the current global biomarker discovery outsourcing service market is about $2.7 billion, and the current global companion diagnostic (CDX) outsourcing market is about $3.5 billion. To meet this demand pharmaceutical companies will increasingly come to largely rely on external resources, primarily through collaboration and/or partnership.
Many drug companies have also established separate departments to specifically focus on the management of R&D involving biomarker and CDX as the management of the discovery of biomarker and development of CDX services requires a totally different expertise and technologies than traditional drug R&D.
Current Biomarker Services Offered to the Pharmaceutical Industry
The diversity and range of specificity of biomarkers are such that no one biomarker, technique, or platform is likely to have all the characteristics necessary to provide a detailed and robust understanding of response. In this regard, research providers have responded by offering an extensive range of tactical or single-point solutions to meet a specific need as we develop drugs.
(Examples are presented in Table — A Transactional Approach to Biomarker Services Supporting Drug Development Projects.)
This transactional approach provides access to niche, or best-in-class academic services, and allows project teams to cherry pick best-in-class providers and thought leaders. However, the project teams are then faced with contracting and managing the data from multiple providers.
We have seen recent examples where up to six different biomarker vendors in four countries were utilized.
Therapeutic Focus and Clinical Input to Biomarker, IVD, and CDX Development
The identification, development, validation, and, ultimately incorporation of biomarkers into clinical practice is critically 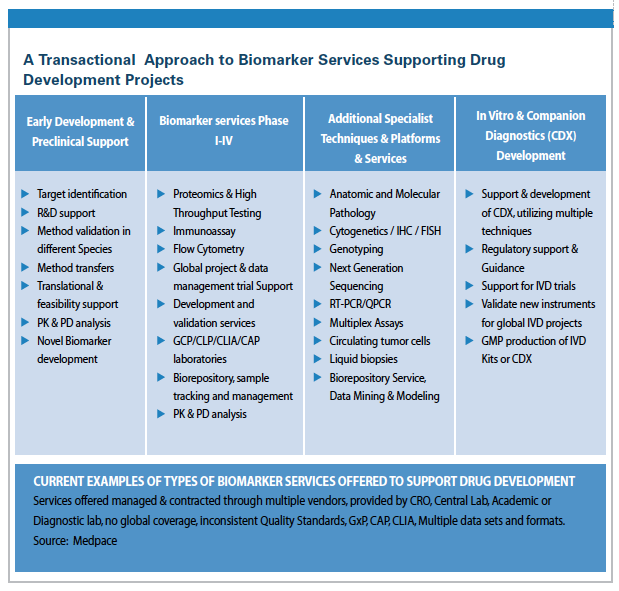 important to a number of disease areas. Given the complexity of a disease such as cancer, the importance of biomarkers is particularly relevant. A number of biological, chemical, and biophysical entities are in development that can be used in cancer diagnosis, prognosis, risk group assessment and treatment stratification, and predicting and monitoring response to therapy.
important to a number of disease areas. Given the complexity of a disease such as cancer, the importance of biomarkers is particularly relevant. A number of biological, chemical, and biophysical entities are in development that can be used in cancer diagnosis, prognosis, risk group assessment and treatment stratification, and predicting and monitoring response to therapy.
While a significant focus is on genomics, biomarkers are also being developed in the areas of proteomics, immunologic metabolomics, and epigenetics.
This approach has already shown significant success with incorporation into clinical practice for a number of different cancers, including the use of biomarkers in patients with chronic myelogenous leukemia (CML) (BCR-ABL), breast and ovarian cancer (BRCA1/BRCA2), breast cancer (HER-2), pancreatic cancer (CA19.9), colon cancer (CEA), and prostate cancer (PSA). Given the current focus on increasing precise and increasingly personalized medicine, clinical trials in cancer now almost universally include the study of biomarkers as secondary and exploratory objectives.
The use of CROs that have an integrated approach to the execution of these complex studies will greatly enhance the probability of successfully completing these important studies.
The Importance of Quality and Technical Guidance at an Early Stage
The use of biomarkers will facilitate the availability of safer and more effective drugs to guide dose selection and to enhance their benefit-risk profile. The FDA states: “Qualification is a conclusion that, within the stated context of use, the results of assessment with a biomarker can be relied upon to adequately reflect a biological process, response, or event and support use of the biomarker during drug or biotechnology product development, ranging from discovery through post approval."
The most valuable and useful biomarkers will be suitably qualified, robust, easy to collect, non-invasive, with adequate stability to ensure they can be processed, transported, and analyzed whilst delivering accurate and reliable data. The importance of experienced technical oversight operating in a GCP/CAP/CLIA-based global quality systems are essential and the value of these benefits should not be overlooked.
The Value of Consistent Global Project and Data Management Services Supporting Biomarker Clinical Trials
The role of the biomarker project manager is crucial to the trial’s success. He or she balances all facets of the project, including communication with biomarker outsourcing managers and clinical teams. They support, coordinate, and track assay development and validation timelines, ensuring supplies and logistics are in place to make sure that the integration and delivery of data occurs on time. However, when the flexibility, consistency, and level of customer service offered from the project managers is suboptimal, it leads to significant clinical project team dissatisfaction, causes delays, budget overburns, and is frequently cited as a source of significant dissatisfaction with vendors.
The Future of Biomarkers
We suggest that the future use of combinations of multiple biomarkers using different techniques and platforms will be the way forward in order to enable improved prediction of drug efficacy and safety. The use of such combinations of biomarkers and service providers may bring its own challenges, including technical issues on how to combine results and different data sets, how to control quality, and most importantly, how to interpret results in different clinical contexts. However, this approach is gaining favor with the pharmaceutical industry as companies seek to gain greater value for money whilst using best-in-class services to accelerate their drug development programs.
This collaboration or modular approach is a hybrid of the functional service model (FSP) that has emerged recently and the full service model (FSM) traditionally offered by the large CRO service providers. (PV)
Medpace is a global full-service clinical research organization (CRO) providing Phase I-IV core development services for drug, biologic, and device programs.
For more information, visit medpace.com.



















