“Let’s talk."
That’s what we said to a cross-section of health care payers to gauge how they felt about using evidence from patient reported outcome surveys (PROs) when making coverage decisions for pharmaceuticals. The input we received during our in-depth conversations provided valuable insights on what payers think about PRO evidence, how they see it being used in the years to come, and what pharmaceutical companies should be doing about it. Their answers also dispelled some common myths on the subject.
The Ratings Game
To obtain input from individuals with varied viewpoints, we conducted one hour, double blind telephone interviews with a diverse group of payer executives. Optum was not identified as a purveyor of PRO products and services during the survey interviews.
The respondents consisted of four commercial plan executives, two accountable care organization representatives, two pharmacy benefit managers, two government officials, a self-insured employer, and an actuary. The survey was conducted in January and February of 2014.
During the survey, we asked the respondents to provide ratings from their perspective on three topics related to the use of PRO evidence both at the time of the survey and five years in the future:
1. The relevance of PRO evidence in making coverage decisions (1 = not relevant / 10 = highly relevant)
2. Whether the respondents would like to see more PRO evidence from pharmaceutical companies (1 = no interest / 10 = high interest)
3. Whether pharma companies should invest in creating PRO evidence to differentiate their drugs (1 = no interest / 10 = high interest)
 What We Found: Overall
What We Found: Overall
The chart shows the overall results from the survey. While the respondents felt the relevance of PRO evidence was on the low side when surveyed, they certainly didn’t think it was going to stay that way. Their ratings predict a 70% jump in relevance between the survey date and five years later. That point of view may very well be spurred by a changing health care environment created by the Affordable Care Act in which payment is being linked to achieving quality standards.
Looking at the second pair of questions, the payers expressed an interest in getting more PRO data right away, and their ratings indicate their interest is only going to get stronger in the coming years. The payers’ responses tell a similar story about how they feel about the value of pharma companies investing in PROs.
What We Found: By Payer
When we looked at the ratings of the respondents individually, we found that not all payers have the same opinions about the value of PROs for making coverage decisions about new drugs. PROs seem to have greater relevance for payers who have long-term responsibility for their population, like ACOs, the VA and self-insured employers. Relevance also seems to rise along with the amount of financial risk the organization assumes.
What We Found: By Disease Type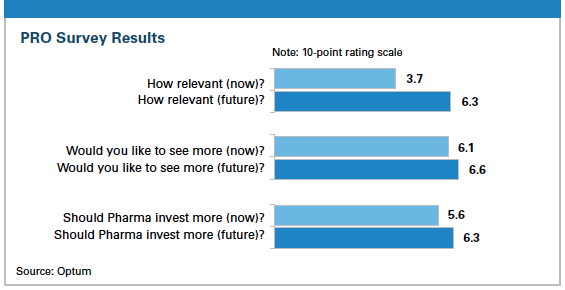
We also found that PRO relevance was stronger when organizations are making decisions about treatments for symptomatic diseases with few objective measures versus asymptomatic diseases. The same held true for chronic diseases when compared to acute diseases. That was surprising.
Myth Busters
Now, about those myths we referred to earlier. As we spoke to the executives, we heard some interesting things that upended three common beliefs regarding PROs and their usage along the pharma developmental timeline.
The first myth that was busted is that U.S. payers don’t care about PROs. It’s clear to us from our discussions that some payers are very attentive to PRO data and, in fact, want more of it.
The second myth is that PRO evidence is only important if it’s on the product label. While PRO-based labels are extremely valuable for product promotion, payers told us they will consider PRO results from pivotal studies whether or not the results are featured on the label.
 And, finally, there is the common belief that it’s OK to put off PRO data collection until late phase studies. What we heard is that the best approach for pharma companies is to collect and interpret the data in the pivotal studies since initial coverage decisions are based on those studies. PRO data from late phase studies isn’t considered.
And, finally, there is the common belief that it’s OK to put off PRO data collection until late phase studies. What we heard is that the best approach for pharma companies is to collect and interpret the data in the pivotal studies since initial coverage decisions are based on those studies. PRO data from late phase studies isn’t considered.
What It All Means
When creating a PRO strategy, it’s critical to keep payers in mind, especially those with a long-term view and more financial risk. That means gathering the kind of evidence they want to see and presenting it in a way they want to see it. For example, it’s important to use PROs to underscore the relationship between self-rated health and future medical costs. It’s also helpful to use standardized PROs that allow for a straightforward interpretation and direct comparisons.
During this enlightening exercise, we learned that payers value PRO evidence as a unique predictor of clinical and economic outcomes, and as a key element of performance and quality ratings. We also learned that payers need to be educated about how PRO tools work and how the data those tools produce can be used to make patient access and intervention decisions. For pharmaceutical companies, these realizations represent an opportunity to substantially improve the chances for product acceptance and marketing success by meeting the key data needs of payer decision makers.(PV)
Optum is a global team of approximately 80,000 people, working collaboratively across the health system to improve care delivery, quality, and cost-effectiveness by focusing on three key drivers of transformative change: engaging the consumer, aligning care delivery, and modernizing the health system infrastructure.
For more information, visit optum.com



















