 A BRIC nation and the largest economy in South America, Brazil is regarded by most industries as a key market. Starting in the mid-1990s the country entered a period of economic stabilization, followed by strong growth in the early 2000s, driven by the commodities sector.
A BRIC nation and the largest economy in South America, Brazil is regarded by most industries as a key market. Starting in the mid-1990s the country entered a period of economic stabilization, followed by strong growth in the early 2000s, driven by the commodities sector.
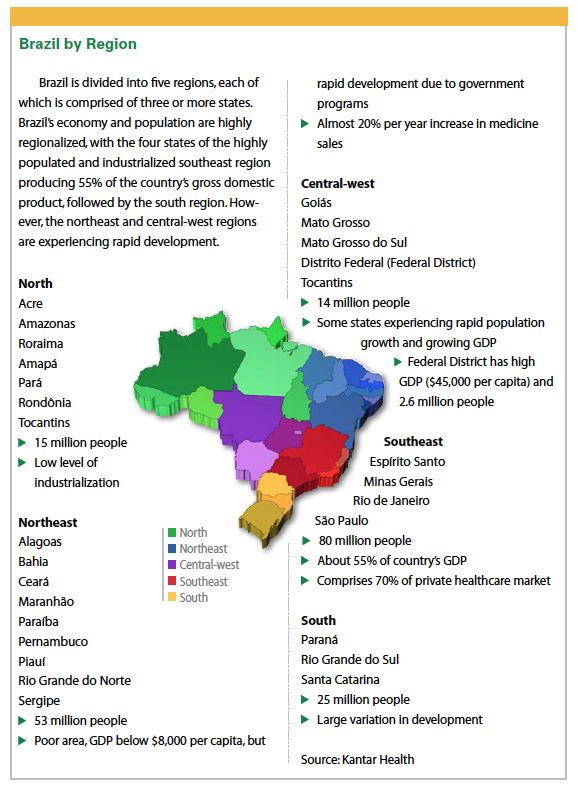
In addition, Brazil experienced significant population growth two to three decades ago that shifted the shape of the age pyramid. As a result of this population growth, Vitor Harada, VP and regional head, Latin America, site management for Quintiles, says Brazil’s age demographics have favored the economy for a number of years, although this has started to slow.
During 2015, the country experienced a turbulent economic and political environment, with fragmented attention to national needs, economic deficit, and rising inflation.
“The economy is in recession, public expenditure is expected to be reduced, and the local currency may be devalued," Mr. Harada says.
But he adds that while the next year or two may be bumpy from an economic standpoint, a return to stability and growth is expected once the government makes expected adjustments in public expenditure and policies.
The Pharmaceutical Sector
Brazil remains the sixth pharmaceutical market globally, and despite economic volatility, favorable growth in pharma sales is expected, says Tracy Francis, a director in McKinsey & Company’s São Paulo, Brazil, office.
According to McKinsey, although the double-digit growth rates the Brazilian pharma market enjoyed over the last decade have slowed, the period to 2020 is expected to deliver growth of around 7% to 10% per annum.
 Deloitte notes that while the country will remain high on the list of emerging markets being targeted by multinational patented-drug companies and international generic manufacturers, both segments will face tough challenges, including: complex fiscal and regulatory frameworks, restrictive pricing policies, and a strong local industry.
Deloitte notes that while the country will remain high on the list of emerging markets being targeted by multinational patented-drug companies and international generic manufacturers, both segments will face tough challenges, including: complex fiscal and regulatory frameworks, restrictive pricing policies, and a strong local industry.
Otávio Clark, M.D., Ph.D., CEO of Evidências-Kantar Health, says virtually all multinational pharmaceutical companies have offices in Brazil and are active in the market.
Experts say companies that are already established in the Brazilian market can leverage regular product launches and local government incentives to encourage growth and scale efficiently, which makes it attractive for many multinational companies.
“Recently an American company purchased one of the largest healthcare insurance operators in Brazil," Dr. Clark says. “Also, the government recently sanctioned a law allowing international companies to invest and even purchase private hospitals in the country. One large hospital in São Paulo has already been purchased. Besides that, many health insurance companies have been acquired by foreign companies."
“Recent changes in legislation and the devaluation of the Brazilian currency are also attracting investors to the healthcare industry, leading to consolidation of the hospital sector, as well as of distributors and pharma chains," Ms. Francis says.
R&D Opportunities
 R&D innovation has traditionally not been at the center of the Brazilian pharma strategy, however the government has been trying to encourage R&D with collaborations such as productive development partnerships (PDPs), for which a technology transfer is applied.
R&D innovation has traditionally not been at the center of the Brazilian pharma strategy, however the government has been trying to encourage R&D with collaborations such as productive development partnerships (PDPs), for which a technology transfer is applied.
Mr. Harada adds that R&D is at a turning point in Brazil.
“Brazilian companies that previously focused primarily on generics are now investing in innovative research – both through their own efforts and through partnerships with other global companies," he says.
In addition, Mr. Harada says the government has implemented incentives to progress biotechnology efforts in the country, which has encouraged local and multinational companies to partner with public laboratories to develop and produce biologic drugs in the country.
“The government also is investing," he says. “For example, there is a much-anticipated dengue vaccine in development that has received a lot of attention given its potential impact on the public health system."
Ms. Francis adds that biotech has been on the agenda of government priorities in local R&D, and since 2009, the government has been establishing public-private partnership deals with multinational manufacturers, local players, and public laboratories for the technology transfer of biologic products to local companies.
According to Dr. Clark, Brazil’s domestic industry remains focused on generics and biosimilars.
“There are at least three major Brazilian groups with large projects in biosimilar development, in association with both foreign and local industries, which are shaking up the market," he says. “Many biosimilar clinical studies are being designed and conducted in Brazil."
He says recently the first biosimilar — infliximab — was registered with ANVISA (Brazil’s national health authority). Infliximab is a monoclonal antibody for treating several chronic inflammatory diseases such as Crohn’s disease, rheumatoid arthritis, and psoriasis.
According to Associação Brasileira das Indústrias de Medicamentos Genéricos (Pró Genéricos), a Brazilian industry group for generics manufacturers, more than 20 best-selling pharmaceuticals lost patent protection in Brazil in 2012 and 2013, which boosted the generics market by more than $400 million. By 2018, the volume market share for generics is expected to exceed 33%.
In terms of R&D spending, most of the investment is private, but the National Development Bank (BNDES) has funds leveraged for projects in the pharmaceutical area, so there is capital available at a competitive interest rate, Mr. Harada adds.
A Healthcare Market Assessment
 Brazil has a universal and free health system guaranteed in its federal constitution. In addition, many Brazilians pay for additional private health insurance, Mr. Harada says.
Brazil has a universal and free health system guaranteed in its federal constitution. In addition, many Brazilians pay for additional private health insurance, Mr. Harada says.
According to Dr. Clark, the private healthcare market accounts for 25% of the population while the public healthcare system accounts for 75% of the population.
One of the biggest challenges is that incorporation of new technologies in the public system has been difficult and slow, mainly due to the lack of government budget, Dr. Clark says.
All new technologies are assessed by a government agency known as CONITEC (National Commission on Technology Incorporation in the Public HealthCare System) before they can be incorporated into the public healthcare system. However, this agency has been almost invariably slow to analyze new treatments, and its lack of clear decision criteria has been subject to criticism by pharmaceutical companies and doctors alike.
“The root of this problem is that the government invests too little in health, less than 3.5% of the gross domestic product (GDP), which is less than many neighboring countries and there is no foreseeable change in this scenario in the coming years," Dr. Clark says.
However, Dr. Clark adds that the public healthcare system has some high-quality programs.
“For example, we have the latest technologies available to treat hepatitis," he says. “The vaccine market is also expanding: the vaccine to prevent cervical cancer is distributed for free here."
According to Ms. Francis, the public healthcare budget has suffered some cuts — estimated in BRL 3.8 billion in 2016 — including priority government programs such as Farmácia Popular and the high- and medium-complexity diseases. The 2016 budget is expected to focus on the sustainability of SUS (universal government coverage for all Brazilians), especially in primary care and the supply of drugs through other pharmaceutical assistance programs.
“Demographic shifts remain the major driver in healthcare spend," she says. “The population is getting older and living longer, contributing to changes in the epidemiologic profile, with higher prevalence of chronic diseases."
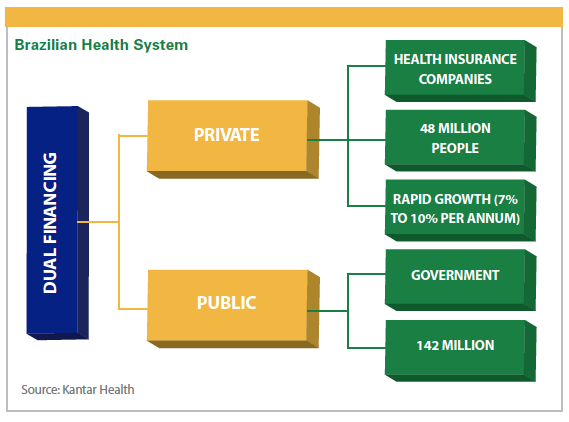 The private healthcare market, meanwhile, has grown significantly in the past two decades, from 20 million people with private health insurance in 1995 to 50 million people in 2015, Dr. Clark says.
The private healthcare market, meanwhile, has grown significantly in the past two decades, from 20 million people with private health insurance in 1995 to 50 million people in 2015, Dr. Clark says.
Mr. Harada says Brazil’s private health insurance plans are tightly regulated, guaranteeing a minimum coverage and quality, depending on the indication and types of illness.
Regulatory Perspective
Brazil’s regulatory health authority, ANVISA, publishes guidelines to help ensure compliance and understanding of their rules since phone and e-mail communications are not accepted by the agency. Good knowledge of the regulatory environment and experience is crucial for a successful registration approval.
According to Interfarma, the Brazilian research-based pharmaceutical manufacturers association, ANVISA took an average of 1,000 days for a generic registration and 500 days for an innovative product registration in 2014.
It is not mandatory that clinical studies be conducted with Brazilian patients; studies performed abroad are acceptable. For new drug applications (NDAs), however, it is necessary to submit Phase II and III studies showing efficacy and safety of the medicine. There is no simplified process in Brazil for NDA approval even if the medicine is already approved by the U.S. FDA or the European Medicines Agency.
While there can be challenges in getting a product established on the Brazilian market, experts say ANVISA has been seeking to work more closely with the pharmaceutical industry to improve approval timelines and procedures. In addition, new regulations are being reviewed to be more harmonized with other health authorities.
Pharmaceutical companies investing in Brazil will need to consider a granular go-to-market approach, Ms. Francis says, who adds that getting products registered is often challenging.
“Best-in-class market access is critical, as the government is not keen to register new high-cost therapies," she says. “This situation ties in with a clinical trial strategy to get local stakeholders on board, access to public formulary, which requires significant knowledge of stakeholders involved in the process, as well as preparation of robust data for the regulators."
Mr. Harada emphasizes the importance of understanding the local market.
“Those who know the Brazil market at a local level understand that although volatility may bring some uncertainty, many times it also brings opportunity," he says. (PV)



















