Gene Therapy for Better Sight
Kathy High, M.D., President and Chief Scientific Officer at Spark Therapeutics, talks about the company’s gene therapy for inherited retinal diseases.
 Thanks to a renewed focus on gene therapy, researchers are uncovering new ways to treat diseases where no therapies exist. (See September 2016, PharmaVOICE.)
Thanks to a renewed focus on gene therapy, researchers are uncovering new ways to treat diseases where no therapies exist. (See September 2016, PharmaVOICE.)
Spark Therapeutics has built a gene therapy platform that strives to turn genes into medicines for patients with inherited diseases, including inherited retinal diseases (IRD), neurodegenerative diseases, as well as diseases that can be addressed by targeting the liver. The company was founded in October 2013 as a result of the technology created at the Children’s Hospital of Philadelphia.
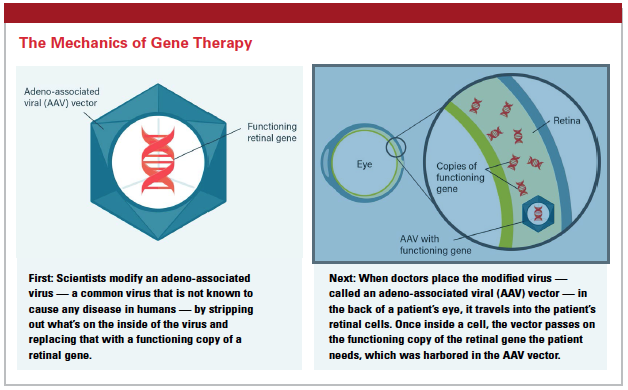
The company’s most advanced program is voretigene neparvovec, an adeno-associated viral gene therapy for IRD caused by biallelic RPE65 mutations.
“Adeno-associated viral vectors are engineered from a very small, naturally occurring virus," says Kathy High, M.D., president and chief scientific officer. “The viral coding sequences are removed and replaced with the therapeutic gene of interest. The end result is a recombinant particle. On the outside it looks like a virus, and on the inside it contains the therapeutic piece of DNA under the control of an appropriate promoter. In this case, we’re using the outside of the virus to bind to a cell and introduce its DNA into the cell, and then that piece of DNA employs the cellular machinery to make the missing protein."
For the Phase III study of voretigene for blindness, the therapy was administered in a surgical procedure in the operating room by a retinal surgeon, and the vector is injected into the retina.
The Food and Drug Administration has granted the company orphan drug and breakthrough designation for voretigene, while the European Medicines Agency (EMA) also has granted orphan product designation.
Spark has initiated a rolling submission of the biologics license application (BLA), which is expected to be completed in early 2017. Dr. High says the company plans to file voretigene with European regulators sometime this year.
The Phase III study of voretigene measured patients’ ability to do a mobility test at a range of light levels, from 400 lux to 1 lux. Lux is a unit of measure of luminosity. All the individuals in the intervention group improved by an average of about two light levels.
 The secondary endpoints included full field light sensitivity, and the people in the intervention group improved by about two logs in terms of their light sensitivity, whereas the people in the control group didn’t change. Patients in the intervention arm also saw improvements in the visual fields; their visual fields went from about 350 sum total degrees, to about 650 sum total degrees. The people in the control group had very little change in the visual fields.
The secondary endpoints included full field light sensitivity, and the people in the intervention group improved by about two logs in terms of their light sensitivity, whereas the people in the control group didn’t change. Patients in the intervention arm also saw improvements in the visual fields; their visual fields went from about 350 sum total degrees, to about 650 sum total degrees. The people in the control group had very little change in the visual fields.
The study contained 20 patients in the intervention arm and nine patients in the control arm.Dr. High notes that after the one-year evaluation, patients in the control arm crossed over to the intervention arm. The only side effects, she says, were related to the procedure and included inflammation and redness.
“The Phase I study involved dose escalation," she says. “We started at a low dose, and then when that was deemed safe, we moved to the next dose. We only injected one eye, in the interest of safety. When the right dose was determined, we did a second Phase I study where we injected the contralateral eye of each of those people. The retina is a relatively immune privileged site, but we nonetheless gave a course of steroids in the perioperative period to reduce any risk of an immune response."
Dr. High points out that in the manufacturing of the vector, the company removes empty capsids to help reduce immune response.
Spark Therapeutics is also conducting a dose-escalating Phase I/II trial to assess safety and efficacy of subretinal administration SPK-7001 for treating choroideremia (CHM). This is an X-linked IRD that usually affects boys during childhood as night blindness and a reduction of visual field.
The company is also conducting preclinical work on two other retinal diseases. Spark is developing RhoNova, an investigational gene therapy for rhodopsin-linked autosomal dominant retinitis pigmentosa (RHO-adRP), an IRD that also leads to visual impairment and in the most severe cases, to blindness. Spark is investigating a potential gene therapy for LHON, an IRD that can cause sudden, severe loss of central vision.
Additionally, Spark has a program using an AAV vector in hemophilia B. In January, the company achieved a pre-specified safety and efficacy profile development milestone in the ongoing hemophilia B Phase I/II trial of SPK-9001, earning it a $15 million payment from development partner Pfizer.
SPK-9001 is a novel bio-engineered adeno-associated virus (AAV) capsid expressing a codon-optimized, high-activity human factor IX variant enabling endogenous production of factor IX. SPK-9001 has received breakthrough therapy and orphan product designations from the FDA.
Finally, Spark initiated a Phase I/II clinical trial of SPK-8011 for Hemophilia A in late 2016. Spark retains global commercialization rights to SPK-8011 for hemophilia A. (PV)


















