A New Targeted Cancer Treatment
 Dr. Joshua Allen, VP for Research and Development at Oncoceutics, talks about his company’s approach to target cancer.
Dr. Joshua Allen, VP for Research and Development at Oncoceutics, talks about his company’s approach to target cancer.
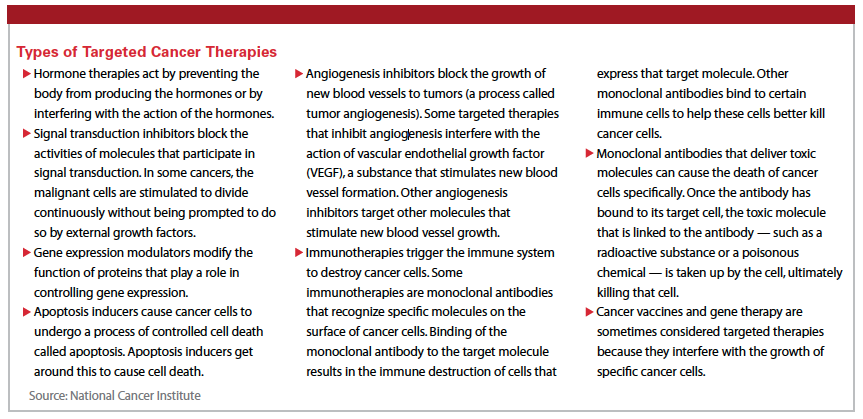
Targeted therapies are the focus of many cancer drug development programs. They are a cornerstone of precision medicine, which uses information about a person’s genes and proteins to prevent, diagnose, and treat disease. Targeted therapies aim to take advantage of the small DNA differences between cancer and normal cells.
These gene changes might cause the cell to make too much of a certain protein, which in turn might make the cell grow and divide too quickly.
Oncoceutics is developing a new approach to targeted therapies. The company’s lead product is ONC201, a first-in-class small molecule that aims to exploit cells’ response to stress.
“Unlike chemotherapy, our drug does not rely on the division rate of tumor cells; it uses a new way to kill tumor cells that has not been exploited before," says Joshua Allen, Ph.D., VP for research and development at Oncoceutics. “It takes advantage of the integrated stress response pathway."
Dr. Allen says ONC201 is able to trigger a response in tumor cells that makes them die but doesn’t affect normal cells.
“Tumor cells divide quickly so everything that is needed for cell division is in overdrive," he says. “If we can add a specific type of stress into that cell, this basically tips the cell over the edge into cell death mode. The molecular pathways engaged by the drug to accomplish this are collectively called the integrated stress response. All cells have this mechanism, but normal cells can cope with stress better since they have lower resting levels of stress. The higher the levels of stress in a cell, the more likely the cell is to die when additional stress is added."
 ONC201 is in Phase II trials for the treatment of several cancers, including glioblastoma, solid tumors, multiple myeloma, non-Hodgkin’s lymphoma, and leukemia. These trials will enroll more than 200 patients. The company recently completed a successful Phase I study in solid tumors. Dr. Allen says depending on how these trials go, the company could enter pivotal trials as soon as 2017.
ONC201 is in Phase II trials for the treatment of several cancers, including glioblastoma, solid tumors, multiple myeloma, non-Hodgkin’s lymphoma, and leukemia. These trials will enroll more than 200 patients. The company recently completed a successful Phase I study in solid tumors. Dr. Allen says depending on how these trials go, the company could enter pivotal trials as soon as 2017.
Dr. Allen adds that preclinical data suggest that the drug has broad applicability across cancers that no longer respond to available treatment options, as well very different types of cancer ranging from blood cancers to brain cancers.
The company has several R&D alliance agreements with leading comprehensive cancer centers, including The University of Texas MD Anderson Cancer Center and the Fox Chase Cancer Center.
Oncoceutics is researching several other compounds for development that build on its flagship product; one is a chemical analogue of ONC201 called ONC212, which appears to work in melanoma.
Oncoceutics is not a traditionally funded venture capital company; it combines private investment with non-dilutive grants and alliances to fund its research.
“We’ve been able to research not only on our first compound, but also research a whole family of related, new compounds through a grant from the National Cancer Institute, so they have funded both pipeline expansion, as well as a brain tumor trial at Massachusetts General Hospital and Dana Farber Cancer Institute," Dr. Allen says.
Additionally, he says, the company has a unique risk-sharing alliance agreement with the MD Anderson Cancer Center.
“This is the first risk-sharing structure of its kind at MD Anderson," Dr. Allen says. “This nontraditional alliance enabled Oncoceutics and MD Anderson to share the risk and potential commercialization of ONC201 in two clinical trials, with MD Anderson receiving a royalty on any eventual sales in lieu of the usual payments for conducting clinical trials."
Dr. Allen says as the company progresses though clinical development, there is interest in collaborations with pharmaceutical companies. (PV)













